It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
reply to post by Phage
So by modifying the vegetation of the world is it having an impact on the atmosphere? Example: Planting more corn for ethynol can effect the
atmosphere it there is lots of it?
reply to post by Phage
Yeah, well, this isn't:
www.ecd.bnl.gov...
It seems as though "forcing" is being used to offset the greenhouse gases from industrial plants. As it states, natural aerosols disperse evenly, while forced aerosols don't. This seems to be a problem that they're currently trying to figure out how to fix.
Yeah, well, this isn't:
www.ecd.bnl.gov...
In several studies we have tried to provide estimates of the uncertainty budget associated with the aerosol forcing. Much of the uncertainty arises from the fact that unlike the long-lived greenhouse gases, whose concentrations are rather uniformly distributed in the atmosphere, the loadings of aerosols are highly variable in space and time, as a consequence of highly localized sources and of sporadic removal, mainly by precipitation. Additionally aerosol microphysical properties are not a universal constant, but depend on sources and composition and evolve as a consequence of chemical and physical processes occurring in the atmosphere. The mass loading, composition, and the microphysical properties of aerosols such as number concentration and size distribution directly affect their direct and indirect radiative forcing of climate.
Reducing the uncertainty in aerosol forcing will require a major effort both in characterizing the present distribution and properties of aerosols and in developing understanding required to represent the processes controlling loading and properties of tropospheric aerosols in numerical models. Model-based descriptions of aerosol forcing need to be incorporated into climate models in order to represent this forcing not just for the present climate but also retrospectively over the industrial period and prospectively for various scenarios of future emissions. Much of our research is directed to developing and evaluating numerical models for representing the geographical distribution of loading of atmospheric aerosols. Our approach has been to use observationally derived meteorological data to drive our models, because the temporal and spatial variation in aerosol loading is governed to great extent by meteorological variability. Meaningful evaluation of the model by comparison with observations thus requires this approach. Much of this work is conducted within the Department of Energy's Atmospheric Science Program (ASP).
An additional major component of our research is directed to developing improved representation of aerosol optical properties and radiative forcing. Much of this work is conducted in conjunction with the Department of Energy's Atmospheric Radiation Measurement (ARM) Program.
Our work is represented in our publications. I welcome inquiries of interest from any and all. Much of our work is conducted in collaboration with others at their institutions or as visiting scientists at Brookhaven National Laboratory. I particularly encourage inquiries from students; you are our future.
Atmospheric Heating and Cooling from Fossil-Fuel Combustion. Our examination of the greenhouse heating influence of fossil fuel combustion versus the aerosol cooling influence was highlighted in the Fall 1994 Newsletter of the DOE Carbon Dioxide Information and Analysis Center CDIAC Communications.
It seems as though "forcing" is being used to offset the greenhouse gases from industrial plants. As it states, natural aerosols disperse evenly, while forced aerosols don't. This seems to be a problem that they're currently trying to figure out how to fix.
reply to post by rickymouse
Is more corn being planted, or is it that more corn is being used for ethanol?
Or maybe cutting down rainforests is a good way of preventing climate change?
What does any of this have to do with "chemtrails making us sick?"
Is more corn being planted, or is it that more corn is being used for ethanol?
Or maybe cutting down rainforests is a good way of preventing climate change?
What does any of this have to do with "chemtrails making us sick?"
reply to post by rickymouse
Visit the link and check out the charts and graphs they have. You may understand it better. I'm still digging around myself to try to gain a more thorough understanding about what this all entails.
Visit the link and check out the charts and graphs they have. You may understand it better. I'm still digging around myself to try to gain a more thorough understanding about what this all entails.
My main problem with the chemtrail theory is that, if it was being sprayed by TPTB, wouldn't the chemicals be affected them too?
Originally posted by Afterthought
It seems as though "forcing" is being used to offset the greenhouse gases from industrial plants. As it states, natural aerosols disperse evenly, while forced aerosols don't. This seems to be a problem that they're currently trying to figure out how to fix.
