It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
This is my personal belief, and probably horribly unscientific. I didn’t worry about C02 warming all that much from the fact the temperature around
here drops like a rock on a clear winter’s night.
But something got my admittedly biased attention, a goofy Scfi show that gave the composition of the Martian atmosphere. Note, the below quote is not from the movie.
Long story short, the Martian atmosphere is really thin, but about 95 percent carbon dioxide.
So, what happens to the temperatures on Mars over a Martian day?
I get that even though the Martian atmosphere is mostly carbon dioxide, it’s too thin to hold heat.
So the question is, “How thick or dense does CO2 need to be to trap heat?”
When you try to google something like “How thick or dense does CO2 need to be to trap heat?” This is the result you get? Below is an example.
In short, the same BS answer the world is heating up because of increasing CO2. Which isn’t the real question. Or even science.
The real question concerning C02 is,”At what concentration of CO2 is great enough in Earth’s atmosphere to notability effect how much heat is retained by an already dense and insulating atmosphere?”
What I also hate in the context of:
What really contributed to the 0.8° Celsius increase? I can see that being caused by deforestation, laying large areas of pavement and concrete, the increase in large buildings, increases in equipment that releases heat (all the cars, factories, power plants, ships spilling heat into the environment), and the increase usage of cooling towers that releases heat into the atmosphere and water vapor that is too a greenhouse gas.
But something got my admittedly biased attention, a goofy Scfi show that gave the composition of the Martian atmosphere. Note, the below quote is not from the movie.
en.m.wikipedia.org...
The atmosphere of the planet Mars is composed mostly of carbon dioxide. The atmospheric pressure on the Martian surface averages 600 pascals (0.087 psi; 6.0 mbar), about 0.6% of Earth's mean sea level pressure of 101.3 kilopascals (14.69 psi; 1.013 bar). It ranges from a low of 30 pascals (0.0044 psi; 0.30 mbar) on Olympus Mons's peak to over 1,155 pascals (0.1675 psi; 11.55 mbar) in the depths of Hellas Planitia. This pressure is well below the Armstrong limit for the unprotected human body. Mars's atmospheric mass of 25 teratonnes compares to Earth's 5148 teratonnes; Mars has a scale height of 11.1 kilometres (6.9 mi)[2] versus Earth's 8.5 kilometres (5.3 mi).[3]
Long story short, the Martian atmosphere is really thin, but about 95 percent carbon dioxide.
So, what happens to the temperatures on Mars over a Martian day?
www.universetoday.com...
Even at the equator, the night time temperatures fall well below zero. On those summer days, it can be around 20 degrees Celsius then plummet to -90 C at night.
I get that even though the Martian atmosphere is mostly carbon dioxide, it’s too thin to hold heat.
So the question is, “How thick or dense does CO2 need to be to trap heat?”
When you try to google something like “How thick or dense does CO2 need to be to trap heat?” This is the result you get? Below is an example.
www.thoughtco.com...
Carbon Dioxide as a Greenhouse Gas
In the atmosphere, carbon dioxide contributes with other molecules to the greenhouse effect. Energy from the sun gets reflected by the surface of the earth, and in the process it is transformed into a wavelength more easily intercepted by greenhouse gases, trapping the heat within the atmosphere instead of letting it reflect out into space. Carbon dioxide’s contribution to the greenhouse effect varies between 10 and 25 % depending on the location, immediately behind water vapor.
An Upward Trend
The concentration of CO2 in the atmosphere has varied over time, with significant ups and downs experienced by the planet over geological times. If we look at the last millennia however we see a steep rise in carbon dioxide clearly starting with the industrial revolution. Since pre-1800 estimates the CO2 concentrations have risen by over 42% to current levels over 400 parts per million (ppm), driven by the burning of fossil fuels and by land clearing.
In short, the same BS answer the world is heating up because of increasing CO2. Which isn’t the real question. Or even science.
The real question concerning C02 is,”At what concentration of CO2 is great enough in Earth’s atmosphere to notability effect how much heat is retained by an already dense and insulating atmosphere?”
What I also hate in the context of:
earthobservatory.nasa.gov...
Earth has increased by about 0.8° Celsius (1.4° Fahrenheit) since 1880.
