It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
a reply to: neutronflux
Yes, statement is false.
That is fine, there are lots of things we do not know. The evidence shows some thing hotter than a usual office fire or plane crash took place. To crack the case we need to follow the evidence to where it leads.
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron. Is that false.
Yes, statement is false.
The statement was there was no source of heat to evaporate steel. It’s like? I don’t know.
That is fine, there are lots of things we do not know. The evidence shows some thing hotter than a usual office fire or plane crash took place. To crack the case we need to follow the evidence to where it leads.
edit on 12-8-2020 by kwakakev because: grammer
a reply to: neutronflux
Yes. This is a source of the iron micro spheres. The next question is where did the heat come from?
Steel burning in atmosphere
Yes. This is a source of the iron micro spheres. The next question is where did the heat come from?
a reply to: kwakakev
Like to change your answer....
originally posted by: kwakakev
a reply to: neutronflux
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron. Is that false.
Yes, statement is false.
Like to change your answer....
Here's How Steel Wool Burns (and Why It Looks Like the Death of Krypton)
www.livescience.com...
The reason a block of iron like a utensil doesn't catch fire is that the surface area is small, relative to the volume, Jason Benedict, an associate professor of chemistry at the University at Buffalo, told Live Science. Rusting iron actually generates some heat in the reaction, but it's a very small amount. In addition, a big block of iron can absorb and dissipate a lot of that heat energy before the block's temperature goes up. (You can see this effect in heating a metal spoon when stirring boiling pasta — a small one very quickly gets too hot to hold, while a bigger spoon takes longer.)
Steel wool, on the other hand, is made of lots of thin strands, and so a lot more iron atoms are in contact with the oxygen in the air. When you add heat (as from a flame), you add energy to the iron, and that makes the iron more likely to react with other elements.
a reply to: kwakakev
AE911's Response to Mick West's Iron Microspheres Talk.
www.metabunk.org...
By Mick West
Post 7
www.metabunk.org...-239750
I was chatting about it with Sarns on Facebook a while back. Basically he can't get past the idea that iron burns, with a low ignition temperature, and makes spheres of various compositions of iron and iron oxide. He thought that the candle flame was super hot, and so was melting the iron filings. I demonstrated (video, below) that it makes no difference what the flame is (butane, candle, paper, wood), the iron filing combust just the same.
He seemed to stop responding after that. But I could have missed it, as I've been busy
Can you burn Iron on a Wood Fire? (9/11 related)
m.youtube.com...
a reply to: neutronflux
No. If the towers where made out of steel wool you would have a good point. A combustion engine made out steel does not burn in normal operating conditions.
Like to change your answer....
No. If the towers where made out of steel wool you would have a good point. A combustion engine made out steel does not burn in normal operating conditions.
originally posted by: kwakakev
a reply to: neutronflux
Like to change your answer....
No. If the towers where made out of steel wool you would have a good point. A combustion engine made out steel does not burn in normal operating conditions.
Now intellectually dishonesty. Good gosh.
The whole argument...
originally posted by: neutronflux
originally posted by: kwakakev
a reply to: neutronflux
How would thermite “vaporize” Iron or steel?
Boiling point of Iron. 5198 Fahrenheit. Steel is a little less.
Thermite burns at at4000 Fahrenheit.
So how would thermite vaporize whole columns?
Good question. What was the source of this heat?
The statement was there was no source of heat to evaporate steel. It’s like? I don’t know. If a particle of steel, iron, rust, or iron oxide found in paints have sufficient surface area to the actual amount of material will burn when exposed to an office fire. You know where a jet [hit a building] or building collapse would knock of rust, small particles of steel, and paint while it was on fire.
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron. Is that false.
Which you stated...
Yes, statement is false.
Then you were soundly proven wrong.
What do you not understand about..
Normally we do not think of iron as being flammable, this is because bulk iron doesn't self-sustain its burning like most flammable materials. But the strands of steel wool are thin enough with enough surface area that heat produced is self-sustaining and will continue to burn through if there is enough air present.
www.instructables.com...
—————-
F’n conspiracists. Shameless in their F’n ignorance.
edit on 12-8-2020 by neutronflux because: Made more clear
a reply to: neutronflux
It is ok that you don't how structural steel turns into iron micro spheres and what caused this heat.
When you call Richard Gage a charlatan for what he understands, you are indirectly calling me the same. Is it fair to make these allegations on something you do not understand?
The statement was there was no source of heat to evaporate steel. It’s like? I don’t know.