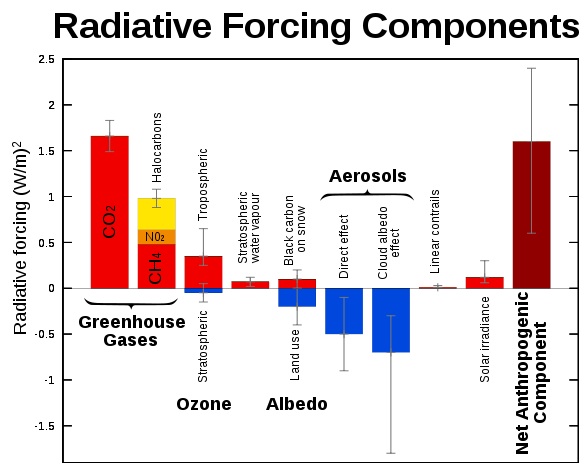
"forcing" is a just the difference between incoming and outgoing radiation. It's a technical term, a measurement. There is no such things as a "forced aerosol"
en.wikipedia.org...
In climate science, radiative forcing is generally defined as the change in net irradiance between different layers of the atmosphere. Typically, radiative forcing is quantified at the tropopause in units of watts per square meter. A positive forcing (more incoming energy) tends to warm the system, while a negative forcing (more outgoing energy) tends to cool it. Sources of radiative forcing include changes in insolation (incident solar radiation) and in concentrations of radiatively active gases and aerosols.
There are many sources of forcing.
edit on 30-1-2012 by Uncinus because: (no reason given)
edit on 30-1-2012 by Uncinus because: (no
reason given)
A bit off topic perhaps, but anybody notice how the OP hasn't returned to this thread after his first (and only) unprovable post?
edit on 1/30/2012 by Chamberf=6 because: add
Originally posted by Afterthought
As it states, natural aerosols disperse evenly, while forced aerosols don't. This seems to be a problem that they're currently trying to figure out how to fix.
And that's not what it states at all. It states:
Much of the uncertainty arises from the fact that unlike the long-lived greenhouse gases, whose concentrations are rather uniformly distributed in the atmosphere, the loadings of aerosols are highly variable in space and time, as a consequence of highly localized sources and of sporadic removal, mainly by precipitation.
So greenhouse gases are dispersed evenly, but aerosols (both natural and anthropogenic) are not.
reply to post by Uncinus
It sounds like it seemed like a good idea but concentrating it instead of natural diversity doesn't have the same effect. Going back to small
spread out farms with diverse planting may be the best option. In areas with extreme CO2 emissions they could use a little more forests or possibly
plantlife that takes up the CO2. I read an article about putting big parks near cities to clean the air but it was labor intensive/expensive to
manage them. I thought trees grew without our help. Do we have to start paying the trees?
edit on 30-1-2012 by rickymouse because: (no reason given)
Originally posted by nuttin4U
Originally posted by nuttin4U
These days...with so much information...the truth is hard to come by. The people that say they're just 'contrails' haven't seen what i've seen, over the past 2 years. I know the difference between 'contrail' and a 'chemtrail'. Unless, a 'contrail' is really a 'chemtrail' and a 'chemtrail' is really a 'contrail'...and we just got the names confused. But, whatever you want to call them..they're UNDENIABLE. It's hard to see how a little bit of exhaust can cause a DRAMATIC change in weather. You can see the streak GROW, throughout the day. It starts like a LOOOOOOOOOOONG line, then starts to expand and get puffy; you can even see the 'clouds' drifting. And guess where they're drifting into: the Pacific Ocean. And you want me to believe it's a 'contrail' that caused it? Hmm, maybe it is.
Ok then, let's let the people decide.
Is this a contrail?
What is this, then?
And how about this?
Or this?
What about this?
So, this is normal?
And this?
Ewwww, that's purty.
Wow, niiiiiiice
I'm loving the way it drifts INTO the ocean.
These pics are old pics...the ones i had available online. I have MUCH better ones, than this. I took some of today's activity...but...why bother loading them...they look the same as these. The only difference today, was the fact that the planes were flying relatively close to each other. A little too close, to be passenger planes. That's for SURE!edit on 29-1-2012 by nuttin4U because: (no reason given)
Can we talk about my pretty pics??? Please??? I need a cloud edumacation. Where are the 'experts'? ProudBird, Unicus...where art thou?