What really contributed to the 0.8° Celsius increase? I can see that being caused by deforestation, laying large areas of pavement and concrete, the increase in large buildings, increases in equipment that releases heat (all the cars, factories, power plants, ships spilling heat into the environment), and the increase usage of cooling towers that releases heat into the atmosphere and water vapor that is too a greenhouse gas.
To simplify further.....
You would think a scale could be created.
With this snapshot of Earth’s atmosphere at this concentration, this density, you have this factor of insulation, with this factor of greenhouse heating to retain this much heat in BTUs. Therefor, with every 100 ppm increase in CO2 will yield this much retention in BTUs of heat resulting in the average temperature increasing by x amount.
My guess is there is no real noticeable heat retention for changes in CO from 200 ppm to 600 ppm in an already thick and insulating atmosphere?
You would think a scale could be created.
With this snapshot of Earth’s atmosphere at this concentration, this density, you have this factor of insulation, with this factor of greenhouse heating to retain this much heat in BTUs. Therefor, with every 100 ppm increase in CO2 will yield this much retention in BTUs of heat resulting in the average temperature increasing by x amount.
My guess is there is no real noticeable heat retention for changes in CO from 200 ppm to 600 ppm in an already thick and insulating atmosphere?
edit on 30-12-2018 by neutronflux because: Added and fixed
I don't get the co2 thing. How's it even in the atmosphere, it's heavier than air?
I do know that plants grow at up to 50% faster at 1000ppm of co2.
-the same one's that pump out oxygen at night.
Greenhouses, etc distribute the co2 from the ceiling because it drops towards the floor due to it's weight.
How tf is it ending up floating up into the atmosphere like claimed?
..or is it "filling" the space along the ground, losing density with altitude, like oxygen?
I do know that plants grow at up to 50% faster at 1000ppm of co2.
-the same one's that pump out oxygen at night.
Greenhouses, etc distribute the co2 from the ceiling because it drops towards the floor due to it's weight.
How tf is it ending up floating up into the atmosphere like claimed?
..or is it "filling" the space along the ground, losing density with altitude, like oxygen?
edit on 12 by Mandroid7 because: Added2
a reply to: neutronflux
I use DuckDuckGo it will not push an idea or product on you.
This was at the top of my DDG search CO2 Tables Calculator - Carbon Dioxide Properties.
If you figure it out let us know what you find.
I use DuckDuckGo it will not push an idea or product on you.
This was at the top of my DDG search CO2 Tables Calculator - Carbon Dioxide Properties.
If you figure it out let us know what you find.
edit on 30-12-2018 by LookingAtMars because: (no reason given)
a reply to: neutronflux
You're kinda forgetting about an atmosphere. Carbon Dioxide is only one element of the Earth's atmosphere. However what it does is prevent heat from escaping from our nitrogen based atmosphere.
Adding more CO2 to the atmosphere increases the ratio of greenhouse gases which effectively "trap" heat.
Skeptical Science
So if you want to compare apples to apples, find another planet with a mostly nitrogen atmosphere first and then add CO2 to it.
You're kinda forgetting about an atmosphere. Carbon Dioxide is only one element of the Earth's atmosphere. However what it does is prevent heat from escaping from our nitrogen based atmosphere.
Earth's atmosphere is 78% nitrogen, 21% oxygen, 0.9% argon, and 0.03% carbon dioxide with very small percentages of other elements. Our atmosphere also contains water vapor. In addition, Earth's atmosphere contains traces of dust particles, pollen, plant grains and other solid particles
Adding more CO2 to the atmosphere increases the ratio of greenhouse gases which effectively "trap" heat.
The effect of adding man-made CO2 is predicted in the theory of greenhouse gases. This theory was first proposed by Swedish scientist Svante Arrhenius in 1896, based on earlier work by Fourier and Tyndall. Many scientist have refined the theory in the last century. Nearly all have reached the same conclusion: if we increase the amount of greenhouse gases in the atmosphere, the Earth will warm up.
Skeptical Science
So if you want to compare apples to apples, find another planet with a mostly nitrogen atmosphere first and then add CO2 to it.
a reply to: kelbtalfenek
I don’t think soo. My argument comes down to this.