It is ok that you don't how structural steel turns into iron micro spheres and what caused this heat.
When you call Richard Gage a charlatan for what he understands, you are indirectly calling me the same. Is it fair to make these allegations on something you do not understand?
a reply to: kwakakev
You
And that process can been conducted at the temperatures at which normal office fires burn. Is that false.
What you get for backing a person that has been repeatedly proven to use falsehoods. Like using photos of columns cut during cleanup as proof of columns cut by thermite. So, if the shoe fits...
Allegations? The falsehoods are well documented, and soundly proven. They are are not allegations. They are documented falsehoods of Richard Gage
You
It is ok that you don't how structural steel turns into iron micro spheres and what caused this heat.
And that process can been conducted at the temperatures at which normal office fires burn. Is that false.
When you call Richard Gage a charlatan for what he understands, you are indirectly calling me the same.
What you get for backing a person that has been repeatedly proven to use falsehoods. Like using photos of columns cut during cleanup as proof of columns cut by thermite. So, if the shoe fits...
Is it fair to make these allegations on something you do not understand
Allegations? The falsehoods are well documented, and soundly proven. They are are not allegations. They are documented falsehoods of Richard Gage
edit on 12-8-2020 by neutronflux because: Added and fixed
a reply to: kwakakev
If the steel in the picture below was raised to a temperature above a normal office fire to the point steel “evaporates” at 5000 Fahrenheit, then way isn’t the steel glowing.
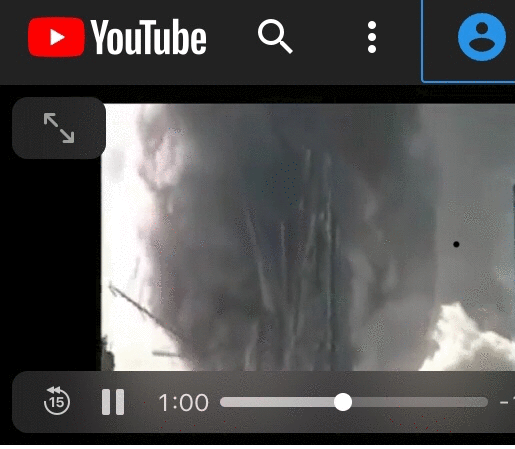
There is no proof and no evidence of steel heated by something other than jet fuel and office fires.
”
If the steel in the picture below was raised to a temperature above a normal office fire to the point steel “evaporates” at 5000 Fahrenheit, then way isn’t the steel glowing.

There is no proof and no evidence of steel heated by something other than jet fuel and office fires.
”
edit on 12-8-2020 by neutronflux because: Added and
edit on 12-8-2020 by neutronflux because: Removed info
a reply to: neutronflux
Your arguments so far have failed to convince me. You saying you do not know does not inspire confidence in your position. If you have the evidence and data, great lets hear it.
So what caused structural steel to turn into iron micro sphere and where did this heat come from?
Allegations? The falsehoods are well documented, and soundly proven.
Your arguments so far have failed to convince me. You saying you do not know does not inspire confidence in your position. If you have the evidence and data, great lets hear it.
So what caused structural steel to turn into iron micro sphere and where did this heat come from?
originally posted by: kwakakev
a reply to: neutronflux
Allegations? The falsehoods are well documented, and soundly proven.
Your arguments so far have failed to convince me. You saying you do not know does not inspire confidence in your position. If you have the evidence and data, great lets hear it.
So what caused structural steel to turn into iron micro sphere and where did this heat come from?
Not suppressing. You don’t seem to understand basic science. And understand Richard Gage is a known and proven Charlatan.
a reply to: kwakakev
The whole argument
—————
originally posted by: neutronflux
Now intellectually dishonesty. Good gosh.
originally posted by: neutronflux
a reply to: kwakakev
I didn’t ask about the debunked work of the Architects and Engineers for Truth. What an oxymoron. Especially the truth part.
Instead of seeing the WTC through some ones created fantasy, I asked:
Do you have proof of planted pyrotechnics cutting columns?
The collapse of the twin towers is right there in the video, audio, seismic record.
Oh may. What do I see for the twin towers...


