It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
a reply to: MagicWand67
so you are just providing an information service to people ? Fair enough - and entirely commendable. Thank you.
I have seen some of those papers, and others look much like others I recall - and they are precisely why I think the whole "we're being sprayed like bugs", "chemtrails" and "geoengineering is destroying the planet" stories are complete nonsense - the atmosphere is being measured by so many different organisations, from all around the world, in so many more and more advanced ways..........and nothing to support these myths or anything like them is being found.
so you are just providing an information service to people ? Fair enough - and entirely commendable. Thank you.
I have seen some of those papers, and others look much like others I recall - and they are precisely why I think the whole "we're being sprayed like bugs", "chemtrails" and "geoengineering is destroying the planet" stories are complete nonsense - the atmosphere is being measured by so many different organisations, from all around the world, in so many more and more advanced ways..........and nothing to support these myths or anything like them is being found.
a reply to: Aloysius the Gaul
While I agree many of the claims made are exaggerated and unproven they do have an ounce of truth to them.
If or when SRM tests take place there would be adverse side effects for humanity.
Listen to what David Keith says about it on the Colbert Report.
Chaff (unrelated to chemtrails) is also a health concern IMO. The USAF uses about 500 tons of it every year. That is a very significant amount considering how many years they have been using it. And while this report says no adverse environmental or health effects have been directly attributed to chaff. I find that very hard to believe.
Human and Environmental Health Issues Related to Use of Radio Frequency Chaff
While I agree many of the claims made are exaggerated and unproven they do have an ounce of truth to them.
If or when SRM tests take place there would be adverse side effects for humanity.
Listen to what David Keith says about it on the Colbert Report.
Chaff (unrelated to chemtrails) is also a health concern IMO. The USAF uses about 500 tons of it every year. That is a very significant amount considering how many years they have been using it. And while this report says no adverse environmental or health effects have been directly attributed to chaff. I find that very hard to believe.
Human and Environmental Health Issues Related to Use of Radio Frequency Chaff
Chaff is a very light material that can remain suspended in air anywhere from 10 minutes to 10 hours and can travel considerable distances from its release point, depending on prevailing atmospheric conditions (USAF 2001).
Training for military personnel, particularly aircraft pilots, in the use of chaff is necessary to deploy this electronic countermeasure effectively. As with most acquired skills, the deployment of chaff must be maintained by practicing in-flight release during training.
It is estimated that the U.S. Armed Forces dispense about 500 tons of chaff per year (USAF 2001), with most chaff being released during training exercises within the continental United States.
Concerns have been raised since the early 1950s by both the public and government officials on the potential impacts of chaff on the environment. In response to these concerns, the Department of Defense (DOD) has sponsored or conducted research to address issues related to the use of chaff by the military including: (a) questions on its persistence and fate in the environment, (b) the effects of chaff on human, livestock, and wildlife health, and (c) the impact of chaff release on natural and cultural resources
a reply to: MagicWand67
Oh the many smaller pieces and how it all may impact the whole world we see in the end.
I had no idea they were using 500 (ahem) TONS of it, per year. I read the whole report and it sets me back a bit to read further, this is a period (with the above number in weight) of serious restrictions and limitations on the use of Chaff over the continental United States. The amount now, as compared to times past. Oh and they make sure it's used high altitude for maximum dispersion. It actually does say that, with the logic being to avoid concentration over areas. ....but it's harmless. Of course.
It's very notable to point out they say in more than one place, the stuff is robust and doesn't break down easily. In some ways presenting as a benefit, since the whole fibers aren't known in listed examples of testing to be health hazards but they seemed uncertain about partially degraded ones. (they were contact irritants to eyes and sinuses tho ..Hmm..)
So it was I saw this doozy... (remember of course, 500 tons over the US a year, in what they call restricted conditions. at up to 100 million fibers a charge, by 8lb charges at a time...and the date makes all this during peace time)
Well, golly, lemmie put down ma banjo a minoot, and I can thank Unkle Samy for nots usin da lead in those pesky fibres anymur. Uf Curse, I sur wish they wouldn't stay 4ever in the envirenmont. Us in dem dar 26 states still been used and aboosed will manage sumhow tho!
Where's that report on ADHD. Dementia and rising levels of developmental issues over a few decades now?
