It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
in science we started learning about atoms and such and me and a mate thought that if the nucleus of an atom is the center and electrons orbit around
it (much like our universe) that our whole universe IS an atom making up something else. we took this as something stupid and of course started going
haha im killing universes while poking things but... gravity what is it? electro magnetic force???. positiveley charged electrons spinning round and
round the center (the sun). anything that me and another friend think up to prove this wrong we find a way to make it possible. if anyone agrees or
knows some way how this is noway possible please reply. because of limited knowlege about atoms, and we cant see close enough to see just something
else, means that it could be possible. as i said anyone know anything to make this wrong??
thanks
DaNReD
thanks
DaNReD
my awesome diagram (not a joke btw its just so u understand me)
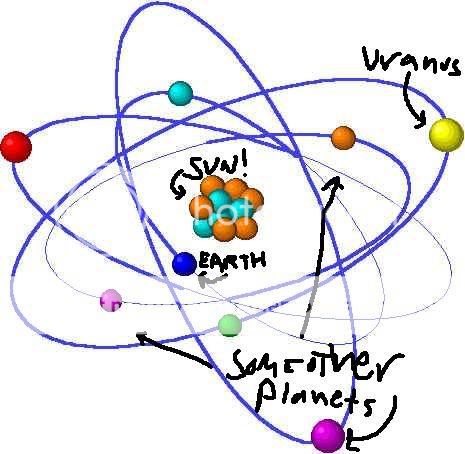

I've often contemplated that our solar system resembles an atom, with the sun as the nucleus and the planets electrons, this would make our galaxy a
molecule and the universe would be the whole thing (whatever that would be).
This would also make sense of the big bang theory (the moment of conception) and explain the fact that everything in the universe is expanding outwards (the act of growing).
This led me to the question of whether humanity is a type of minuscule parasite infecting "god" for want of a better word, then I give up because it makes my head hurt. lol
It's just a theory and we'll never know.
This would also make sense of the big bang theory (the moment of conception) and explain the fact that everything in the universe is expanding outwards (the act of growing).
This led me to the question of whether humanity is a type of minuscule parasite infecting "god" for want of a better word, then I give up because it makes my head hurt. lol
It's just a theory and we'll never know.
its very possible thing in nature tend to repeat itself in bigger bolder ways an alot works the same way as other things... so Yes I could totally see
this. takes you back tot he men in black thing with our universe is inside a bus station locker box.
maybe we are a puddle in another great galaxy that is a drop of water from a leaky sink in another bigger universe an so forth....
if you wanna go insane, just keep thinkin of where our universe ends......
maybe we are a puddle in another great galaxy that is a drop of water from a leaky sink in another bigger universe an so forth....
if you wanna go insane, just keep thinkin of where our universe ends......
The model of the atom being referenced is called the Bohr model and is VERY outdated. The similarity is actually caused because the Bohr model is
BASED on the solar system.
The scientific community has long since moved on to more accurate models.
The scientific community has long since moved on to more accurate models.
An atom is an atom (its already claimed its territory) so the universe is something else entirely . In other words what ever it is it would have to
have another name or you'd get the two confused.
Scientist have enough troubles without giving two different concepts the same name.
But its not the first time somebody wondered what you's are a wondering
Scientist have enough troubles without giving two different concepts the same name.
But its not the first time somebody wondered what you's are a wondering
if so then...
why did they base it on the universe in the first place?
because the information they had was much similar?
i really dont know wot im talking about but anyway.
its a possibility. someone in another universe containing ours could just breathe the air around them and we would be dead and we wouldnt even know it. anyways thanks for replies
DaN
why did they base it on the universe in the first place?
because the information they had was much similar?
i really dont know wot im talking about but anyway.
its a possibility. someone in another universe containing ours could just breathe the air around them and we would be dead and we wouldnt even know it. anyways thanks for replies
DaN
Your drawing looks like a carbon atom which is the basic element for life. I think our Solar System looks more like an atom with the Sun being the
Nucleus. And there are probably atoms we haven't studied out there in our vast universe. Since we are part of the same energy we have the smallest
atom up to the biggest atom. Maybe our universe is one big molecule and our Solar System along with others are the atoms with protons, electrons
etc.. I've thought of this too and think it all looks the same.
[edit on 4/25/2008 by Solarskye]
[edit on 4/25/2008 by Solarskye]
reply to post by DaNReD
oh wow great ponderings !!
Just the other day I was thinking something very similar.
(pity the links don't come up when quoting but never mind...you will have to click on the link to see the images, oh thats if you want to of course)
I really wonder now if we are nothing more than energy within energy and perhaps as small as an atom? And all the atoms make up one big atom... such as what you have described !? I know, sounds a bit way out there?
starred and flagged!
Thanks for sharing your thoughts!
[edit on 25-4-2008 by Thurisaz]
oh wow great ponderings !!
Just the other day I was thinking something very similar.
here
It has been hard to find an image of what I saw in meditation.
