It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
originally posted by: DenyObfuscation
a reply to: Arbitrageur
Sort of yes, though to be more accurate, call them a form of energy storage rather than a battery since a battery has certain characteristics the hydrogen storage lacks.
Ok, Ill try to remember that. I just called it a different type of battery to point out that the fuel cell provides electricity for the motor as opposed to the hydrogen being combusted in the engine like gasoline/diesel/propane, etc.
The OP seems to be looking at ways to use hydrogen combustion in an engine. The article he just presented doesn't support his goal.
One thing that can be confusing is the use of the word generator. In the above article the use of the word (Hydrogen) generator, is different than an electrical generator. An electrical generator creates electricity through the revolutions of magnetic polls. A hydrogen generator passes electricity through electrodes, which separates water into gas. Different thing. The forklifts in the above article do not run on electricity any more than an automobile does. Both contain a battery, but is not the primary fuel.
I would be interested in seeing what kind of results GM has seen from these experiments.
edit on 8-9-2015 by TheChrome because: (no reason
given)
a reply to: TheChrome
machinedesign.com...
The forklifts in the above article do not run on electricity any more than an automobile does. Both contain a battery, but is not the primary fuel.
It was relatively easy to change the 5,500-lb forklifts over to fuel cells because they already used electricity. The Hydrogenic HyPm fuel cell supplies 14 kW, and it is smaller and lighter than the lead-acid batteries it replaces.
... Like battery-powered lifts, hydrogen-fueled versions are quiet and emission free, a big advantage for tooling around indoors. But batteries are drained enough over 8 hr to lower the output voltage and make the lift sluggish. Fuel cells, on the other hand, consistently put out the same output voltage.
machinedesign.com...
edit on 8-9-2015 by DenyObfuscation because: add a line
originally posted by: TheChrome
A hydrogen generator passes electricity through electrodes, which separates water into gas.
You are very confused. The hydrogen generator in that article generates hydrogen by electrolysis, and they then store the hydrogen. The forklifts run on electricity, (originally batteries) but the modified one instead of using a rechargeable battery, uses a hydrogen fuel cell (eg.en.wikipedia.org...) using the hydrogen generated in the factory.
The forklifts in the above article do not run on electricity any more than an automobile does.
Wrong it does run on electricity.
edit on 8-9-2015 by hellobruce because: (no reason given)
a reply to: DenyObfuscation
Okay, you are right, I missed the part where they were using batteries in conjunction with the Hydrogen. Must have been late after a long day when I read the article Ha Ha. I also noticed they were processing the gas into storage tanks, then fueling the forklifts. They seemed to be running a dual battery powered/hydrogen vehicle. This is a little different than the application I was referring to, which would be a water tank and HHO generator on the vehicle itself. Using the standard 12V battery to create the electrolysis and the alternator recharging the battery. I don't have a lot of spare time to dive into this right now, but I think it has potential and I am certainly tossing around ideas on paper and in my head.
Okay, you are right, I missed the part where they were using batteries in conjunction with the Hydrogen. Must have been late after a long day when I read the article Ha Ha. I also noticed they were processing the gas into storage tanks, then fueling the forklifts. They seemed to be running a dual battery powered/hydrogen vehicle. This is a little different than the application I was referring to, which would be a water tank and HHO generator on the vehicle itself. Using the standard 12V battery to create the electrolysis and the alternator recharging the battery. I don't have a lot of spare time to dive into this right now, but I think it has potential and I am certainly tossing around ideas on paper and in my head.
originally posted by: DenyObfuscation
a reply to: TheChrome
I missed the part where they were using batteries in conjunction with the Hydrogen.
That's because, they're not. The hydrogen fuel cell produces electricity.
Watch some youtube videos on the subject. The output is gas, not electricity.
I am learning more and more about the research since I started this thread, which is good. I think more heads need to meet on this subject due to it's potential.
originally posted by: TheChrome
Watch some youtube videos on the subject. The output is gas, not electricity.
WTF are you babbling about now? The forklifts run on electricity. Instead of a battery, they are using a hydrogen fuel cell that produces electricity using hydrogen as a fuel.
application I was referring to, which would be a water tank and HHO generator on the vehicle itself. Using the standard 12V battery to create the electrolysis and the alternator recharging the battery.
