It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
originally posted by: intrptr
a reply to: soficrow
RE: "In West Africa, pigs commonly dig up Ebola victims' graves. Thing is, pigs catch Ebola and worst of all - Ebola transmission in pigs is airborne."
F--- me!
Talk about vectoring.
....I can't even imagine what other scenarios like this might be undiscovered as yet. ...
Birds fly a long ways on migrations, for instance. I wonder if the vultures can get it from carrion like dead pigs that get it from like dead humans? ...
uh huh. Take the Ebola-carrier fruit bat, for instance.
Fruit bats: Africa’s greatest mammal migration
Forget the wildebeest of the Serengeti – Africa’s greatest mammal migration involves 8 million animals and takes place in the air.
….The bats are frequently attacked by birds of prey, such as fish eagles, while crocodiles snap up any unfortunate individuals that fall to the forest floor.
….In the 1980s, DW Thomas of Aberdeen University used sightings to estimate that these mammals travelled some 1,500km during the course of a year. But when Heidi monitored their movements, she discovered this to be a major underestimation.
She fitted four individuals with satellite-tracking collars, then plotted their movements on a map. She found that after their departure in December, her study bats had covered a whopping 1,000km in just one month. “They disappeared off the radar somewhere over the Democratic Republic of Congo (DRC),” says Heidi. “The collars could have been lost or damaged, or the bats eaten.”
However, previous data showed that one particular bat, named Hercules, had travelled 1,900km in six months. The DRC may not even have been his final destination – with a return trip to Kasanka later in the year, Hercules would have made a round trip of at least 3,800km, a staggering distance. The full route will not be known until more tracking studies are carried out.
a reply to: intrptr
From another thread:
]originally posted by: jadedANDcynical
a reply to: Drinthearctic
I know this wasn't addressed to me, but seeing as I recently went looking for the answer to that particular question myself, I hope you don't mind if I share what I've found:
From a google scholar search summary of:
Mosquitoes and People: Environmental Ethics Concerns
From another thread:
]originally posted by: jadedANDcynical
a reply to: Drinthearctic
Also wondering if mosquitos can spread it like malaria?
I know this wasn't addressed to me, but seeing as I recently went looking for the answer to that particular question myself, I hope you don't mind if I share what I've found:
1 However, laboratory work indicates mosquitoes are unlikely to be transmitters of Ebola virus.
From a google scholar search summary of:
Mosquitoes and People: Environmental Ethics Concerns
a reply to: intrptr
Recognized insect 'transmitters' tend to 'sequester' viruses inside their bodies/cells - and bugs are not on the list for transmitting Ebola in this way. ...We've already discussed the potential for more casual transportation.
Recognized insect 'transmitters' tend to 'sequester' viruses inside their bodies/cells - and bugs are not on the list for transmitting Ebola in this way. ...We've already discussed the potential for more casual transportation.
Transmission
Viruses that cause haemorrhagic fevers are transmitted by mosquitoes (dengue, yellow fever, RVF), ticks (CCHF), rodents (Hantavirus, Lassa) or bats (Ebola, Marburg). For Ebola and Marburg viruses, humans have been infected from contact with tissues of diseased non-human primates (monkeys and apes) and other mammals, but most human infections have resulted from direct contact with the body fluids or secretions of infected patients. Humans who develop CCHF usually become infected from a tick bite but can also acquire the virus from direct contact with blood or other infected issues from livestock or from infected patients. RVF can be acquired either by mosquito bite or by direct contact with blood or tissues of infected animals (mainly sheep), including consumption of unpasteurized milk. Lassa fever virus is carried by rodents and transmitted by excreta, either as aerosols or by direct contact. Some viral haemorrhagic fevers have been amplified in hospitals by nosocomial transmission resulting from unsafe procedures, use of contaminated medical devices (including needles and syringes) and unprotected exposure to contaminated body fluids.
a reply to: jadedANDcynical
I'd like to believe that, but I didn't see the "pigs digging up Ebola graves", either.
Thanks for the reply. soficrow has another link about it above this post.
I'd like to believe that, but I didn't see the "pigs digging up Ebola graves", either.
Thanks for the reply. soficrow has another link about it above this post.
a reply to: intrptr
Sofi's post seems to indicate the same thing. The way viruses generally transmit via an insect vector is by becoming encapsulated within the cells of the insect through biological processes. That being the case, ebola is unlikely to be transmitted via that method.
Now, I could conceive that a mosquito biting an infected person, and then through contacting the sweat of the victim, transmitting the virus to a new host by it's feet when it lands on an uninfected person; the same could be said of any insect.
As far as the pigs digging up the bodies, it makes sense that any scavenger would take advantage of a shallow grave containing a (to it's mind) feast. Hyenas, vultures, and a host of other animals which feed on carrion would be liable to spread the disease equally.
All of that being said, I did find this older research which raises several alarming possibilities. Let me state that the author indicates that everything within is somewhat outside the norm of the thinking about filoviruses at the time:
In other words, he'd fit in quite well right here at ATS.