The real question concerning C02 is,”At what concentration of CO2 is great enough in Earth’s atmosphere to notability effect how much heat is retained by an already dense and insulating atmosphere?”
I don’t think soo. My argument comes down to this.
The real question concerning C02 is,”At what concentration of CO2 is great enough in Earth’s atmosphere to notability effect how much heat is retained by an already dense and insulating atmosphere?”
a reply to: LookingAtMars
Than you for the link, but to use the calculator: “registration required”
I really do try to minimize my internet footprint.
Than you for the link, but to use the calculator: “registration required”
I really do try to minimize my internet footprint.
a reply to: neutronflux
That's a bummer. I don't like making accounts on the web either. I did a search with different key words and found this.
Does Carbon Dioxide ‘Trap Heat’?
That's a bummer. I don't like making accounts on the web either. I did a search with different key words and found this.
Does Carbon Dioxide ‘Trap Heat’?
Problem #2—None of these table top “greenhouse effect in a bottle” experiments test CO2 at 560 ppm vs. CO2 at 280ppm The big debate about CO2’s effect on global surface-level air temperatures is what will happen when atmospheric CO2 doubles in concentration from pre-industrial times, i.e., increases from 0.026% (280 ppm) of the atmosphere to 0.056% (560 ppm). Yes, 0.056% is a “doubling” of the amount of CO2 in the air from pre-industrial times but it is still a minute amount. None of these table-top “greenhouse effect in a bottle” experiments test the effect of a CO2 level of 0.056% vs. a CO2 level of 0.026%. They all test CO2 levels of 50-100% compared to regular air and even then on average they still only get a temperature rise of several degrees due to CO2‘s lower specific heat. Based on computer models, the “climate sensitivity of carbon dioxide” hypothesis asserts that a doubling of CO2 levels from pre-industrial times, from ~260 ppm to ~560 ppm, will result in 2-6 °C of global warming. These table top “greenhouse effect in a bottle” experiments demonstrate that the atmosphere would have to be 50-100% CO2 (500,000 – 1,000,000 ppm) to get that much warming but even then the extra warming would not be from a “greenhouse effect”, but rather would be a result of CO2’s lower specific heat value.
edit on 30-12-2018 by LookingAtMars because: (no reason given)
Here is another example..
Who gives a rat’s ass about ice samples being actual proof.
This is how real science is supposed to work.
CO2 is known to have x properties. This results in C02 adding to x amount in greenhouse warming, and it’s insulation value of x both will cause this amount in BTUs for heat retention to cause the earth’s average temperature to increase x amount for every 50 ppm increase in CO2 in the earths atmosphere.
The calculated increase in temperature per 50 ppm increase of CO2 is supported by historical temperatures and historical CO2 concertation as captured in ice core samples.
Bad science is: “the earth’s temperature is rising because we can prove CO2 increased from ice core samples.”
Good Science is: “using the known properties of CO2, we created a model where the earth’s average temperature will increase this much by every 50 ppm increase of CO2 in the atmosphere. We understand the greenhouse effect by comparing Venus, Earth, and Mars. Historical temperatures and historical CO2 levels as captured by Ice Core samples shows the model to be about 70 percent correct for the historical temperature trend.”
Why does CO2 get most of the attention when there are so many other heat-trapping gases?
www.ucsusa.org...
CO2 has caused most of the warming and its influence is expected to continue
CO2 has contributed more than any driver to climate change between 1750 and 2011.
The Intergovernmental Panel on Climate Change (IPCC) issued a global climate assessment in 2013 that compared the influence of three changes to the environment resulting from human activity between 1750 and 2011: the emission of key heat-trapping gases and tiny particles known as aerosols, as well as land use change.
By measuring the abundance of heat-trapping gases in ice cores, the atmosphere, and other climate drivers along with models, the IPCC calculated the “radiative forcing” (RF) of each climate driver—in other words, the net increase (or decrease) in the amount of energy reaching Earth’s surface attributable to that climate driver.
Who gives a rat’s ass about ice samples being actual proof.
This is how real science is supposed to work.