Oh the many smaller pieces and how it all may impact the whole world we see in the end.
I had no idea they were using 500 (ahem) TONS of it, per year. I read the whole report and it sets me back a bit to read further, this is a period (with the above number in weight) of serious restrictions and limitations on the use of Chaff over the continental United States. The amount now, as compared to times past. Oh and they make sure it's used high altitude for maximum dispersion. It actually does say that, with the logic being to avoid concentration over areas. ....but it's harmless. Of course.
It's very notable to point out they say in more than one place, the stuff is robust and doesn't break down easily. In some ways presenting as a benefit, since the whole fibers aren't known in listed examples of testing to be health hazards but they seemed uncertain about partially degraded ones. (they were contact irritants to eyes and sinuses tho ..Hmm..)
So it was I saw this doozy... (remember of course, 500 tons over the US a year, in what they call restricted conditions. at up to 100 million fibers a charge, by 8lb charges at a time...and the date makes all this during peace time)
Source...Pg 3
Chaff fibers are approximately 60% glass and 40% aluminum by weight. Lead was used as a weighting material in early versions, but this metal is no longer incorporated into chaff (GAO 1998, USAF 1997). Chaff fibers are also coated with a lipid to prevent clumping.
Well, golly, lemmie put down ma banjo a minoot, and I can thank Unkle Samy for nots usin da lead in those pesky fibres anymur. Uf Curse, I sur wish they wouldn't stay 4ever in the envirenmont. Us in dem dar 26 states still been used and aboosed will manage sumhow tho!
Where's that report on ADHD. Dementia and rising levels of developmental issues over a few decades now?
a reply to: Wrabbit2000
I wonder which genius had the bright idea to use lead in chaff.
There's certainly no doubt about it's harmful effects.
I wonder which genius had the bright idea to use lead in chaff.
There's certainly no doubt about it's harmful effects.
a reply to: MagicWand67
The amount of lead in 500 tons of chaff would have paled to insignificance next to the amount of lead in gas over a year - it was an inconsequential pollutant IMO, and hitting on it is just more scaremongering.
ETA: And also nothing to do with geoengineering, SRM, or even chemtrails!!
The amount of lead in 500 tons of chaff would have paled to insignificance next to the amount of lead in gas over a year - it was an inconsequential pollutant IMO, and hitting on it is just more scaremongering.
ETA: And also nothing to do with geoengineering, SRM, or even chemtrails!!
edit on 2-7-2014 by Aloysius the Gaul because: (no reason given)
originally posted by: MagicWand67
a reply to: Aloysius the Gaul
While I agree many of the claims made are exaggerated and unproven they do have an ounce of truth to them.
And what is that ounce of truth?
If or when SRM tests take place there would be adverse side effects for humanity.
As is well known and identified in most research papers into the mattr - it is also one of the reasons the scientific consensus is not to try it with atmospheric sulphur, for example.
originally posted by: MagicWand67
Chaff (unrelated to chemtrails) is also a health concern IMO.
There is some question about whether the aluminum deposited through chaffing exercises is toxic. A research survey by the U.S. Fish and Wildlife Service in 2005, prompted by concerns about the potential for impact of chaff upon endangered pronghorn antelopes, noted the existence of studies that concluded that chaff containing aluminum would not be harmful to either humans or animals: “Although there is potential for inhalation hazard to wildlife, two reports found that the risk is negligible. A study in the United Kingdom found that chaff particle size was too large for inhalation in humans and livestock (USAF 1997). Therefore, chaff is considered too large to be respired. When broken down, chaff would not cause adverse effects to terrestrial wildlife because aluminum and silicon are not very toxic when inhaled (NRL 1999).” The survey also noted a 1999 study of a worst case scenario at a military base in Nevada where chaffing exercises had been frequently run. That study, by the Naval Research Laboratory, “concluded that the deposition of chaff did not result in the accumulation of toxic substances in soils and that inhalation and ingestion exposure to domestic livestock and non-domestic grazers was not a concern due to the large size of chaff fibers, and because ingestion of unreasonably large amounts of chaff would be required to produce toxic effects.” Conducting its own worst-case scenario research, the Fish and Wildlife Service determined that “Sonoran pronghorn are not likely to be at risk from aluminum toxicity at BMGR due to chaff releases.”
link to study
Can you find any peer reviewed study that finds Chaff dangerous?