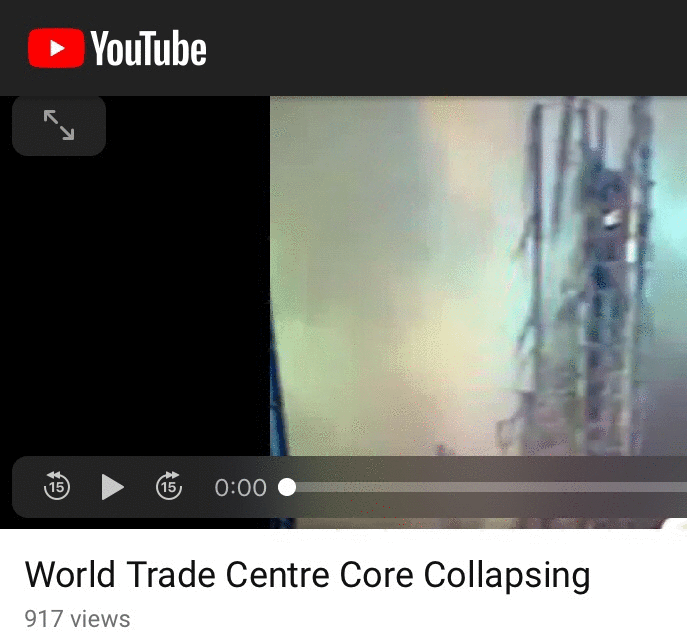

The core columns fell last in long segments. Proving Richard Gage wrong and fallible. And pushing a false narrative.
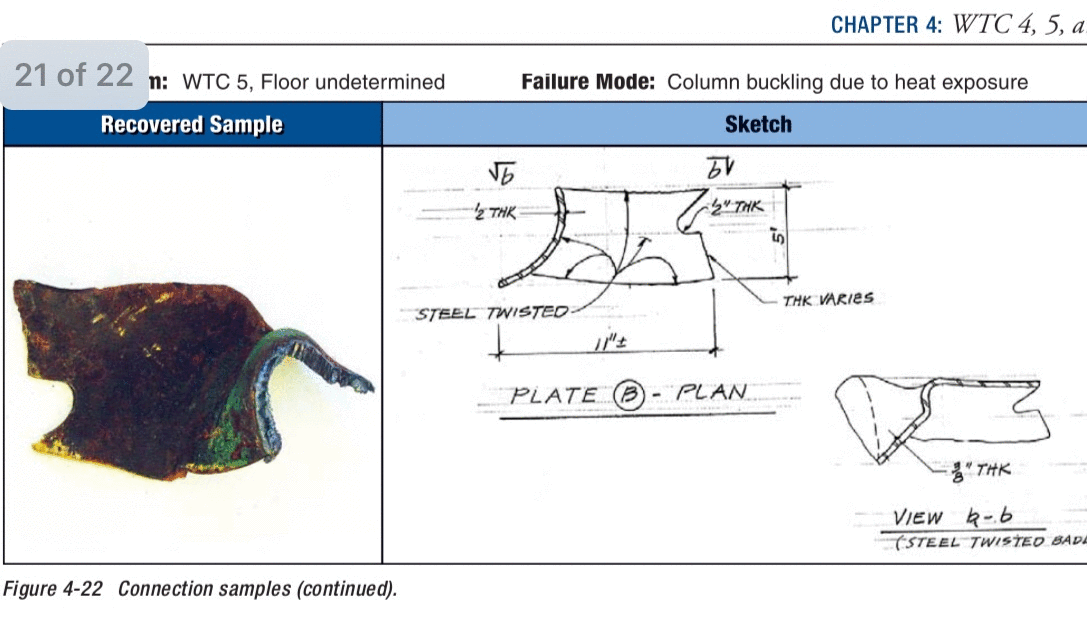
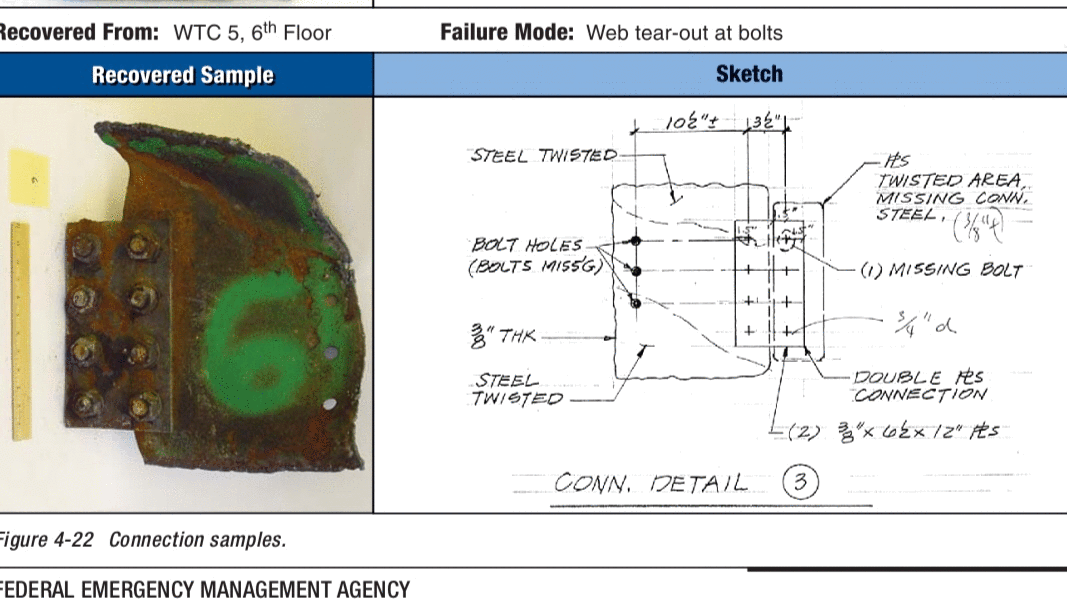
What was that about fire related failures being impossible.