I'm still waiting for 1 of the infamous 'contrail' theorists to explain these pics. They're all from the same day. So, what say you? These are all 'NORMAL', right? Nothing more than 'contrails'?
woke up to a sky full of trails this morning in west texas. Still lingering in the air, took photos but cant seem to post.
edit on 30-1-2012
by AHUGE1 because: (no reason given)
Originally posted by Uncinus
Originally posted by Afterthought
As it states, natural aerosols disperse evenly, while forced aerosols don't. This seems to be a problem that they're currently trying to figure out how to fix.
And that's not what it states at all. It states:
Much of the uncertainty arises from the fact that unlike the long-lived greenhouse gases, whose concentrations are rather uniformly distributed in the atmosphere, the loadings of aerosols are highly variable in space and time, as a consequence of highly localized sources and of sporadic removal, mainly by precipitation.
So greenhouse gases are dispersed evenly, but aerosols (both natural and anthropogenic) are not.
Right. Anthropogenic aerosols.
Anthropogenic means caused by humans.
www.answers.com...
www.ecd.bnl.gov...
Over the course of decades, adding carbon dioxide to the atmosphere will lead to an enhanced greenhouse
effect by trapping additional heat in the lower atmosphere, leading to increases in temperature. On the other hand, energy by-products in the form of aerosols can reflect incoming sunlight and also make clouds more reflective and longer lasting, resulting in atmospheric cooling on a much shorter timescale. Offsetting
factors like these must be accounted for in any effective climate model.
Scientists at Brookhaven National Laboratory model anthropogenic sulfate aerosol emissions to understand their potential to increase atmospheric reflectance and modify global radiative heat balance.
The ultimate objective? To furnish policymakers with predictive models of sufficient detail and reliability to develop local, regional, and national policies and plans with confidence.
reply to post by Phage
I figure that once we can prove that they are spraying aerosols into the atmosphere and why, then we can look at what types of chemicals and substances they are using in these aerosols that may be causing illnesses.
What does any of this have to do with "chemtrails making us sick?"
I figure that once we can prove that they are spraying aerosols into the atmosphere and why, then we can look at what types of chemicals and substances they are using in these aerosols that may be causing illnesses.
reply to post by Afterthought
Look.....let's show how those who keep banging on about so-called "chemtrails" will take any ridiculous claim that they find written on the Internet, and trot it out as "truth"....whilst completely ignoring actual science, and the explanations from those individuals here who actually DO have the knowledge and life experience to know crap from science
In your post on page back, you linked to this site:
From that, were some excerpts. THIS was first (and it is almost completley wrong):
(I say "almost", because they get this part correct, at least: "Normal contrails are formed above 30,000 feet..." The rest is a load of hogwash).
"...and last only a few seconds."
LIE. Well, a half-truth, so give them one more fraction of a point. But, the innuendo there is clear....they are lying if they mean to imply that "all" contrails only last "a few seconds".
"They are created rarely and are caused by extreme heat."
AGAIN, a mixed bag of LIE and fact. "rarely" is a LIE....."extreme heat" is descriptively somewhat correct, if inexact and a poor choice of phrase, in terms of a scientific explanation.
But, this is where it goes out intl La La Land:
"Designed to appear as normal contrails, chemtrails are formed between 12,000 and 20,000 feet altitude...."
Utter rubbish, and more LIE. Now, they just make crap up to suit their agenda.
And, this is the funniest of them all. It shows the level of intellectual prowess that they (do not) possess:
"...and come from spray nosels that are attached to chemtrail planes: airplanes that spray."
What else is there to say? Those who know better, can understand that this source is worthless as a factual presenter of information on the topic. "nosels"....? Really?
In any case, as has been pleaded many times...IF these "chemtrail planes".....planes with "nosels" attached to "spray" exist....WHERE is the evidence, and WHERE are the photos? The eyewitnesses to the airplanes on the ground, or in flight at low altitude, where they are clearly visible....takeoff and landing situations.
Where are the "whistleblowers"?
Just one verifiable and credible whistleblower? Just one?? After about 15 years, where are they?
Look.....let's show how those who keep banging on about so-called "chemtrails" will take any ridiculous claim that they find written on the Internet, and trot it out as "truth"....whilst completely ignoring actual science, and the explanations from those individuals here who actually DO have the knowledge and life experience to know crap from science
In your post on page back, you linked to this site:
www.thebestofrawfood.com...