CO2 is known to have x properties. This results in C02 adding to x amount in greenhouse warming, and it’s insulation value of x both will cause this amount in BTUs for heat retention to cause the earth’s average temperature to increase x amount for every 50 ppm increase in CO2 in the earths atmosphere.
The calculated increase in temperature per 50 ppm increase of CO2 is supported by historical temperatures and historical CO2 concertation as captured in ice core samples.
Bad science is: “the earth’s temperature is rising because we can prove CO2 increased from ice core samples.”
Good Science is: “using the known properties of CO2, we created a model where the earth’s average temperature will increase this much by every 50 ppm increase of CO2 in the atmosphere. We understand the greenhouse effect by comparing Venus, Earth, and Mars. Historical temperatures and historical CO2 levels as captured by Ice Core samples shows the model to be about 70 percent correct for the historical temperature trend.”
edit on 30-12-2018 by neutronflux because: Added and fixed
edit on 30-12-2018 by neutronflux because: Added and fixed
a reply to: neutronflux
I am sure when the Sun comes up in Hawaii someone will be around to set us straight
I am sure when the Sun comes up in Hawaii someone will be around to set us straight
It is all BS.
It is about the Global Elites trying to control all us useless eaters and nothing more. The fact that there are still people that think this is about the planet warming just shows how stupid people can be.
It is about the Global Elites trying to control all us useless eaters and nothing more. The fact that there are still people that think this is about the planet warming just shows how stupid people can be.
Quote that touches upon this issue:
According to the Arrhenius equation for calculating how CO2 catches heat the warming on Mars should be larger. “The Mars atmosphere is highly amenable to empirical testing of Arrhenius’ equation because its climate-system contains no real significant feedback-mechanisms to complicate or modify the direct and immediate response of the CO2 in terms of the amount of radiative forcing that it is supposed to produce”. When the Arrhenius equation is applied to the planet of Mars things go horribly wrong. There the observed global warming (according to NASA’s revised Fact Sheet) is about 0.2°K (or about 0.5 W/m2). The revised NASA data shows an effective temperature for Mars of 209.8°K (See NASA Mars Fact Sheet) and an average global surface temperature of ~210°K. This is interesting when one considers that Mars has a CO2 density approximately 27 times higher than on Earth — at 165 kg/m2 and 6 kg/m2 respectively and on Earth CO2 is claimed to increase the global average surface temperature by 6°K (or 32 W/m2). The CO2 on Mars apparently produces 30 times less warming despite being 27 times more abundant.
originally posted by: Metallicus
It is all BS.
It is about the Global Elites trying to control all us useless eaters and nothing more. The fact that there are still people that think this is about the planet warming just shows how stupid people can be.
That really doesn’t add to the argument? As in proof one way or another.
The climate has and will always change. The Earth, solar system, galaxy is always changing by degrees. Change will happen with, or without people. Put ever bit of effort, and make all destitute, to try to prevent the earth’s average temperature from changin three degrees, and one random gamma burst could wipe all life from earth.
I think it’s more important to expand into the solar system than worry about three degrees. The earth just has to many people that could be used to colonize the solar system. people were made to adapt and explore, not overpopulate a planet.
edit on 30-12-2018 by neutronflux because: Added adapt
originally posted by: LookingAtMars
a reply to: neutronflux
I am sure when the Sun comes up in Hawaii someone will be around to set us straight
Now that's funny! ha

Anyone want to talk about the rate at which CO2 effects temperature changes?
Just a little FYI, the more CO2 the less overall effect each additional ppm causes. The reason for this has to do with the wavelengths that CO2 absorbs, and at some point it has absorbed nearly all of the energy in the wavelengths it absorbs.