The closest thing to it was this: (but the colours are wrong as it is a diagram. I saw my energy spiraling perfectly around me. I was in the centre, the positive +++) and started off like this:
a neutral atom
then I saw other 'stardust/energy' joining up with me, I changed to this:
Carbon-12: 6 protons + 6 neutrons
(pity the links don't come up when quoting but never mind...you will have to click on the link to see the images, oh thats if you want to of course)
I really wonder now if we are nothing more than energy within energy and perhaps as small as an atom? And all the atoms make up one big atom... such as what you have described !? I know, sounds a bit way out there?
starred and flagged!
Thanks for sharing your thoughts!
[edit on 25-4-2008 by Thurisaz]
Thought the same thoughts when i first saw the model of the atom ive heard it referred to as "the paper weight theory". It basically states we are
nothing more then an atom in a paper weight on some ones desk in a larger universe .
However there is a reason for the similarity basic laws of magnetic pull and rotation apply to the universe as well as the atom, hence the similarities.
However there is a reason for the similarity basic laws of magnetic pull and rotation apply to the universe as well as the atom, hence the similarities.
Part of the reason that this misconception occurs is that in elementary school science classes the atom is represented with spheres for electrons,
protons and neutrons.
However, in real life, the atom is a pure waveform there are in fact no balls or spheres to see under an electron microscope. Here is what an actual hydrogen molecule looks like (after it is colorized based on charge):
Next......
[edit on 4/25/2008 by Choronzon]
However, in real life, the atom is a pure waveform there are in fact no balls or spheres to see under an electron microscope. Here is what an actual hydrogen molecule looks like (after it is colorized based on charge):
Next......
[edit on 4/25/2008 by Choronzon]
Here are some references regarding the atom and its form.
As has been noted the Bohr model for the atom was discarded long ago.
Electrons don't orbit the nuclei as planets orbit the sun.
en.wikipedia.org...
science.howstuffworks.com...
web.jjay.cuny.edu...
Google Search: Structure of the Atom
[edit on 2008/4/25 by GradyPhilpott]
As has been noted the Bohr model for the atom was discarded long ago.
Electrons don't orbit the nuclei as planets orbit the sun.
Electron cloud is a term used, if not originally coined, by the Nobel Prize laureate and acclaimed educator Richard Feynman in The Feynman Lectures on Physics for discussing "exactly what is an electron?". This intuitive model provides a simplified way of visualizing an electron as a solution of the Schrödinger equation. In the electron cloud analogy, the probability density of an electron, or wavefunction, is described as a small cloud moving around the atomic or molecular nucleus, with the opacity of the cloud proportional to the probability density.
The electron cloud formulation better describes many observed phenomena, including the double slit experiment, the periodic table and chemical bonding, and atomic interactions with light. Although lacking in certain details, the intuitive model roughly predicts the experimentally observed wave-particle duality....
In the electron cloud model, rather than following fixed orbits, electrons bound to an atom are observed more frequently in certain areas around the nucleus called orbitals. The electron cloud can transition between electron orbital states, and each state has a characteristic shape and energy, all predicted by the Schrödinger equation, which has infinitely many solutions. Experimental results motivated this conceptual refinement of the Bohr model.
en.wikipedia.org...
en.wikipedia.org...
science.howstuffworks.com...
web.jjay.cuny.edu...
Google Search: Structure of the Atom
[edit on 2008/4/25 by GradyPhilpott]
reply to post by Quest
Definately. They now use the word "orbitals" to describe electron clouds. Which, despite the name are not orbits.
www.orbitals.com...
And the last few posts above mine describe it just as well
[edit on 25-4-2008 by TheBandit795]
Definately. They now use the word "orbitals" to describe electron clouds. Which, despite the name are not orbits.
www.orbitals.com...
And the last few posts above mine describe it just as well
[edit on 25-4-2008 by TheBandit795]
I have often sat and pondered the question posed by the OP. Are we(the universe) just a particle part of an even larger universe, and do the particles
in our universe represent other universes, ad infinitum? An interesting philosophical question for the time being, and one that I don't see modern
science answering any time soon.
reply to post by Choronzon
Awesome, thanks for that. I pondered with the same theory as well but the typical animated atom has made it easy for us to make that misconception. If the solar system was more like an atom wouldn't earth have an indefinite orbit? Anyway there are more threads like this but the titles are never easy to find.
[edit on 25-4-2008 by beaverg]
Awesome, thanks for that. I pondered with the same theory as well but the typical animated atom has made it easy for us to make that misconception. If the solar system was more like an atom wouldn't earth have an indefinite orbit? Anyway there are more threads like this but the titles are never easy to find.
[edit on 25-4-2008 by beaverg]
I wonder how the proportions would scale up...
Say take the distance between the nucleus and the electrons (I seem to rember something along the lines of if the nucleus was football size and placed in the centre of wemberly stadium - the closest electron would be pea sized and wizzing round the M25... no idea if that's right)
Scale up the nucleus to the size of our sun and see where the planets/electrons should be.
I just can't be bothered to find the numbers right now - consider it home work fellow ATSers
Say take the distance between the nucleus and the electrons (I seem to rember something along the lines of if the nucleus was football size and placed in the centre of wemberly stadium - the closest electron would be pea sized and wizzing round the M25... no idea if that's right)
Scale up the nucleus to the size of our sun and see where the planets/electrons should be.