Well, WTC 5 documented failures proves that wrong.
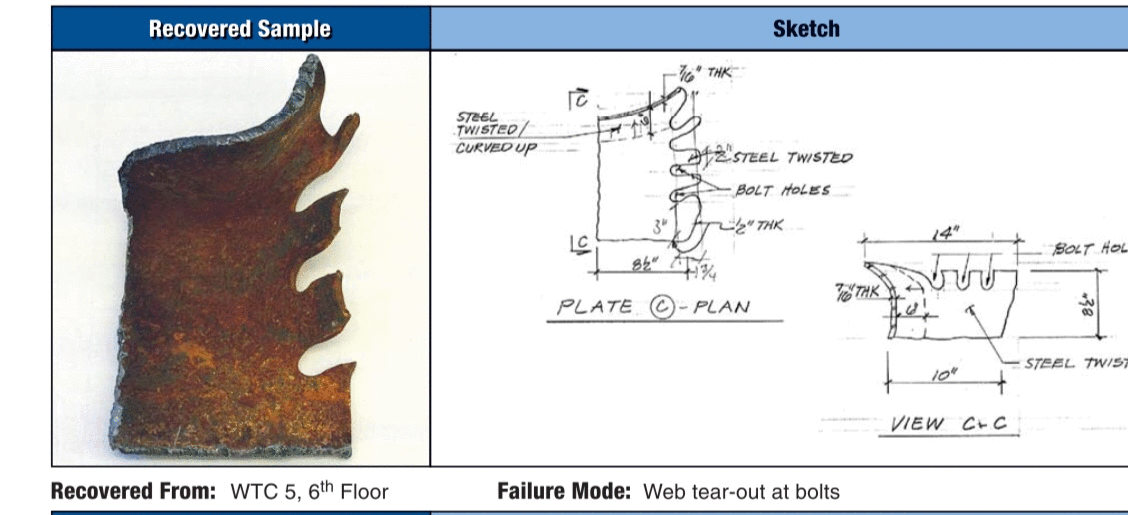


Funny I can go straight to the “evidence”. While yours is presented through a “filter”
The whole argument
—————
originally posted by: neutronflux
originally posted by: kwakakev
a reply to: neutronflux
Like to change your answer....
No. If the towers where made out of steel wool you would have a good point. A combustion engine made out steel does not burn in normal operating conditions.
Now intellectually dishonesty. Good gosh.
originally posted by: neutronflux
a reply to: kwakakev
I didn’t ask about the debunked work of the Architects and Engineers for Truth. What an oxymoron. Especially the truth part.
Instead of seeing the WTC through some ones created fantasy, I asked:
Do you have proof of planted pyrotechnics cutting columns?
The collapse of the twin towers is right there in the video, audio, seismic record.
Oh may. What do I see for the twin towers...




The core columns fell last in long segments. Proving Richard Gage wrong and fallible. And pushing a false narrative.
What was that about fire related failures being impossible.
Well, WTC 5 documented failures proves that wrong.





Funny I can go straight to the “evidence”. While yours is presented through a “filter”
Do you have proof of planted pyrotechnics cutting columns?