a reply to: MagicWand67
I'm enjoying reading this thread ... I just had to pause and say ... HOLY SMOKES!
Okay ... carry on with the conversation.
I'm enjoying reading this thread ... I just had to pause and say ... HOLY SMOKES!
Okay ... carry on with the conversation.
I would like to revisit this paper one more time. Which describes the timeline in which certain details of research of SRM are broken down into 5
phases. I want to focus on the first 2 phases because I do not think we have reached phase 3 yet.
A Framework to Prevent the Catastrophic Effects of Global Warming using Solar Radiation Management (Geo-Engineering)
Now if we look at the details of phase 1 and based on all the available info and studies. I think that Phase 1's goals have been accomplished. A few times over even.
Lab research - done
Computer models - done
Set rules to regulate - done (United Nations)
detailed cost analysis - done (Aurora report)
Acquire funding - done (Bill Gates and friends)
Appoint governing body - ? (SRMGI) (IPCC) (USGCRP) (RoyalSociety) (CFR) (UN)
So we've met all the goals of phase 1. Except maybe the last one of appointing a governing body. Then comes phase 2. Which begins with "Careful real World testing of sub scale versions of SRM".
But wait, remember this quote from this paper?
Cooling the Earth Through Solar Radiation Management:
Basically says we don't need or want a governing body while we do our tests. It mentions that other people fly planes and shoot rockets all the time. Why shouldn't we be allowed to spray our sulfuric acid up there too.
A Framework to Prevent the Catastrophic Effects of Global Warming using Solar Radiation Management (Geo-Engineering)
5. Proposed Timeline
This Framework contemplates a five phase approach that would likely achieve its objective of guaranteeing prevention of catastrophic sea level rise within five years.
Phase I – Laboratory Research and Institutional Development: A consortium to include the national leaders in SRM, would conduct preliminary research and technical development work and draft a detailed plan to accomplish the necessary pilot scale testing of SRM, to include funding requirements.
The ideal leader of this consortium would be Professor Wood (with significant assistance by Professor Caldeira and his colleagues), and would include institutional experts such as Professor Barrett at Johns Hopkins. Most physical research would involve laboratory scale physics and chemistry, as well as computer simulations, modeling, and analyses of the kind routinely conducted by climate scientists today.
Simultaneously, the institutional research branch would identify alternative means to regulate and manage SRM use, to include formation of a specific objective such as presented in the first Element above. The plan would include a detailed proposal for formation of a control institution to test and regulate the use of SRM. The plan would ideally be reviewed and accepted by experts from a very wide spectrum of relevant disciplines (18 months, $3.5 million estimated).
Phase II: Careful real world testing of subscale versions of SRM at gradually increasing scales to verify any remaining questions and development of revised implementation plan; appointment and organization of the SRM control organization (18 months). Phase III: Review research results and propose and take comment on an SRM schedule of events. This would be the first major action of the international SRM control body. It would include a reexamination of the objective to ensure adequate global support (18 months).
Now if we look at the details of phase 1 and based on all the available info and studies. I think that Phase 1's goals have been accomplished. A few times over even.
Lab research - done
Computer models - done
Set rules to regulate - done (United Nations)
detailed cost analysis - done (Aurora report)
Acquire funding - done (Bill Gates and friends)
Appoint governing body - ? (SRMGI) (IPCC) (USGCRP) (RoyalSociety) (CFR) (UN)
So we've met all the goals of phase 1. Except maybe the last one of appointing a governing body. Then comes phase 2. Which begins with "Careful real World testing of sub scale versions of SRM".
But wait, remember this quote from this paper?
Cooling the Earth Through Solar Radiation Management:
So long as modest low-level field studies designed to answer these questions are done in an open and transparent manner, we believe they should not be subject to any formal international process of vetting and approval. Countries and firms routinely fly various aircraft in the stratosphere, or send rockets through the stratosphere into space. These activities release significant quantities of particles and gases.