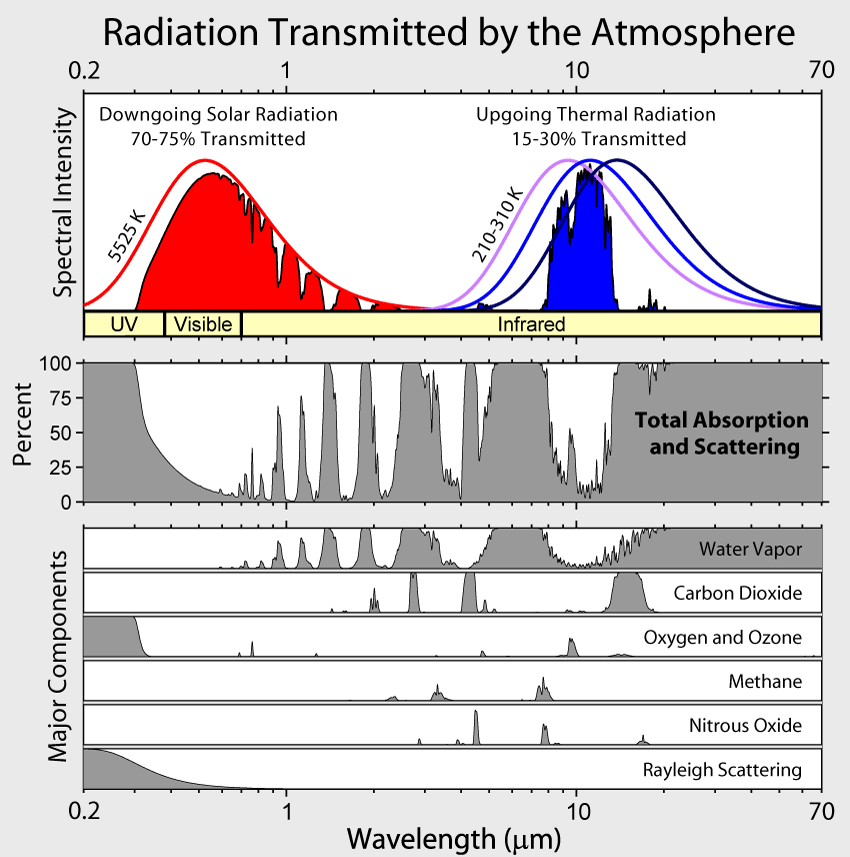
Just a little FYI, the more CO2 the less overall effect each additional ppm causes. The reason for this has to do with the wavelengths that CO2 absorbs, and at some point it has absorbed nearly all of the energy in the wavelengths it absorbs.

originally posted by: dubiousatworst
Anyone want to talk about the rate at which CO2 effects temperature changes?
Just a little FYI, the more CO2 the less overall effect each additional ppm causes. The reason for this has to do with the wavelengths that CO2 absorbs, and at some point it has absorbed nearly all of the energy in the wavelengths it absorbs.
That's true and relevant to the OP's question of how much CO2 there needs to be to have effect. Below is a little graph I made (click here to see it in better quality) showing the warming effect from successive 20ppmv increments. The first 20ppmv of CO2 does the majority of warming and after that it has much less effect, assuming one accepts the veracity of the Arrhenius equation.

edit on 31-12-2018 by Nathan-D because: (no reason given)
*Double-Post*
edit on 31-12-2018 by Nathan-D because: (no reason given)
I did find this
I did find
Using 50% to be conservatively low on the amount of inferred energy of the 1050 W/m2 from the sun comes to 525 w/m2. Supposedly “greenhouse gas radiation had increased by 3.5 watts per square meter”
3.5 W/m2 is .7 percent of the 525W/m2 infrared energy the sun directs at the earth.
The infrared retention can easily be explained by:
Side note, to combat that “3.5 W/m2” increase in infrared retention, just use “Gold, manganese and copper also absorb IR radiation well. According to Next Energy News, the U.S. Department of Energy is using these three metals to design nanoantennas, which will convert waste heat in the form of IR radiation into electricity”
Anyway, this is my beef with articles like “Carbon Dioxide Is Warming the Planet (Here's How)”
One, they give a fact like “an increase of 3.5W/m2 infrared in the atmosphere compared to preindustrial times.” But never give the context of how much infrared energy the Earth is bathed in from the sun which is around 525 Watts a square meter. (And how do they know for sure the amount of infrared retention preindustrial revolution?)
Two, the article completely ignores rising sea levels will absorb more IR. (One must wonder if there is a connection with IR absorbing and CO2 released by the oceans.) Less snow means less IR reflected to space. Increased water vapor release by increased cooling tower usage results in more IR absorption. Then take in account the increase amount of pavement, concrete, and glass covering the earth absorbing more IR.