I do see the iron micro spheres as proof of something cutting columns. Thermite is one possible explanation. I am open to another explanation but yet to find anything that measures up.
a reply to: kwakakev
Maybe in a normal situation, but WTC was not normal
First start with the aircraft impacts which dumped thousands of gallons of jet fuel in building and started massive fires on multiple floors
The aircraft impacts cut the plumping for sprinklers and standpipes cutting off any chance of controlling fires
Damage to elevators meant no firemen were able to reach the fires before collapse
Add in large amount of synthetic materials AKA plastics in such things like floor/wall coverings. furniture, office equipment . Plastics are made from petroleum and burn with 50-100% more heat energy than organic (wood paper cloth)
Aircraft impact had smashed large holes in building to admit plenty of air Combine that with that was 1000 feet above
ground level where winds speeds are much higher than at ground
What have Is uncontrolled fires attacking steel where the fire proofing had been dislodged
That is fine, there are lots of things we do not know. The evidence shows some thing hotter than a usual office fire or plane crash took place. To crack the case we need to follow the evidence to where it leads.
Maybe in a normal situation, but WTC was not normal
First start with the aircraft impacts which dumped thousands of gallons of jet fuel in building and started massive fires on multiple floors
The aircraft impacts cut the plumping for sprinklers and standpipes cutting off any chance of controlling fires
Damage to elevators meant no firemen were able to reach the fires before collapse
Add in large amount of synthetic materials AKA plastics in such things like floor/wall coverings. furniture, office equipment . Plastics are made from petroleum and burn with 50-100% more heat energy than organic (wood paper cloth)
Aircraft impact had smashed large holes in building to admit plenty of air Combine that with that was 1000 feet above
ground level where winds speeds are much higher than at ground
What have Is uncontrolled fires attacking steel where the fire proofing had been dislodged
originally posted by: kwakakev
Do you have proof of planted pyrotechnics cutting columns?
I do see the iron micro spheres as proof of something cutting columns. Thermite is one possible explanation. I am open to another explanation but yet to find anything that measures up.
But there was no cut columns. The cores fell last.

So your basing your argument off a false claim by Richard Gage. A known charlatan that pushes fake evidence.
With no evidence of heat from 4000 degree Fahrenheit fires, with no evidence of thermite fueled fires, with no evidence of what would be close to something supposedly around 1000 pyrotechnic charges per building.
With no evidence such charges were ever plated.
With no way a system to ignite 1000 pyrotechnic per building would survive air plane impacts, being hit by debris, survive wide spread fires. Fires in WTC 7’s case that burned for hours.
With micro iron sphere formation not being exclusive to Thermite.
You have misconceptions, not evidence.
edit on 13-8-2020 by neutronflux because: Added and fixed
edit on 13-8-2020 by neutronflux because: Added and fixed
a reply to: neutronflux
You have posted evidence of the iron micro spheres. Are you calling yourself a charlatan that pushes fake evidence?
Something caused enough heat to melt the structural steel for these micro spheres to form. I want to know what it was?
So your basing your argument off a false claim by Richard Gage. A known charlatan that pushes fake evidence.
You have posted evidence of the iron micro spheres. Are you calling yourself a charlatan that pushes fake evidence?
Something caused enough heat to melt the structural steel for these micro spheres to form. I want to know what it was?
a reply to: kwakakev
You
In what context? Please quote the actual post of mine your referring to. Or just more intellectually dishonesty by you.
Again...
originally posted by: neutronflux
a reply to: kwakakev
If the steel in the picture below was raised to a temperature above a normal office fire to the point steel “evaporates” at 5000 Fahrenheit, then way isn’t the steel glowing.

There is no proof and no evidence of steel heated by something other than jet fuel and office fires.
originally posted by: neutronflux
Now intellectually dishonesty. Good gosh.
The whole argument...
originally posted by: neutronflux
The statement was there was no source of heat to evaporate steel. It’s like? I don’t know. If a particle of steel, iron, rust, or iron oxide found in paints have sufficient surface area to the actual amount of material will burn when exposed to an office fire. You know where a jet [hit a building] or building collapse would knock of rust, small particles of steel, and paint while it was on fire.
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron. Is that false.
Which you stated...
Then you were soundly proven wrong.
What do you not understand about..
—————-
F’n conspiracists. Shameless in their F’n ignorance.
You
You have posted evidence of the iron micro spheres.
In what context? Please quote the actual post of mine your referring to. Or just more intellectually dishonesty by you.
Something caused enough heat to melt the structural steel for these micro spheres to form. I want to know what it was?