From that, were some excerpts. THIS was first (and it is almost completley wrong):
What are chemtrails?
Normal contrails are formed above 30,000 feet and last only a few seconds. They are created rarely and are caused by extreme heat. Designed to appear as normal contrails, chemtrails are formed between 12,000 and 20,000 feet altitude and come from spray nosels that are attached to chemtrail planes: airplanes that spray.
(I say "almost", because they get this part correct, at least: "Normal contrails are formed above 30,000 feet..." The rest is a load of hogwash).
"...and last only a few seconds."
LIE. Well, a half-truth, so give them one more fraction of a point. But, the innuendo there is clear....they are lying if they mean to imply that "all" contrails only last "a few seconds".
"They are created rarely and are caused by extreme heat."
AGAIN, a mixed bag of LIE and fact. "rarely" is a LIE....."extreme heat" is descriptively somewhat correct, if inexact and a poor choice of phrase, in terms of a scientific explanation.
But, this is where it goes out intl La La Land:
"Designed to appear as normal contrails, chemtrails are formed between 12,000 and 20,000 feet altitude...."
Utter rubbish, and more LIE. Now, they just make crap up to suit their agenda.
And, this is the funniest of them all. It shows the level of intellectual prowess that they (do not) possess:
"...and come from spray nosels that are attached to chemtrail planes: airplanes that spray."
What else is there to say? Those who know better, can understand that this source is worthless as a factual presenter of information on the topic. "nosels"....? Really?
In any case, as has been pleaded many times...IF these "chemtrail planes".....planes with "nosels" attached to "spray" exist....WHERE is the evidence, and WHERE are the photos? The eyewitnesses to the airplanes on the ground, or in flight at low altitude, where they are clearly visible....takeoff and landing situations.
Where are the "whistleblowers"?
Just one verifiable and credible whistleblower? Just one?? After about 15 years, where are they?
edit on Mon 30 January 2012 by ProudBird because: (no reason given)
reply to post by nuttin4U
I've seen where there's three planes wide and expanding exhaust gasses that turn to clouds around here occasionally. I haven't a clue what they are trying to do. Most people never look up to see what's going on and without opening their eyes deny that things happen. I don't know if what they are doing is bad for us. I just hope someone doing this has tested it out well for unforseen problems. I live in the UP of Michigan, there aren't many people here so they could test things like this up here with less casualties. Problem is many people here look into the sky and see these things and wonder. If something is found to be negative we will take action as a whole to sue the government. They should test these things where people do not pay attention if they are smart.
I've seen where there's three planes wide and expanding exhaust gasses that turn to clouds around here occasionally. I haven't a clue what they are trying to do. Most people never look up to see what's going on and without opening their eyes deny that things happen. I don't know if what they are doing is bad for us. I just hope someone doing this has tested it out well for unforseen problems. I live in the UP of Michigan, there aren't many people here so they could test things like this up here with less casualties. Problem is many people here look into the sky and see these things and wonder. If something is found to be negative we will take action as a whole to sue the government. They should test these things where people do not pay attention if they are smart.
Originally posted by ProudBird
reply to post by Afterthought
Look.....let's show how those who keep banging on about so-called "chemtrails" will take any ridiculous claim that they find written on the Internet, and trot it out as "truth"....whilst completely ignoring actual science, and the explanations from those individuals here who actually DO have the knowledge and life experience to know crap from science
In your post on page back, you linked to this site:
www.thebestofrawfood.com...
From that, were some excerpts. THIS was first (and it is almost completley wrong):
What are chemtrails?
Normal contrails are formed above 30,000 feet and last only a few seconds. They are created rarely and are caused by extreme heat. Designed to appear as normal contrails, chemtrails are formed between 12,000 and 20,000 feet altitude and come from spray nosels that are attached to chemtrail planes: airplanes that spray.
(I say "almost", because they get this part correct, at least: "Normal contrails are formed above 30,000 feet..." The rest is a load of hogwash).
"...and last only a few seconds."
LIE. Well, a half-truth, so give them one more fraction of a point. But, the innuendo there is clear....they are lying if they mean to imply that "all" contrails only last "a few seconds".
"They are created rarely and are caused by extreme heat."