But the article claims “Carbon Dioxide Is Warming the Planet (Here's How)” is all because of CO2, and ignores other factors like they don’t exist.
Three. The science of the article is backwards. They give a fact of how much less IR radiation is making it to space, then blame CO2. Good science would be first create a model using the known properties of CO2 to come up with a theoretical amount of how much a given increase in CO2 in the atmosphere will resort in how much of an increase in IR retention. Then that should be compared to what is actually occurring.
What you have is basically this. Saying temperatures are increasing, so it has to be because CO2 is rising is like saying the Earth is the center of the universe because everything appears to orbit the earth.
Carbon Dioxide Is Warming the Planet (Here's How)
www.livescience.com...
Overall, they found that greenhouse gas radiation had increased by 3.5 watts per square meter compared with preindustrial times, a rise of just over 2 percent. Other researchers have noted "missing" infrared wavelengths in radiation into space, a phenomenon that happens because these missing wavelengths get stuck in the atmosphere.
I did find
en.m.wikipedia.org...
If the extraterrestrial solar radiation is 1367 watts per square meter (the value when the Earth–Sun distance is 1 astronomical unit), then the direct sunlight at Earth's surface when the Sun is at the zenith is about 1050 W/m2, but the total amount (direct and indirect from the atmosphere) hitting the ground is around 1120 W/m2.[5] In terms of energy, sunlight at Earth's surface is around 52 to 55 percent infrared (above 700 nm), 42 to 43 percent visible (400 to 700 nm), and 3 to 5 percent ultraviolet (below 400 nm).[6]
Using 50% to be conservatively low on the amount of inferred energy of the 1050 W/m2 from the sun comes to 525 w/m2. Supposedly “greenhouse gas radiation had increased by 3.5 watts per square meter”
3.5 W/m2 is .7 percent of the 525W/m2 infrared energy the sun directs at the earth.
The infrared retention can easily be explained by:
Materials That Absorb Infrared Rays
sciencing.com...
Earth’s atmosphere absorbs the majority of the IR radiation that comes from the sun. In addition, carbon dioxide, ozone and oxygen also absorb much of the IR radiation, allowing very little to actually reach the ground. Aside from water vapor, bodies of water on the surface of the Earth also absorb IR wavelengths well. Glass, Plexiglas, wood, brick, stone, asphalt and paper all absorb IR radiation. While regular silver-backed mirrors reflect visible light waves, allowing you to see your reflection, they absorb infrared radiation. Gold, manganese and copper also absorb IR radiation well. According to Next Energy News, the U.S. Department of Energy is using these three metals to design nanoantennas, which will convert waste heat in the form of IR radiation into electricity.
Side note, to combat that “3.5 W/m2” increase in infrared retention, just use “Gold, manganese and copper also absorb IR radiation well. According to Next Energy News, the U.S. Department of Energy is using these three metals to design nanoantennas, which will convert waste heat in the form of IR radiation into electricity”
Anyway, this is my beef with articles like “Carbon Dioxide Is Warming the Planet (Here's How)”
One, they give a fact like “an increase of 3.5W/m2 infrared in the atmosphere compared to preindustrial times.” But never give the context of how much infrared energy the Earth is bathed in from the sun which is around 525 Watts a square meter. (And how do they know for sure the amount of infrared retention preindustrial revolution?)
Two, the article completely ignores rising sea levels will absorb more IR. (One must wonder if there is a connection with IR absorbing and CO2 released by the oceans.) Less snow means less IR reflected to space. Increased water vapor release by increased cooling tower usage results in more IR absorption. Then take in account the increase amount of pavement, concrete, and glass covering the earth absorbing more IR.
But the article claims “Carbon Dioxide Is Warming the Planet (Here's How)” is all because of CO2, and ignores other factors like they don’t exist.
Three. The science of the article is backwards. They give a fact of how much less IR radiation is making it to space, then blame CO2. Good science would be first create a model using the known properties of CO2 to come up with a theoretical amount of how much a given increase in CO2 in the atmosphere will resort in how much of an increase in IR retention. Then that should be compared to what is actually occurring.