A requirement for formal prior approval of small field studies, just because they are directed at learning about
SRM and its limitations, is probably unenforceable because judging intent is often impossible. Such a regulation would, at best, make conducting modest low-level SRM research extremely difficult and, at worst, impossible
Basically says we don't need or want a governing body while we do our tests. It mentions that other people fly planes and shoot rockets all the time. Why shouldn't we be allowed to spray our sulfuric acid up there too.
a reply to: network dude
Okay, let's examine chaff a little closer then.
Remember this article from earlier in the thread?
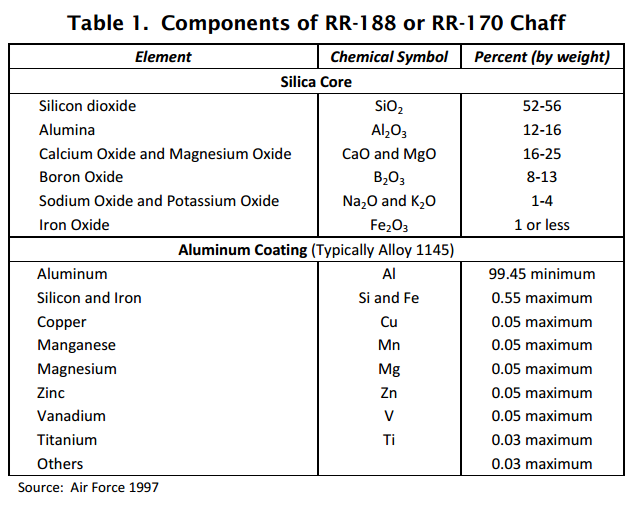
On average somewhere around 75% of RR-188 chaff is composed of Aluminum and Silica and 25% is composed of other oxides.
Some chaff can be up to 13% Boron Trioxide
Occupational saftey sheet for boron oxide
500 tons of chaff = 1,000,000 lbs
13% of 1,000,000lbs = 130,000 lbs of Boron trioxide each year
Let's look at Calcium and Magnesium oxide
Are we really supposed to believe this is not bad for us to breath, drink, and land on our skin?
Characteristics of Chaff
Okay, let's examine chaff a little closer then.
Remember this article from earlier in the thread?
Weird Weather Radar 'Blob' Tied To 'Chaff' Used In Military Test
"What we were able to see from the dual-pol radar data looked similar to military chaff cases previously, but the primary difference was that the winds weren't blowing the stuff away," Havin said.
"The releases were happening primarily below 3,300 feet [1,000 meters] above the ground and the low-level winds that afternoon were almost nonexistent (less than 3 mph [4.8 km/h]), so the chaff was basically pluming outward over a good portion of the Huntsville metro area."
In fact, the chaff was visible on their radar for more than nine hours, and the news stories lingered even longer.
"Officially, Redstone Arsenal disclosed that it was a military test using RR-188 military chaff," Havin said, referring to aircraft used to spread a cloud of aluminum-coated silica in the case of RR-188.

On average somewhere around 75% of RR-188 chaff is composed of Aluminum and Silica and 25% is composed of other oxides.
Some chaff can be up to 13% Boron Trioxide
Occupational saftey sheet for boron oxide
Boron Trioxide
Boron trioxide (or diboron trioxide) is one of the oxides of boron. It is a white, glassy solid with the formula B2O3. It is almost always found as the vitreous (amorphous) form; however, it can be crystallized after extensive annealing (that is, under prolonged heat). It is known as one of the most difficult compounds to crystallize
.......
Boron trioxide is produced by treating borax withsulfuric acid in a fusion furnace. At temperatures above 750 °C, the molten boron oxide layer separates out from sodium sulfate. It is then decanted, cooled and obtained in 96–97% purity.
....
Boron oxide will also form when Diborane (B2H6) reacts with oxygen in the air or trace amounts of moisture:
.....
