It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
Hi all, Sometimes we dont know and even dont care what happen around us, but i bet if you see this spectacular image you will say WOW i dont know that
can be so Amazing!


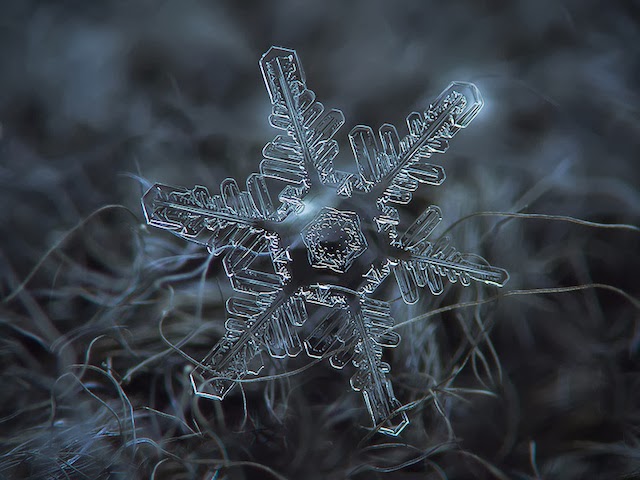


Event in Very Strange Shape :




what you say, this Snow shape lookslike something very strange, i wonder if this shape like a code, or something like that
altering-perspectives.com...





Event in Very Strange Shape :




Photographer Alexey Kljatov takes incredible close-up photos of snowflakes in his backyard in Moscow.
“I capture snowflakes on the open balcony of my house, mostly on glass surface, lighted by an LED flashlight from the opposite side of the glass, and sometimes in natural light, using dark woolen fabrics as background.”
what you say, this Snow shape lookslike something very strange, i wonder if this shape like a code, or something like that
altering-perspectives.com...
reply to post by cheesy
These are absolutely beautiful thank you for sharing! Its wonderful the different captivating patterns that nature can produce for us. *drooling*
These are absolutely beautiful thank you for sharing! Its wonderful the different captivating patterns that nature can produce for us. *drooling*
reply to post by cheesy
S&F, I like the pics very much. I remember in grade school for class we would catch them on cold black construction paper, and then try to draw them the best we could to see if there are two alike.
Can you believe that where I am in Alaska its raining.......
Sigh. I miss the white stuff
S&F, I like the pics very much. I remember in grade school for class we would catch them on cold black construction paper, and then try to draw them the best we could to see if there are two alike.
Can you believe that where I am in Alaska its raining.......
Sigh. I miss the white stuff
edit on 8-12-2013 by AK907ICECOLD because: (no reason given)
Super cool pics cheesy
Sorry about your comet btw I was hoping for a good show just for you cos you deserved it
Sorry about your comet btw I was hoping for a good show just for you cos you deserved it
Cheesy, you are Mr. Cool. S&F
Here's some more of Alexey's work. Including more snowflakes. Alexey Kljatov
Here's some more of Alexey's work. Including more snowflakes. Alexey Kljatov
I honestly believe it has to do with frequency resonance in the atmosphere. Sound creates phenomenal shapes and the ice happens to freeze it perfectly
in space and time. Just a theory, no proof or anything.
Beyond being spectacularly beautiful to look at, these shapes allow us to see, in close up, what wondrous shapes are created under no outside influences. They are absolutely the most perfect shapes and every single one is original. Impressive to say the least.
Excellent pictures!
*S&F*
Beyond being spectacularly beautiful to look at, these shapes allow us to see, in close up, what wondrous shapes are created under no outside influences. They are absolutely the most perfect shapes and every single one is original. Impressive to say the least.
Excellent pictures!
*S&F*
boymonkey74
Super cool pics cheesy
Sorry about your comet btw I was hoping for a good show just for you cos you deserved it
haha dont wory about Ison, He is fine..Just Sleep a while..
Do you see the patern? how can that patern make? very singular form..very wonderful World we live in..
Klassified
Cheesy, you are Mr. Cool. S&F
Here's some more of Alexey's work. Including more snowflakes. Alexey Kljatov