What you have is basically this. Saying temperatures are increasing, so it has to be because CO2 is rising is like saying the Earth is the center of the universe because everything appears to orbit the earth.
a reply to: neutronflux
So, even with Mars' atmospheric pressure only being .6% of Earth's and its atmosphere being "mostly" CO2, would that equal out to a similar amount of total CO2 in the air that is available to trap hear, much less, or much more that Earth's?
I think that's a big part of your question and maybe part of the implication, because if the total amount is still more than that on Earth, one might conclude that it's obvious that CO2 does little to trap in the heat and limit temperature fluctuations. If it's far less, one might conclude that it's obvious that it's the limited amount of CO2 compared to earth that allows such dramatic fluctuations.
Personally, after decades of reading and researching this topic, I have concluded that water vapor (clouds) and solar activity has MUCH more to do with Earth's temperatures than the 0.04% of CO2 that is in the atmosphere.
I hope that you find an answer to the question--I remember reading it once, but didn't commit the answer nor the source to memory.
So, even with Mars' atmospheric pressure only being .6% of Earth's and its atmosphere being "mostly" CO2, would that equal out to a similar amount of total CO2 in the air that is available to trap hear, much less, or much more that Earth's?
I think that's a big part of your question and maybe part of the implication, because if the total amount is still more than that on Earth, one might conclude that it's obvious that CO2 does little to trap in the heat and limit temperature fluctuations. If it's far less, one might conclude that it's obvious that it's the limited amount of CO2 compared to earth that allows such dramatic fluctuations.
Personally, after decades of reading and researching this topic, I have concluded that water vapor (clouds) and solar activity has MUCH more to do with Earth's temperatures than the 0.04% of CO2 that is in the atmosphere.
I hope that you find an answer to the question--I remember reading it once, but didn't commit the answer nor the source to memory.
new topics
-
BIDEN Admin Begins Planning For January 2025 Transition to a New President - Today is 4.26.2024.
2024 Elections: 2 hours ago -
Big Storms
Fragile Earth: 4 hours ago -
Where should Trump hold his next rally
2024 Elections: 6 hours ago -
Shocking Number of Voters are Open to Committing Election Fraud
US Political Madness: 7 hours ago -
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections: 8 hours ago -
Falkville Robot-Man
Aliens and UFOs: 8 hours ago -
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 9 hours ago -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 10 hours ago -
Ireland VS Globalists
Social Issues and Civil Unrest: 10 hours ago -
Biden "Happy To Debate Trump"
2024 Elections: 11 hours ago
top topics
-
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 9 hours ago, 17 flags -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 10 hours ago, 15 flags -
Blast from the past: ATS Review Podcast, 2006: With All Three Amigos
Member PODcasts: 13 hours ago, 13 flags -
Biden "Happy To Debate Trump"
2024 Elections: 11 hours ago, 13 flags -
Ireland VS Globalists
Social Issues and Civil Unrest: 10 hours ago, 9 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 13 hours ago, 8 flags -
Shocking Number of Voters are Open to Committing Election Fraud
US Political Madness: 7 hours ago, 6 flags -
BIDEN Admin Begins Planning For January 2025 Transition to a New President - Today is 4.26.2024.
2024 Elections: 2 hours ago, 6 flags -
What is the white pill?
Philosophy and Metaphysics: 12 hours ago, 6 flags -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 11 hours ago, 5 flags
active topics
-
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs • 12 • : pianopraze -
BIDEN Admin Begins Planning For January 2025 Transition to a New President - Today is 4.26.2024.
2024 Elections • 14 • : WeMustCare -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order • 29 • : 19Bones79 -
Big Storms
Fragile Earth • 15 • : Caver78 -
What is the white pill?
Philosophy and Metaphysics • 22 • : AlexandrosOMegas -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 315 • : CriticalStinker -
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections • 58 • : cherokeetroy -
Biden "Happy To Debate Trump"
2024 Elections • 51 • : rickymouse -
Hate makes for strange bedfellows
US Political Madness • 53 • : 19Bones79 -
SETI chief says US has no evidence for alien technology. 'And we never have'
Aliens and UFOs • 79 • : SchrodingersRat