That would very quickly stop working,
as you lose efficiency in producing hydrogen,
then lose more efficiency running the engine of the gas,
then lose even more efficiency in the generator,
then lose even more efficiency in charging the battery!
edit on 8-9-2015 by hellobruce because: (no reason given)
originally posted by: hellobruce
originally posted by: TheChrome
Watch some youtube videos on the subject. The output is gas, not electricity.
WTF are you babbling about now? The forklifts run on electricity. Instead of a battery, they are using a hydrogen fuel cell that produces electricity using hydrogen as a fuel.
application I was referring to, which would be a water tank and HHO generator on the vehicle itself. Using the standard 12V battery to create the electrolysis and the alternator recharging the battery.
That would very quickly stop working,
as you lose efficiency in producing hydrogen,
then lose more efficiency running the engine of the gas,
then lose even more efficiency in the generator,
then lose even more efficiency in charging the battery!
The energy from the battery passes through the generator, which converts water into gas. The gas combusts fueling the engine and alternator. Because for every action there is an equal and opposite reaction, you must find the energy gain that compensates the energy draw. Energy can neither be lost nor destroyed. That is the law of energy conservation. However, as I have said before there are other laws that come in to play such as Thermodynamics. That is why it is an interesting thing to look into. An example of a crude generator is below:
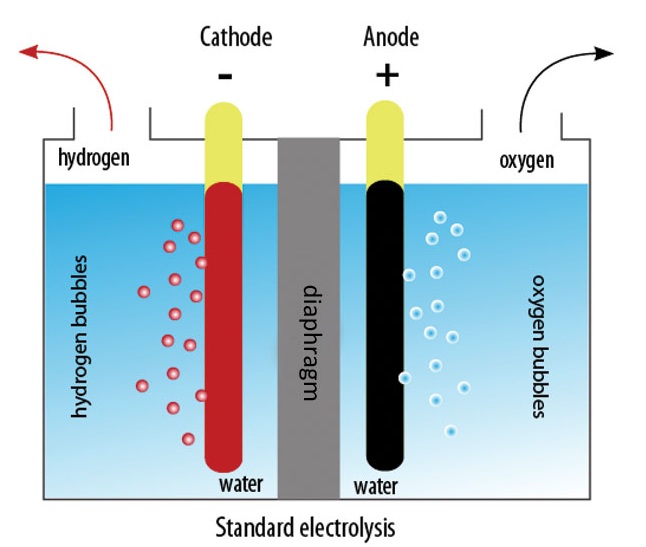
a reply to: TheChrome
Just considering the electrical involvement in the overall cycle, the losses can be easily estimated.
If there's current flowing and any electrical resistance anywhere in the circuit, the lost energy as heat is I^2.R and that doesn't include the other losses imposed by cell construction, motor/alternator windage & hysteresis etc. If it was constructed exclusively with superconductors you'd get better efficiency but still not 100% and definitely never greater than 100%.
Just considering the electrical involvement in the overall cycle, the losses can be easily estimated.
If there's current flowing and any electrical resistance anywhere in the circuit, the lost energy as heat is I^2.R and that doesn't include the other losses imposed by cell construction, motor/alternator windage & hysteresis etc. If it was constructed exclusively with superconductors you'd get better efficiency but still not 100% and definitely never greater than 100%.
originally posted by: TheChrome
The energy from the battery passes through the generator, which converts water into gas. The gas combusts fueling the engine and alternator.
It still would not work for very long!
Because for every action there is an equal and opposite reaction,
WTF do the laws of motion have to do with this???
you must find the energy gain that compensates the energy draw.
How?
Energy can neither be lost nor destroyed.
But in your example it is converted to noise, heat, is used to overcome friction, hysteresis loss in the generator, heat losses in the wires etc. etc.
originally posted by: Pilgrum
a reply to: TheChrome
Just considering the electrical involvement in the overall cycle, the losses can be easily estimated.