What is alarming about his conjecture? Here are a couple of examples:
In other words what shows up as Ebola in humans, might not be Ebola in other animals, but is something else that mutates into ebola and then appears in humans.
Of course, this is non-traditional thinking, but something to be considered as a possibility.
How would this happen, and are there other examples of what is being suggested?
That's quite a bit to wrap one's brain around, but the following should be easy enough to grasp:
Bushmeat, but sofi shows that the research indicates that all subsequent transmission beyond the index case has been human to human with no further zoonotic reinfection occurring.
More from the paper:
Holy random intermittent occurrences, Batman!
More specifically to the question of insect vectoring:
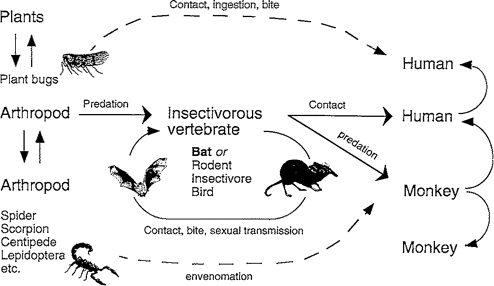
So mixed results of very incomplete studies. All subtypes need to be tested against all possible vectors present within the potentially affected area(s).
Ecology of Marburg and Ebola Viruses: Speculations and Directions for Future Research
Sofi's post seems to indicate the same thing. The way viruses generally transmit via an insect vector is by becoming encapsulated within the cells of the insect through biological processes. That being the case, ebola is unlikely to be transmitted via that method.
Now, I could conceive that a mosquito biting an infected person, and then through contacting the sweat of the victim, transmitting the virus to a new host by it's feet when it lands on an uninfected person; the same could be said of any insect.
As far as the pigs digging up the bodies, it makes sense that any scavenger would take advantage of a shallow grave containing a (to it's mind) feast. Hyenas, vultures, and a host of other animals which feed on carrion would be liable to spread the disease equally.
All of that being said, I did find this older research which raises several alarming possibilities. Let me state that the author indicates that everything within is somewhat outside the norm of the thinking about filoviruses at the time:
The enigma surrounding the natural transmission cycles of Ebola (EBO) and Marburg (MBG) viruses remains a challenge for scientific research. Since my charge in preparing this paper was to present hypotheses, to stimulate nonlinear thinking, and to suggest new avenues for investigation, what follows is speculation, extrapolation, and conjecture. The reader should keep in mind that certain hypotheses may not be consistent with the prevailing views of some filovirologists.
In other words, he'd fit in quite well right here at ATS.
What is alarming about his conjecture? Here are a couple of examples:
An interesting possibility is that filoviruses may be arthropod or plant viruses, with non—blood-feeding arthropods transmitting the virus to intermediate hosts or humans during oral ingestion or envenomation. Paradoxically, in Africa, Ebola virus disease has high lethality and high seroprevalence as determined by the IFA test. If the seroreactivity is confirmed by more specific tests, then the Ebola virus serogroup in Africa probably contains an antigenically cross-reactive, enzootic, nonpathogenic agent(s). Such viruses may have separate life cycles or may give rise to virulent strains by mutation.
In other words what shows up as Ebola in humans, might not be Ebola in other animals, but is something else that mutates into ebola and then appears in humans.
Of course, this is non-traditional thinking, but something to be considered as a possibility.
How would this happen, and are there other examples of what is being suggested?
A possible explanation for the infrequent emergence of virulent EBO strains is that these viruses arise by gradual or stepwise mutation from an “enzootic” virus rather than being sustained in a cryptic maintenance cycle. The analog in arbovirology is the alphavirus, Venezuelan equine encephalitis, which is transformed by stepwise mutation from a nonpathogenic enzootic virus (ID subtype) to a virulent epizootic virus (IC subtype) having a different range of vectors and hosts [54]. Single-site mutations can give rise to profound shifts in virulence. Variants of rabies virus selected by monoclonal antibodies [55] or chemical mutagenesis [56) were found to be nonpathogenic on the basis of a specific, single amino acid change (arginine to glutamine) at position 333 in the G protein. It is possible that a single-site mutation or a few stepwise mutations could explain the sudden conversion of a nonpathogenic to a virulent EBO strain.
That's quite a bit to wrap one's brain around, but the following should be easy enough to grasp:
Bats (particularly solitary microchiropteran species) are leading contenders as reservoir hosts. Virus transfer to humans occurs by contact with the primary reservoir or via an intermediate animal that acquired infection from the reservoir and is, in turn, hunted by humans.
Bushmeat, but sofi shows that the research indicates that all subsequent transmission beyond the index case has been human to human with no further zoonotic reinfection occurring.
More from the paper:
As will be pointed out later, it is possible that the filoviruses represent a diverse complex of agents with different transmission cycles, as is the case for the lyssavirus and vesiculovirus genera, raising the difficulty of interpreting heterologous cross-reacting antibodies. Virulent strains of EBO and MBG virus may not persist in a local reservoir but rather cause wandering epizootics that return at long intervals to any one location. It is also possible that the virulent filovirus strains do not have sustained transmission cycles at all but arise from time to time by mutation from enzootic variants.