See also
boron suboxide
boric acid
sassolite
Boric Acid
Toxicology
Based on mammalian median lethal dose (LD50) rating of 2,660 mg/kg body mass, boric acid is poisonous if taken internally or inhaled in large quantities. The Thirteenth Edition of the Merck Index indicates that the LD50 of boric acid is 5.14 g/kg for oral dosages given to rats, and that 5 to 20 g/kg has produced death in adult humans. For comparison's sake, the LD50 of salt is reported to be 3.75 g/kg in rats according to the Merck Index. But this is highly misleading, as humans have been known to die from amounts only a small fraction of the LD50. "The minimal lethal dose of ingested boron (as boric acid) was reported to be 2–3 g in infants, 5–6 g in children, and 15–20 g in adults", according to the Agency for Toxic Substances and Disease Registry. [9]
Long term exposure to boric acid may be of more concern, causing kidney damage and eventually kidney failure (see links below). Although it does not appear to be carcinogenic, studies in dogs have reported testicular atrophy after exposure to 32 mg/kg bw/day for 90 days. This level is far lower than the LD50.[10]
Diborane
The toxic effects of diborane are primarily due to its irritant properties. Short-term exposure to diborane can cause a sensation of tightness of the chest, shortness of breath, cough, and wheezing. These signs and symptoms can occur immediately or be delayed for up to 24 hours. Skin and eye irritation can also occur. Studies in animals have shown that diborane causes the same type of effects observed in humans.[citation needed]
People exposed for a long time to low amounts of diborane have experienced respiratory irritation, seizures, fatigue, drowsiness, confusion, and occasional transient tremors.
500 tons of chaff = 1,000,000 lbs
13% of 1,000,000lbs = 130,000 lbs of Boron trioxide each year
Let's look at Calcium and Magnesium oxide
Calcium Oxide
Health issues
Because of vigorous reaction of quicklime with water, quicklime causes severe irritation when inhaled or placed in contact with moist skin or eyes. Inhalation may cause coughing, sneezing, labored breathing. It may then evolve into burns with perforation of the nasal septum, abdominal pain, nausea and vomiting. Although quicklime is not considered a fire hazard, its reaction with water can release enough heat to ignite combustible materials.
Magnesium Oxide
Precautions
Magnesium oxide is easily made by burning magnesium ribbon, which produces a very bright white light, and a powdery ash. The bright flame is very hard to extinguish and it emits a harmful intensity of UV light. Inhalation of magnesium oxide fumes can cause metal fume fever.[19] When burned in open air, the magnesium gets hot enough to produce noticeable amounts of yellow magnesium nitride. Burning in a covered crucible, letting in just enough air to support combustion, will reduce the burning temperature, minimizing the production of the nitride.
Are we really supposed to believe this is not bad for us to breath, drink, and land on our skin?
Characteristics of Chaff
edit on 2-7-2014 by MagicWand67 because: (no reason given)
edit on 2-7-2014 by MagicWand67 because: (no reason given)
originally posted by: MagicWand67
a reply to: network dude
Are we really supposed to believe this is not bad for us to breath, drink, and land on our skin?
the evidence you provided seems clear enough - it is toxic in great enough quantities or concentrations - sort of like every otehr substance we know of.
Clearly 130,000lbs/year spread across the USA is no great health hazard - but best to keep away from any concentrations.
And nor is it related to geoengineering, solar radiation management, or even chemtrails.
Why so far off topic??
a reply to: Aloysius the Gaul
I'd beg to differ. 130,000 lbs a year (or 500 tons of overall chaff a year by the Air Force statement recently) is then something to multiply by how many years running back to the 1950's some of the above links say it's been used for?
Particularly when the mention of lead not being used any longer is cited to a report of the late 90's for something happening decades prior to it? Just how much lead by literal weight did get distributed in a relatively fine form, in an even distribution?
This type of pollution would be assumed as accidental or reckless at worst (but then, it is a conspiracy site, so who knows...) but even post-lead years? Respiratory issues are becoming epidemic across this nation along with everything else we're seeing everyone around us starting to have issues with. Just how is this to encounter in partial states of breaking down? That seemed to be the health question.
ANYTHING we learn has been falling across our environment and on our heads for 6 decades is a very BIG deal to my thinking.
The training isn't the issue...the sheer VOLUME of impact over LONG years is what I stop cold with. It's not Chemtrails. It's quite likely worse. Especially as that lead, in tiny amounts x's billions of fibers with it is likely breaking down across the entire nation.
What a thing to be a cumulative environmental toxin.
I'd beg to differ. 130,000 lbs a year (or 500 tons of overall chaff a year by the Air Force statement recently) is then something to multiply by how many years running back to the 1950's some of the above links say it's been used for?