Wowww look at that..how can nature produce something very tiny beautiful like that..
Tq for the Link Sir..!
reply to post by cheesy
Here cheesy.
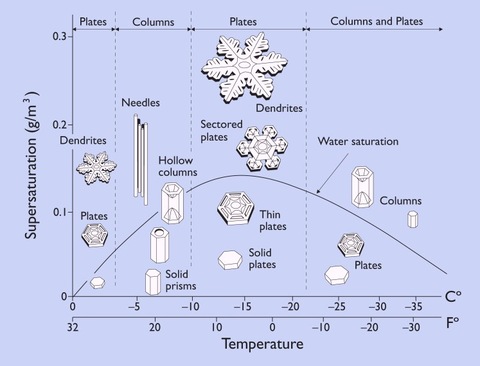
Maybe it is the frequency produced by thermal radiation between varying temperatures. The visible spectrum will produce a certain kind of flake where as higher frequencies makes a more complex one.
Here cheesy.
Water molecules -- made of one oxygen and two hydrogen atoms each -- are ultimately responsible for the familiar six-sided shape we associate with snowflakes.
At low humidity you get simple plates and simple hexagonal blocks, Libbrecht said. At higher humidity, more branched structures.

But the physics is far from exact, Libbrecht said.
"It's a mystery as to why [snowflake shapes] go from plates to columns to plates to columns as the temperature lowers," he said. "That's one of the things I've been trying to understand. It has been a mystery for about 75 years, and it's still unsolved."
Maybe it is the frequency produced by thermal radiation between varying temperatures. The visible spectrum will produce a certain kind of flake where as higher frequencies makes a more complex one.
edit on 8-12-2013 by eisegesis because: (no reason given)
eisegesis
reply to post by cheesy
Here cheesy.
wow thats amazing! so that how the mystery solve..TQ sir!
Every one seems to have six sides with a six pointed star in the middle.
What does this MEAN?
What does this MEAN?
ItCameFromOuterSpace
Every one seems to have six sides with a six pointed star in the middle.
What does this MEAN?
I dont Know what its mean by streching it arm by 6,.ummm yeah its mystery my friend..
reply to post by ItCameFromOuterSpace
It's called six-fold radial symmetry an in snowflakes, it results from the underlying crystalline structure of (water) ice.
source
More detailed:
source
It's called six-fold radial symmetry an in snowflakes, it results from the underlying crystalline structure of (water) ice.
The six-fold symmetry of a snow crystal ultimately derives from the hexagonal geometry of the ice crystal lattice. But the lattice has molecular dimensions, so it's not trivial how this nano-scale symmetry is transferred to the structure of a large snow crystal.
The way it works is through faceting. No long-range forces are necessary to form facets; they appear simply because of how the molecules hook up locally in the lattice (see Crystal Faceting for how this works). From faceting we get hexagonal prisms, which are large structures with six-fold symmetry. Eventually arms sprout from the corners of a prism, and six corners means six arms.
Faceting is how the geometry of the water molecule is transferred to the geometry of a large snow crystal.
source
More detailed:
These bonds make water an unusual substance. When the temperature drops and water starts to solidify, the hydrogen bonding becomes very important. The hydrogen bonding dictates the shape of the ice crystals. You’ve learned that each water molecule is linked to four other water molecules in a tetrahedral arrangement.
As the water freezes, these tetrahedrons come closer together and crystallize into a six-ring or hexagonal structure. Look at the image to see how this happens. Each point on the hexagon is an oxygen atom, and each side is a hydrogen bonded to one oxygen. As the water approaches freezing temperature, the water molecules continue to crystallize in this tetrahedral arrangement.
But water does something unlike most substances. As it nears freezing, instead of continuing to contract, it expands slightly from about 4 degrees to 0 degrees Celsius as the motion of the molecules slows with the cold, and the hydrogen bonds extend the molecules to their fullest distance from each other. It’s like a ring of people holding hands, elbows bent, and then gradually straightening their arms to the fullest extension so that they’re at the greatest distance from each other. When water molecules do this, the hexagonal structure expands into a larger and larger hexagonal structure.
source
edit on 8-12-2013 by theantediluvian because: (no reason
given)
reply to post by cheesy
Because nature and the universe it is encompassed within is the very expression of beautiful. Everything, From the Microcosmic to the Macrocosmic is amazingly beautiful in one way or another. Even deadly virus' under intense magnification can be beautiful.