If there's current flowing and any electrical resistance anywhere in the circuit, the lost energy as heat is I^2.R and that doesn't include the other losses imposed by cell construction, motor/alternator windage & hysteresis etc. If it was constructed exclusively with superconductors you'd get better efficiency but still not 100% and definitely never greater than 100%.
I agree with you. I think any technology that achieves 100% would make whoever invented it rich, since nothing near that has ever been achieved. I know the basic principals of electricity from college, but I am not an expert in that area. With what I do, you calculate a lot of friction loss. As minute as that may seem, the friction of water moving through the hose or pipe to the generator creates loss. The friction of the gas moving out of the generator and to the engine creates loss. So there is a lot to be calculated, but if you can figure out a way to produce a relatively high efficiency rate, then you're onto something. I started this thread because the technology, apparently has enough validity to be investigated. Can you clarify what you mean by Hysteresis in this case? What I think of Hysteresis is ex. when a pressure gauge is driven beyond it's limits, and won't return to it's original value.
originally posted by: TheChrome
but if you can figure out a way to produce a relatively high efficiency rate, then you're onto something.
Not really, you cannot ignore those losses, your system will always stop working after a short time. Also look at how much power you will have to generate to produce hundreds of liters of Hydrogen.
Can you clarify what you mean by Hysteresis in this case?
It is just another loss in the generator.
I don't see anything wrong with the Wikipedia article on hysteresis, refer to that. What you describe sounds more like a damaged guage.
originally posted by: TheChrome
Can you clarify what you mean by Hysteresis in this case? What I think of Hysteresis is ex. when a pressure gauge is driven beyond it's limits, and won't return to it's original value.
It sounds to me like you're trying to do the same thing as those guys in the video I posted earlier here, are you?
originally posted by: Arbitrageur
I don't see anything wrong with the Wikipedia article on hysteresis, refer to that. What you describe sounds more like a damaged guage.
originally posted by: TheChrome
Can you clarify what you mean by Hysteresis in this case? What I think of Hysteresis is ex. when a pressure gauge is driven beyond it's limits, and won't return to it's original value.
It sounds to me like you're trying to do the same thing as those guys in the video I posted earlier here, are you?
I respectfully want to ask if you studied engineering in college? This is not a wiki war, wiki links are basic.
a reply to: TheChrome
The hysteresis I mentioned is an unavoidable energy loss in a magnetic circuit with polarity reversals going on such as in motors, generators, transformers etc. It is also used in reference to heating/cooling systems or called the 'dead band' of a thermostat.
What it represents is the energy required to establish a magnetic field of a given polarity, then reverse that field to the opposite polarity and some ferro-magnetic materials are better at it than others, but none are loss-free. The characteristic of hysteresis is like a double 'S' with S's joined at the tips and the gap between them representing the effort required to reverse the field. In terms of transformers it's bundled up with the 'iron losses'. A transformer is probably as close as you can get to the 'perfect machine' as it has no moving parts (therefore no windage loss) so the losses are the sum of magnetic fringing, copper loss (I^2.R) and iron loss to produce an efficiency near 95% in a good design.
All off the top of my head so this is just my understanding of it.
The hysteresis I mentioned is an unavoidable energy loss in a magnetic circuit with polarity reversals going on such as in motors, generators, transformers etc. It is also used in reference to heating/cooling systems or called the 'dead band' of a thermostat.
What it represents is the energy required to establish a magnetic field of a given polarity, then reverse that field to the opposite polarity and some ferro-magnetic materials are better at it than others, but none are loss-free. The characteristic of hysteresis is like a double 'S' with S's joined at the tips and the gap between them representing the effort required to reverse the field. In terms of transformers it's bundled up with the 'iron losses'. A transformer is probably as close as you can get to the 'perfect machine' as it has no moving parts (therefore no windage loss) so the losses are the sum of magnetic fringing, copper loss (I^2.R) and iron loss to produce an efficiency near 95% in a good design.
All off the top of my head so this is just my understanding of it.
edit on 9/9/2015 by Pilgrum because: (no reason given)
a reply to: Pilgrum
The whole point of the thread, is that dry cells are more efficient than wet cells. Most dry cell designs use stainless steel from what I have seen. What kind of efficiency would be achieved using super conductive metals or alloys? I don't know the answer to this, as I am just staring to gather information. It would seem that a Tantalum or Niobium alloy might present a greater efficiency as an anode/cathode.