Particularly when the mention of lead not being used any longer is cited to a report of the late 90's for something happening decades prior to it? Just how much lead by literal weight did get distributed in a relatively fine form, in an even distribution?
This type of pollution would be assumed as accidental or reckless at worst (but then, it is a conspiracy site, so who knows...) but even post-lead years? Respiratory issues are becoming epidemic across this nation along with everything else we're seeing everyone around us starting to have issues with. Just how is this to encounter in partial states of breaking down? That seemed to be the health question.
ANYTHING we learn has been falling across our environment and on our heads for 6 decades is a very BIG deal to my thinking.
The training isn't the issue...the sheer VOLUME of impact over LONG years is what I stop cold with. It's not Chemtrails. It's quite likely worse. Especially as that lead, in tiny amounts x's billions of fibers with it is likely breaking down across the entire nation.
What a thing to be a cumulative environmental toxin.
a reply to: Aloysius the Gaul
I wouldn't want to be one of those people in Huntsville when that chaff cloud was tested.
Why can't they test this stuff above unpopulated areas at least?
I wouldn't want to be one of those people in Huntsville when that chaff cloud was tested.
Why can't they test this stuff above unpopulated areas at least?
a reply to: Wrabbit2000
My feelings exactly.
That boron oxide is nasty stuff. It breaks down in water to form boric acid. 5-20 grams of boric acid is deadly.
What's the average amount of chaff released in a single test? How many pounds?
We could calculate how much boron oxide was floating above that city that day.
What a thing to be a cumulative environmental toxin.
My feelings exactly.
That boron oxide is nasty stuff. It breaks down in water to form boric acid. 5-20 grams of boric acid is deadly.
What's the average amount of chaff released in a single test? How many pounds?
We could calculate how much boron oxide was floating above that city that day.
a reply to: MagicWand67
8lbs per cannister fired was an average quoted in the Air Force report above. So, the volume represented to get 500 tons of total material weight from 8lb shots? Well... I guess I just never gave any thought to how MUCH of a thing like that may be used over us. Really? Small amounts wouldn't bother me. 60 years of all different levels in air all over the nation?
Well.. Economy of scale does unpleasant things to outcomes, in my view.
8lbs per cannister fired was an average quoted in the Air Force report above. So, the volume represented to get 500 tons of total material weight from 8lb shots? Well... I guess I just never gave any thought to how MUCH of a thing like that may be used over us. Really? Small amounts wouldn't bother me. 60 years of all different levels in air all over the nation?
Well.. Economy of scale does unpleasant things to outcomes, in my view.
a reply to: Aloysius the Gaul
Did you read the Occupational Safety Sheet on Boron Oxide?
Did you read the symptoms of acute exposure?
Unfortunately the document won't let me cut and paste from it.
But don't those symptoms resemble all the complaints of those crazy chemmies?
And nor is it related to geoengineering, solar radiation management, or even chemtrails.
Did you read the Occupational Safety Sheet on Boron Oxide?
Did you read the symptoms of acute exposure?
Unfortunately the document won't let me cut and paste from it.
But don't those symptoms resemble all the complaints of those crazy chemmies?
a reply to: Wrabbit2000
So about 1 pound of boron oxide is released in a single burst.
That's enough to kill about 50 people if they were spoon fed.
Of course it's all spread out so no worries.
We all get to share in the benefits of boron oxide chaff.
And let's not forget we have that new smart nano chaff to look forward to also.
So about 1 pound of boron oxide is released in a single burst.
That's enough to kill about 50 people if they were spoon fed.
Of course it's all spread out so no worries.
We all get to share in the benefits of boron oxide chaff.
And let's not forget we have that new smart nano chaff to look forward to also.
edit on 3-7-2014 by MagicWand67 because: (no reason given)
a reply to: MagicWand67
Well, it's late and math isn't my thing...but I get 500 short tons converting to roughly 1,000,000lbs. Divide that by 8 (which is a real real rough thing, as canister size does vary) and it comes to 125,000 canisters to account for the weight the AF said it expended in one year, domestically, during restricted operation with it.
Yup.. I'm sure that's harmless over decades. Uncle Sammy says so, and we all know Uncle is always here to help and look out for our very best interests.