Again...
originally posted by: neutronflux
a reply to: kwakakev
If the steel in the picture below was raised to a temperature above a normal office fire to the point steel “evaporates” at 5000 Fahrenheit, then way isn’t the steel glowing.

There is no proof and no evidence of steel heated by something other than jet fuel and office fires.
originally posted by: neutronflux
originally posted by: kwakakev
a reply to: neutronflux
Like to change your answer....
No. If the towers where made out of steel wool you would have a good point. A combustion engine made out steel does not burn in normal operating conditions.
Now intellectually dishonesty. Good gosh.
The whole argument...
originally posted by: neutronflux
originally posted by: kwakakev
a reply to: neutronflux
How would thermite “vaporize” Iron or steel?
Boiling point of Iron. 5198 Fahrenheit. Steel is a little less.
Thermite burns at at4000 Fahrenheit.
So how would thermite vaporize whole columns?
Good question. What was the source of this heat?
The statement was there was no source of heat to evaporate steel. It’s like? I don’t know. If a particle of steel, iron, rust, or iron oxide found in paints have sufficient surface area to the actual amount of material will burn when exposed to an office fire. You know where a jet [hit a building] or building collapse would knock of rust, small particles of steel, and paint while it was on fire.
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron. Is that false.
Which you stated...
Yes, statement is false.
Then you were soundly proven wrong.
What do you not understand about..
Normally we do not think of iron as being flammable, this is because bulk iron doesn't self-sustain its burning like most flammable materials. But the strands of steel wool are thin enough with enough surface area that heat produced is self-sustaining and will continue to burn through if there is enough air present.
www.instructables.com...
—————-
F’n conspiracists. Shameless in their F’n ignorance.
edit on 13-8-2020 by neutronflux because: Added and fixed
a reply to: neutronflux
link . metabunk.org
Good question. I don't know. I have never seen structural steel vaporize before.
Incorrect. The iron micro spheres are evidence of this. What happens to the structural steel you post is evidence of this.
This does not explain how structural steel turns into iron micro spheres.
In what context? Please quote the actual post of mine your referring to. Or just more intellectually dishonesty by you.
link . metabunk.org
If the steel in the picture below was raised to a temperature above a normal office fire to the point steel “evaporates” at 5000 Fahrenheit, then way isn’t the steel glowing.
Good question. I don't know. I have never seen structural steel vaporize before.
There is no proof and no evidence of steel heated by something other than jet fuel and office fires.
Incorrect. The iron micro spheres are evidence of this. What happens to the structural steel you post is evidence of this.
Steel and iron will burn in a normal atmosphere with sufficient surface area to mass when exposed to a flame well below the boiling/evaporation point of steel/iron.
This does not explain how structural steel turns into iron micro spheres.
edit on 13-8-2020 by kwakakev because: added metabunk link
new topics
-
What is the white pill?
Philosophy and Metaphysics: 1 hours ago -
Mike Pinder The Moody Blues R.I.P.
Music: 2 hours ago -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 5 hours ago -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 9 hours ago
top topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies: 15 hours ago, 34 flags -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 9 hours ago, 18 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 2 hours ago, 5 flags -
Is AI Better Than the Hollywood Elite?
Movies: 17 hours ago, 4 flags -
What is the white pill?
Philosophy and Metaphysics: 1 hours ago, 4 flags -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 5 hours ago, 2 flags -
Maestro Benedetto
Literature: 16 hours ago, 1 flags
active topics
-
God's Righteousness is Greater than Our Wrath
Religion, Faith, And Theology • 32 • : andy06shake -
Las Vegas UFO Spotting Teen Traumatized by Demon Creature in Backyard
Aliens and UFOs • 14 • : pianopraze -
Gaza Terrorists Attack US Humanitarian Pier During Construction
Middle East Issues • 73 • : FlyersFan -
SETI chief says US has no evidence for alien technology. 'And we never have'
Aliens and UFOs • 76 • : andy06shake -
Russia Ukraine Update Thread - part 3
World War Three • 5734 • : Arbitrageur -
So this is what Hamas considers 'freedom fighting' ...
War On Terrorism • 264 • : FlyersFan -
Candidate TRUMP Now Has Crazy Judge JUAN MERCHAN After Him - The Stormy Daniels Hush-Money Case.
Political Conspiracies • 792 • : Threadbarer -
What is the white pill?
Philosophy and Metaphysics • 6 • : kwaka -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works • 19 • : FlyersFan -
Starburst galaxy M82 - Webb Vs Hubble
Space Exploration • 7 • : Scratchpost