Holy random intermittent occurrences, Batman!
More specifically to the question of insect vectoring:
The susceptibility to infection of arthropod taxa can be assessed by virus replication after intrathoracic inoculation of virus. Turell et al. [8] reported that EBO virus, subtype Reston (EBO-R), failed to replicate in Culex or Aedes mosquitoes and in Ornithodoros ticks. However, Kunz et al. [9] previously reported that MBG virus could persist in Aedes mosquitoes for 3 weeks or more, indicating that certain arthropods exposed towasp.Altho the virus could be transient or persistent carriers of infection. Many potential blood-feeding arthropod vectors (phlebotomine flies, culicoids, ixodid ticks, mites, fleas, and wingless flies associated with bats) have not been tested by experimental inoculation. The susceptibility of insects used by humans or wild vertebrate reservoir hosts as a source of food (including termites, moths, and larvae [grubs]) also has not been explored. As will be discussed below, there has been recent speculation about the possible role of leafhoppers in filovirus ecology; therefore, experimental studies should determine the host range of EBO virus for plant-feeding bugs.

So mixed results of very incomplete studies. All subtypes need to be tested against all possible vectors present within the potentially affected area(s).
EBO and MBG viruses are similar in genome organization and have extensive sequence homology but do not share cross-reactive antigens [33]. The ecology of EBO viruses is certainly more complex than that of MBG. Operational hypotheses on the ecology of EBO virus must take into account the factors discussed below.
Geographic distribution
At least 4 genetic subtypes of EBO virus have been recognized: Zaire (EBO-Z), Côte d'Ivoire (EBO-CI) [34], Sudan (EBO-S) [35], and EBO-R. EBO-R was recovered in the Philippines [36] and from monkeys imported from the Philippines to the United States and Europe.
...
The occurrence of a distinct EBO virus subtype in the Philippines has important implications for filovirus ecology, since it implies that EBO group viruses may have been carried by migratory hosts at some point in their evolution.
Ecology of Marburg and Ebola Viruses: Speculations and Directions for Future Research
a reply to: jadedANDcynical
Awesome post. Like the out of the box approach. We have never been here with Ebola free ranging in populated areas. Ebola is definitely out of the box.
If you read the HotZone by Richard Preston they surmise that a couple of past outbreaks began as near as they can tell in or around a place called Kitum cave (spelling). Its a strange place, not formed by water but over centuries, completely by elephants goring for salt minerals in the cave walls. Every kind of critter, mold and crystal grows there, many explores and medical teams have gone there looking for Ebola and have never found the repository.
Hella read if you got the time…
Awesome post. Like the out of the box approach. We have never been here with Ebola free ranging in populated areas. Ebola is definitely out of the box.
]
In other words what shows up as Ebola in humans, might not be Ebola in other animals, but is something else that mutates into ebola and then appears in humans.
If you read the HotZone by Richard Preston they surmise that a couple of past outbreaks began as near as they can tell in or around a place called Kitum cave (spelling). Its a strange place, not formed by water but over centuries, completely by elephants goring for salt minerals in the cave walls. Every kind of critter, mold and crystal grows there, many explores and medical teams have gone there looking for Ebola and have never found the repository.
Hella read if you got the time…
new topics
-
Ireland VS Globalists
Social Issues and Civil Unrest: 20 minutes ago -
Biden "Happy To Debate Trump"
Mainstream News: 51 minutes ago -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 1 hours ago -
What is the white pill?
Philosophy and Metaphysics: 2 hours ago -
Mike Pinder The Moody Blues R.I.P.
Music: 3 hours ago -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 6 hours ago -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 10 hours ago
top topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies: 16 hours ago, 34 flags -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 10 hours ago, 18 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 3 hours ago, 7 flags -
What is the white pill?
Philosophy and Metaphysics: 2 hours ago, 5 flags -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 1 hours ago, 4 flags -
Biden "Happy To Debate Trump"
Mainstream News: 51 minutes ago, 4 flags -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 6 hours ago, 2 flags -
Ireland VS Globalists
Social Issues and Civil Unrest: 20 minutes ago, 1 flags
active topics
-
Is AI Better Than the Hollywood Elite?
Movies • 22 • : 5thHead -
Biden "Happy To Debate Trump"
Mainstream News • 14 • : Hakaiju -
King Charles 111 Diagnosed with Cancer
Mainstream News • 324 • : Oldcarpy2 -
Ireland VS Globalists
Social Issues and Civil Unrest • 2 • : TimBurr -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works • 20 • : Hakaiju -
President Biden is Angry at A.G. Merrick Garland for Not Being Loyal to the Biden Family.
Above Politics • 20 • : WeMustCare -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs • 4 • : gortex -
New whistleblower Jason Sands speaks on Twitter Spaces last night.
Aliens and UFOs • 75 • : baablacksheep1 -
What is the white pill?
Philosophy and Metaphysics • 15 • : Astyanax -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 278 • : cherokeetroy