(wouldn't it be amazing to find that the symptoms people attribute to chem trails, which may be 99% or more misidentified, came from spraying of a totally different form, never likely thought to be such a causative agent)
Well, it's late and math isn't my thing...but I get 500 short tons converting to roughly 1,000,000lbs. Divide that by 8 (which is a real real rough thing, as canister size does vary) and it comes to 125,000 canisters to account for the weight the AF said it expended in one year, domestically, during restricted operation with it.
Yup.. I'm sure that's harmless over decades. Uncle Sammy says so, and we all know Uncle is always here to help and look out for our very best interests.
(wouldn't it be amazing to find that the symptoms people attribute to chem trails, which may be 99% or more misidentified, came from spraying of a totally different form, never likely thought to be such a causative agent)
edit on 7/3/2014 by Wrabbit2000 because: (no reason given)
a reply to: Wrabbit2000
I must say I'm still a bit shocked myself.
I just found this info today when trying to answer networkdude's question.
----------
Here's another paper on the Air Force's use of SF6 (Sulfur Hexa Flouride)
Now this stuff may be quite harmless to humans but it's the worst thing to put in the atmosphere as far as the green house effect.
SF6 in tracer gas Applications
I must say I'm still a bit shocked myself.
I just found this info today when trying to answer networkdude's question.
----------
Here's another paper on the Air Force's use of SF6 (Sulfur Hexa Flouride)
Now this stuff may be quite harmless to humans but it's the worst thing to put in the atmosphere as far as the green house effect.
SF6 in tracer gas Applications
new topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies: 4 hours ago -
Maestro Benedetto
Literature: 6 hours ago -
Is AI Better Than the Hollywood Elite?
Movies: 6 hours ago -
Las Vegas UFO Spotting Teen Traumatized by Demon Creature in Backyard
Aliens and UFOs: 9 hours ago -
2024 Pigeon Forge Rod Run - On the Strip (Video made for you)
Automotive Discussion: 10 hours ago -
Gaza Terrorists Attack US Humanitarian Pier During Construction
Middle East Issues: 11 hours ago
top topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies: 4 hours ago, 26 flags -
Krystalnacht on today's most elite Universities?
Social Issues and Civil Unrest: 16 hours ago, 9 flags -
Supreme Court Oral Arguments 4.25.2024 - Are PRESIDENTS IMMUNE From Later Being Prosecuted.
Above Politics: 15 hours ago, 8 flags -
Weinstein's conviction overturned
Mainstream News: 14 hours ago, 8 flags -
Gaza Terrorists Attack US Humanitarian Pier During Construction
Middle East Issues: 11 hours ago, 8 flags -
Massachusetts Drag Queen Leads Young Kids in Free Palestine Chant
Social Issues and Civil Unrest: 13 hours ago, 7 flags -
Las Vegas UFO Spotting Teen Traumatized by Demon Creature in Backyard
Aliens and UFOs: 9 hours ago, 6 flags -
Meadows, Giuliani Among 11 Indicted in Arizona in Latest 2020 Election Subversion Case
Mainstream News: 12 hours ago, 5 flags -
2024 Pigeon Forge Rod Run - On the Strip (Video made for you)
Automotive Discussion: 10 hours ago, 4 flags -
Is AI Better Than the Hollywood Elite?
Movies: 6 hours ago, 3 flags
active topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies • 17 • : BingoMcGoof -
Gaza Terrorists Attack US Humanitarian Pier During Construction
Middle East Issues • 29 • : 19Bones79 -
Supreme Court Oral Arguments 4.25.2024 - Are PRESIDENTS IMMUNE From Later Being Prosecuted.
Above Politics • 90 • : Lumenari -
Las Vegas UFO Spotting Teen Traumatized by Demon Creature in Backyard
Aliens and UFOs • 12 • : KrustyKrab -
SHORT STORY WRITERS CONTEST -- April 2024 -- TIME -- TIME2024
Short Stories • 23 • : DontTreadOnMe -
Truth Social goes public, be careful not to lose your money
Mainstream News • 130 • : Astyanax -
Is AI Better Than the Hollywood Elite?
Movies • 13 • : Justoneman -
Hate makes for strange bedfellows
US Political Madness • 47 • : 19Bones79 -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 689 • : daskakik -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 265 • : Astrocometus