AGAIN, a mixed bag of LIE and fact. "rarely" is a LIE....."extreme heat" is descriptively somewhat correct, if inexact and a poor choice of phrase, in terms of a scientific explanation.
But, this is where it goes out intl La La Land:
"Designed to appear as normal contrails, chemtrails are formed between 12,000 and 20,000 feet altitude...."
Utter rubbish, and more LIE. Now, they just make crap up to suit their agenda.
And, this is the funniest of them all. It shows the level of intellectual prowess that they (do not) possess:
"...and come from spray nosels that are attached to chemtrail planes: airplanes that spray."
What else is there to say? Those who know better, can understand that this source is worthless as a factual presenter of information on the topic. "nosels"....? Really?
In any case, as has been pleaded many times...IF these "chemtrail planes".....planes with "nosels" attached to "spray" exist....WHERE is the evidence, and WHERE are the photos? The eyewitnesses to the airplanes on the ground, or in flight at low altitude, where they are clearly visible....takeoff and landing situations.
Where are the "whistleblowers"?
Just one verifiable and credible whistleblower? Just one?? After about 15 years, where are they?
edit on Mon 30 January 2012 by ProudBird because: (no reason given)
All you have, ProudBird, is quick answers and rebuttles. You still have yet to answer my questions...regarding my pics. Don't act like they don't exist. Those pics all depict 'normal' contrails....right???? Come on....rebuttle them.
reply to post by Gmoneycricket
Of course they add certain substances to gasoline and jet fuel. They do it to lower emissions, to prevent freezing of the fuel etc. So yes, there are added chemicals coming out the exhaust of internal combustion engines. You seem to be inferring that the purpose of those additives is nefarious, without ANY evidence. Saying jet aircraft are spraying "chemtrails" out the exhaust because of anti gel and antifreeze additives in the fuel is beyond illogical and blatantly intellectually dishonest.
Of course they add certain substances to gasoline and jet fuel. They do it to lower emissions, to prevent freezing of the fuel etc. So yes, there are added chemicals coming out the exhaust of internal combustion engines. You seem to be inferring that the purpose of those additives is nefarious, without ANY evidence. Saying jet aircraft are spraying "chemtrails" out the exhaust because of anti gel and antifreeze additives in the fuel is beyond illogical and blatantly intellectually dishonest.
Too bad I need a login in order to read the link that's provided here:
www.ecd.bnl.gov...
So, SO2 and sulfate can be caused by burning fuels as well as natural occurances such as volcanos.
www.ifc.org...$FILE/HandbookSulfurOxides.pdf
So, seeing as aerosols can be naturally caused as well as manufactured from burning fuel, I don't see it all that difficult to believe that sulfates are being distributed into the atmosphere. Considering the previous information I posted, they are in fact trying to figure out how to immitate clouds in order to cool the surface of the Earth. Combine these two "projects" and you have the "tip of the iceberg" evidence for the creation of chemtrails, why they're creating them, and how people are getting sick.
www.ecd.bnl.gov...
Dynamical influences on the distribution and loading of SO2 and sulfate over North America, the North Atlantic and Europe in April 1987. Benkovitz C. M., Miller M. A., Schwartz S. E. and Kwon O-U. Geochem. Geophys. Geosyst. 2, Paper no. 2000GC000129 (2001). www.agu.org...
So, SO2 and sulfate can be caused by burning fuels as well as natural occurances such as volcanos.
www.ifc.org...$FILE/HandbookSulfurOxides.pdf
Periodic episodes of very high concentrations of sulfur dioxide are believed to cause most of the
health and vegetation damage attributable to sulfur emissions. Depending on wind, temperature, humidity, and topography, sulfur dioxide can concentrate concentrate close to ground level.
Exposure to sulfur dioxide in the ambient air has been associated with reduced lung function, increased
incidence of respiratory symptoms and diseases, irritation of the eyes, nose, and throat, and
premature mortality. Children, the elderly, and those already suffering from respiratory ailments,
such as asthmatics, are especially at risk. Health impacts appear to be linked especially to brief exposures
to ambient concentrations above 1,000 μg/ m3 (acute exposures measured over 10 minutes).
Some epidemiologic studies, however, have shown an association between relatively low annual mean
levels and excess mortality. It is not clear whether long-term effects are related simply to annual
mean values or to repeated exposures to peak values. Health effects attributed to sulfur oxides are due
to exposure to sulfur dioxide, sulfate aerosols, and sulfur dioxide adsorbed onto particulate matter.