The whole point of the thread, is that dry cells are more efficient than wet cells. Most dry cell designs use stainless steel from what I have seen. What kind of efficiency would be achieved using super conductive metals or alloys? I don't know the answer to this, as I am just staring to gather information. It would seem that a Tantalum or Niobium alloy might present a greater efficiency as an anode/cathode.
originally posted by: TheChrome
a reply to: Pilgrum
The whole point of the thread, is that dry cells are more efficient than wet cells. Most dry cell designs use stainless steel from what I have seen. What kind of efficiency would be achieved using super conductive metals or alloys?
Even using Unobtanium you have to remember....
And for those that don't understand electrolysis, it can never give you more energy out in the form of hydrogen and oxygen to be combusted than you put in in the form of electricity. Ever.
originally posted by: Bedlam
I've never seen any that wasn't blatant measurement error or a scam. If "resonance" or "quantum" is part of it, you should re-read it and mentally substitute "bull#" for those words, see if it improves the text some.
My company is testing Hydrogen fuel cells... what do you think about them? I personally think the next big jump in technology will be energy storage, this will propel us past gas cars, electrical grids etc....
new topics
-
Where should Trump hold his next rally
Politicians & People: 14 minutes ago -
Shocking Number of Voters are Open to Committing Election Fraud
US Political Madness: 58 minutes ago -
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections: 1 hours ago -
Falkville Robot-Man
Aliens and UFOs: 1 hours ago -
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 2 hours ago -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 3 hours ago -
Ireland VS Globalists
Social Issues and Civil Unrest: 3 hours ago -
Biden "Happy To Debate Trump"
2024 Elections: 4 hours ago -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 4 hours ago -
What is the white pill?
Philosophy and Metaphysics: 6 hours ago
top topics
-
A Warning to America: 25 Ways the US is Being Destroyed
New World Order: 14 hours ago, 21 flags -
Blast from the past: ATS Review Podcast, 2006: With All Three Amigos
Member PODcasts: 6 hours ago, 11 flags -
Mike Pinder The Moody Blues R.I.P.
Music: 6 hours ago, 8 flags -
Biden "Happy To Debate Trump"
2024 Elections: 4 hours ago, 8 flags -
Australian PM says the quiet part out loud - "free speech is a threat to democratic dicourse"...?!
New World Order: 3 hours ago, 6 flags -
James O’Keefe: I have evidence that exposes the CIA, and it’s on camera.
Whistle Blowers and Leaked Documents: 2 hours ago, 5 flags -
What is the white pill?
Philosophy and Metaphysics: 6 hours ago, 5 flags -
Ireland VS Globalists
Social Issues and Civil Unrest: 3 hours ago, 4 flags -
RAAF airbase in Roswell, New Mexico is on fire
Aliens and UFOs: 4 hours ago, 4 flags -
Putin, Russia and the Great Architects of the Universe
ATS Skunk Works: 9 hours ago, 3 flags
active topics
-
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 300 • : burritocat -
Where should Trump hold his next rally
Politicians & People • 2 • : charlest2 -
Candidate TRUMP Now Has Crazy Judge JUAN MERCHAN After Him - The Stormy Daniels Hush-Money Case.
Political Conspiracies • 807 • : xuenchen -
Biden "Happy To Debate Trump"
2024 Elections • 36 • : DontTreadOnMe -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 701 • : Thoughtful3 -
This is our Story
General Entertainment • 2 • : ByeByeAmericanPie -
Mike Pinder The Moody Blues R.I.P.
Music • 2 • : DontTreadOnMe -
The functionality of boldening and italics is clunky and no post char limit warning?
ATS Freshman's Forum • 14 • : Encia22 -
MULTIPLE SKYMASTER MESSAGES GOING OUT
World War Three • 99 • : cherokeetroy -
Gov Kristi Noem Shot and Killed "Less Than Worthless Dog" and a 'Smelly Goat
2024 Elections • 25 • : FlyersFan