Because nature and the universe it is encompassed within is the very expression of beautiful. Everything, From the Microcosmic to the Macrocosmic is amazingly beautiful in one way or another. Even deadly virus' under intense magnification can be beautiful.
reply to post by cheesy
S&F for Cheesy! Hey snowflake geometry is very KOOL! I didn't see my first snowflake until I was 10-years-old when I went to Winter Camp and boy oh boy was I a happy camper!!!!! It was magical.
Thank you for inspiring me this Holiday Season. Now every time I see a snowman I will think of this thread.
Ta ta!
Toni
S&F for Cheesy! Hey snowflake geometry is very KOOL! I didn't see my first snowflake until I was 10-years-old when I went to Winter Camp and boy oh boy was I a happy camper!!!!! It was magical.
Thank you for inspiring me this Holiday Season. Now every time I see a snowman I will think of this thread.
Ta ta!
Toni
dub post
edit on PM123120131211pm0931pm by Antoniastar because: (no reason given)
reply to post by cheesy
Breathtaking pics Cheezy. Best thing I've seen in a while. I don't care what kind of science we come up with to explain these things, the fact that they are SO beautiful to us makes you wonder why... what's the purpose of all this beauty if not to be appreciated by beings such as ourselves?
S+F Nice job
Breathtaking pics Cheezy. Best thing I've seen in a while. I don't care what kind of science we come up with to explain these things, the fact that they are SO beautiful to us makes you wonder why... what's the purpose of all this beauty if not to be appreciated by beings such as ourselves?
S+F Nice job
edit on 8-12-2013 by Bone75 because: (no reason given)
These are cool and all, but just because they have six branches doesn't make them sacred geometry.
I wonder if mother earth is responsible for making crop circles, like she is responsible for making snowflakes? That one snowflake definitely looked
like it had code embedded in it.
edit on 9-12-2013 by jaws1975 because: (no reason given)
new topics
-
In four years, you don’t have to vote again.
2024 Elections: 1 hours ago -
Prize or Zonk
General Chit Chat: 3 hours ago -
I've realized something about Politics recently.
US Political Madness: 5 hours ago -
France bans head coverings but allows Christianity to be mocked at Olympics.
Political Conspiracies: 7 hours ago -
Woo time it aint over yet
Paranormal Studies: 8 hours ago -
BIDEN-HARRIS Preparing to Also Make Us Pay For Illegal-Aliens to Attend College Prep Programs.
Political Ideology: 11 hours ago
top topics
-
The Main Question and A Bonus Question
General Conspiracies: 13 hours ago, 13 flags -
Is Kamala Good for America ?
Political Issues: 13 hours ago, 12 flags -
BIDEN-HARRIS Preparing to Also Make Us Pay For Illegal-Aliens to Attend College Prep Programs.
Political Ideology: 11 hours ago, 9 flags -
Did Russia just attack a NATO nation?
The Gray Area: 16 hours ago, 6 flags -
The Most Crime Ridden Games Ever
World Sports: 13 hours ago, 6 flags -
In four years, you don’t have to vote again.
2024 Elections: 1 hours ago, 5 flags -
A Preliminary Question
General Conspiracies: 14 hours ago, 3 flags -
Olympics France 2024 Opening Ceremony
World Sports: 14 hours ago, 3 flags -
Woo time it aint over yet
Paranormal Studies: 8 hours ago, 3 flags -
France bans head coverings but allows Christianity to be mocked at Olympics.
Political Conspiracies: 7 hours ago, 2 flags
active topics
-
The Acronym Game .. Pt.4
General Chit Chat • 145 • : SecretKnowledge2 -
In four years, you don’t have to vote again.
2024 Elections • 7 • : wAnchorofCarp -
Shots Fired At Trump Rally Trump Looks Wounded
US Political Madness • 1849 • : daskakik -
The Most Crime Ridden Games Ever
World Sports • 6 • : SecretKnowledge2 -
Olympics France 2024 Opening Ceremony
World Sports • 11 • : putnam6 -
France bans head coverings but allows Christianity to be mocked at Olympics.
Political Conspiracies • 22 • : BedevereTheWise -
I've realized something about Politics recently.
US Political Madness • 7 • : wAnchorofCarp -
Prediction watch how fast the war in Ukraine ends.
Dreams & Predictions • 65 • : annonentity -
Lue Elizondo Announces New UFO Book "IMMINENT" Coming August 2024
Aliens and UFOs • 53 • : baablacksheep1 -
Is Kamala Good for America ?
Political Issues • 193 • : covent