Alone, sulfur dioxide will dissolve in the watery fluids of the upper respiratory system and be absorbed into the bloodstream. Sulfur dioxide reacts with other substances in the atmosphere to form sulfate aerosols. Since most
sulfate aerosols are part of PM2.5 (fine particulate matter, with an aerodynamic diameter of less
than 2.5 microns), they may have an important role in the health impacts associated with fine
particulates. However, sulfate aerosols can be transported long distances through the atmosphere
before deposition occurs. Average sulfate aerosol concentrations are about 40% of average fine particulate
levels in regions where fuels with high sulfur content are commonly used. Sulfur dioxide adsorbed on particles can be carried deep into the pulmonary system. Therefore, reducing concentrations of particulate matter may also reduce the health impacts of sulfur dioxide. Acid aerosols affect respiratory and sensory functions.
So, seeing as aerosols can be naturally caused as well as manufactured from burning fuel, I don't see it all that difficult to believe that sulfates are being distributed into the atmosphere. Considering the previous information I posted, they are in fact trying to figure out how to immitate clouds in order to cool the surface of the Earth. Combine these two "projects" and you have the "tip of the iceberg" evidence for the creation of chemtrails, why they're creating them, and how people are getting sick.
edit on 30-1-2012 by Afterthought because: (no reason given)
reply to post by nuttin4U
He probably wont acknowledge them, or if he does he will come up with a certain set of circumstances where contrails become 'persisting contrails' and then go on to explain how the persistent permanence theory dictates that in very rare but actually very common conditions its possible for these contrails to yadda yadda, you get the point. He's done it to me as well, and you're dead on in that he is all quick replies that specialize in shrugging off the questions usually. Good pics, keep it up brother, cheers
He probably wont acknowledge them, or if he does he will come up with a certain set of circumstances where contrails become 'persisting contrails' and then go on to explain how the persistent permanence theory dictates that in very rare but actually very common conditions its possible for these contrails to yadda yadda, you get the point. He's done it to me as well, and you're dead on in that he is all quick replies that specialize in shrugging off the questions usually. Good pics, keep it up brother, cheers
new topics
-
Shocking Number of Voters are Open to Committing Election Fraud
US Political Madness: 6 minutes ago -
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections: 52 minutes ago -
Falkville Robot-Man
Aliens and UFOs: 1 hours ago -
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 1 hours ago -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 2 hours ago -
Ireland VS Globalists
Social Issues and Civil Unrest: 3 hours ago -
Biden "Happy To Debate Trump"
Mainstream News: 3 hours ago -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 3 hours ago -
What is the white pill?
Philosophy and Metaphysics: 5 hours ago -
Mike Pinder The Moody Blues R.I.P.
Music: 5 hours ago
top topics
-
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 13 hours ago, 21 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 5 hours ago, 7 flags -
Biden "Happy To Debate Trump"
Mainstream News: 3 hours ago, 7 flags -
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 1 hours ago, 5 flags -
What is the white pill?
Philosophy and Metaphysics: 5 hours ago, 5 flags -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 2 hours ago, 5 flags -
Ireland VS Globalists
Social Issues and Civil Unrest: 3 hours ago, 4 flags -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 3 hours ago, 4 flags -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 9 hours ago, 3 flags -
Falkville Robot-Man
Aliens and UFOs: 1 hours ago, 1 flags
active topics
-
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections • 19 • : FlyersFan -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works • 26 • : RussianTroll -
Shocking Number of Voters are Open to Committing Election Fraud
US Political Madness • 0 • : FlyersFan -
Candidate TRUMP Now Has Crazy Judge JUAN MERCHAN After Him - The Stormy Daniels Hush-Money Case.
Political Conspiracies • 803 • : xuenchen -
Re-election Tactic - JOE BIDEN Hints He May Put Books in the Homes of Black People.
2024 Elections • 30 • : WeMustCare -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order • 3 • : Athetos -
Biden "Happy To Debate Trump"
Mainstream News • 35 • : WeMustCare -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 297 • : cherokeetroy -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 699 • : Thoughtful3 -
Ditching physical money
History • 22 • : StudioNada