I just can't be bothered to find the numbers right now - consider it home work fellow ATSers
We are now discovering quantum theories that are much smaller than atoms and can change when needed to. As we do as a human race. We adjust to the
changes or environments around us. Are we part of that smaller than an atom quantum theory equation? Look at the double slit experiment and imagine
us as the light. With as many races that we have on our beautiful planet and the interference from all of us clashing together and trying to become
the top dog we are more separated than ever. W#watch this and see.
And then you have to throw in everything else into the mix that involves the human race not just light. Just a thought about how the smallest and biggest play a role in this universe. Are we part of the electrons that are a part of the matter that makes us up. If so is this why we have free will or can change our minds and do different things when we want to. And if we think someone is watching us do we go back to being normal?
[edit on 4/25/2008 by Solarskye]
[edit on 4/25/2008 by Solarskye]
And then you have to throw in everything else into the mix that involves the human race not just light. Just a thought about how the smallest and biggest play a role in this universe. Are we part of the electrons that are a part of the matter that makes us up. If so is this why we have free will or can change our minds and do different things when we want to. And if we think someone is watching us do we go back to being normal?
[edit on 4/25/2008 by Solarskye]
[edit on 4/25/2008 by Solarskye]
Perhaps, this particular universe is smaller than an 'Atom' but never too large to imagine.
Originally posted by DaNReD
if anyone agrees or knows some way how this is noway possible please reply. because of limited knowlege about atoms, and we cant see close enough to see just something else, means that it could be possible.
You're not really so far off base, it's just that the truth is a bit different than your teachers may themselves have known. The structure of the various atoms in the periotic chart are far more complicated and interesting than the simple analogy to a solar system.
Perhaps the shape of a galaxy or the entire universe itself might be closer to what is being discovered:
There are different models of the structure of the atom. One of the first models was created by Niels Bohr, a Danish physicist. He proposed a model in which electrons circle the nucleus in "orbits" around the nucleus, much in the same way as planets orbit the sun. Each orbit represents an energy level which can be determined using equations generated by Planck and others discussed in more detail below. The Bohr model was later proven to be incorrect.
The "accepted" model is the quantum model. In the quantum model, we state that the electron cannot be found precisely, but we can predict the probability, or likelihood, of an electron being at some location in the atom.
-snip-
Chemists care about where electrons are in an atom or a molecule. In the early models, we believed that electrons move like billiard balls, and followed the rules of classical physics. The graphic below attempts to show that earlier models thought that we could identify the exact path, position, velocity, etc. of an electron or electrons in an atom:
A more accurate picture is that the electron(s) reside in a "cloud" that surrounds the nucleus of the atom. This concept is shown in the graphic below:
www.shodor.org...
I've always been taught that atoms are the building blocks of life so why not start at the smallest ( Quantum) to the atom to insects to plants to
smaller animals to us humans to our trees, atmosphere and beyond. If we are indeed connected then we all come from the same energy. Our world, our
solar system, our galaxy, our universe and other things we have no way of seeing big or small have a connection and live together.
[edit on 4/25/2008 by Solarskye]
[edit on 4/25/2008 by Solarskye]
new topics
-
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 9 minutes ago -
Ireland VS Globalists
Social Issues and Civil Unrest: 40 minutes ago -
Biden "Happy To Debate Trump"
Mainstream News: 1 hours ago -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 1 hours ago -
What is the white pill?
Philosophy and Metaphysics: 2 hours ago -
Mike Pinder The Moody Blues R.I.P.
Music: 3 hours ago -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 6 hours ago -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 11 hours ago
top topics
-
President BIDEN's FBI Raided Donald Trump's Florida Home for OBAMA-NORTH KOREA Documents.
Political Conspiracies: 16 hours ago, 35 flags -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 11 hours ago, 20 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 3 hours ago, 7 flags -
What is the white pill?
Philosophy and Metaphysics: 2 hours ago, 5 flags -
Biden "Happy To Debate Trump"
Mainstream News: 1 hours ago, 5 flags -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 1 hours ago, 4 flags -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 6 hours ago, 3 flags -
Ireland VS Globalists
Social Issues and Civil Unrest: 40 minutes ago, 2 flags -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 9 minutes ago, 0 flags
active topics
-
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works • 23 • : RussianTroll -
Biden "Happy To Debate Trump"
Mainstream News • 21 • : PorkChop96 -
A Warning to America: 25 Ways the US is Being Destroyed
New World Order • 28 • : SchrodingersRat -
What is the white pill?
Philosophy and Metaphysics • 17 • : RealityDestroyer -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 280 • : Vermilion -
Ireland VS Globalists
Social Issues and Civil Unrest • 3 • : nugget1 -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order • 0 • : FlyInTheOintment -
Candidate TRUMP Now Has Crazy Judge JUAN MERCHAN After Him - The Stormy Daniels Hush-Money Case.
Political Conspiracies • 793 • : matafuchs -
Is AI Better Than the Hollywood Elite?
Movies • 22 • : 5thHead -
King Charles 111 Diagnosed with Cancer
Mainstream News • 324 • : Oldcarpy2
