It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
a reply to: 1947boomer
I think they made the right call by not recommending as well.
Would be nice to have seen what their recommendation would have been on the vaccine originally, but the FDA did not ask them then.
I think they made the right call by not recommending as well.
Would be nice to have seen what their recommendation would have been on the vaccine originally, but the FDA did not ask them then.
originally posted by: ColoradoTemplar
a reply to: NHow1982
Yep. He will probably threaten to take us out behind the gym and teach us a lesson.
you better watch out, With those F22's and his six inch pythons he's a force to be reckoned with
a reply to: ColoradoTemplar
I do not keep my hopes that high, see when the corrupted FDA pharma lapdogs are doing this is because obviously the side effects of the jabs in children are worst than it has been reported and they have enough data to see it.
Is despicable.
They are maiming and killing children with the jabs.
Is ok to adults but not children.
I do not keep my hopes that high, see when the corrupted FDA pharma lapdogs are doing this is because obviously the side effects of the jabs in children are worst than it has been reported and they have enough data to see it.
Is despicable.
They are maiming and killing children with the jabs.
Is ok to adults but not children.
originally posted by: americanbuffalo1
a reply to: ColoradoTemplar
Well he did say he was losing his patience. Maybe he will suffer an aneurysm due to this wrench in his works.
A third one would likely be fatal...
a reply to: ColoradoTemplar
I really hope Canada and others follow suit and dont include booster shots. Something is odd with their odd.
I really hope Canada and others follow suit and dont include booster shots. Something is odd with their odd.
originally posted by: ColoradoTemplar
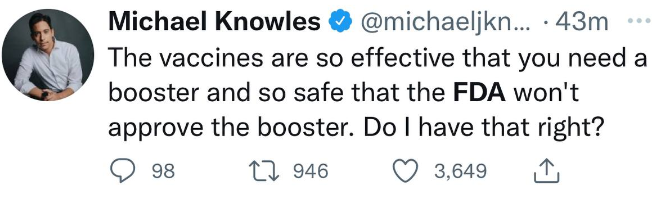
So maybe finally there are a few people in the FDA that are seeing the data and having 2nd doubts about the vaccine all together:
thehill.com...
A federal advisory panel said Friday the Food and Drug Administration (FDA) should not approve a third booster dose of Pfizer and BioNTech's COVID-19 vaccine for everyone 16 or older, but indicated they would be open to boosters for older people.
The panel voted overwhelmingly against the recommendation, 16 to 2.
Friday's vote from the FDA's vaccine advisory committee deals a major blow to the Biden administration's plan to administer booster doses beginning Sept. 20.
Lets see what Biden has to say now.
Approved unannimously for 65+ and those at high risk of contracting severe Covid-19
www.statnews.com...
edit on
17-9-2021 by NHow1982 because: (no reason given)
originally posted by: MiddleInsite
It's interesting that the FDA all of a sudden has credibility, when clearly to many here, it did not.
I always find it funny that with most here, that when you agree with the ruling/finding/decision, well then we can trust it. Otherwise, they are a bunch of government hacks.
I don't see how you can not trust them, and in a split second, you can.
Do you see how ridiculous that is?
So you are saying you trust the FDA and the info they distribute to the public?
a reply to: ColoradoTemplar
Who said we trust the FDA ?
Nobody . The point of this post is to show the circus crumbling. Using the FDA's own flip flops.
No we aren't going to trust the FDA no matter what they say . They've lost that credibility long time ago .
This is just a highlight reel of the circus that is covid
a reply to: ColoradoTemplar
Vaccines reduce to less than 75% effectiveness after 6 months, and the FDA has determined that a Booster shot is NOT needed.
Sounds like we've been in a Pandemic of money payouts and election fraud over the past 19 months.
A cool $5 Trillion worth of payouts!
Barely-lethal Covid-19 was the excuse.
Democrats feel the Suicides, Alcoholism, Family violence, intentional Senior Deaths, were just acceptable losses.
Vaccines reduce to less than 75% effectiveness after 6 months, and the FDA has determined that a Booster shot is NOT needed.
Sounds like we've been in a Pandemic of money payouts and election fraud over the past 19 months.
A cool $5 Trillion worth of payouts!
Barely-lethal Covid-19 was the excuse.
Democrats feel the Suicides, Alcoholism, Family violence, intentional Senior Deaths, were just acceptable losses.
edit on 9/17/2021 by carewemust because: (no reason given)
originally posted by: ColoradoTemplar
Yep. He will probably threaten to take us out behind the gym and teach us a lesson.
More concerned about the White House applying some "reimagine and rebuild" to the FDA so they'll give the correct answers.
Or Biden taking it upon himself, and finding powers to mandate boosters.
This isn't over yet.
NO one has been able to meet my COVID challenge yet...
Find me a single story of....."Joe/Jane in the prime of their perfectly healthy 25 year old or so lives..were taken by COVID way to early.." certainly with "600k+" deaths the media should have at least one of these stories (verifiable) to advance their narrative....last time someone sent me such a story, the person in question did not actually exist...
The vaccine probably kills more young healthy people than the actual virus....remember all vaccines have side effects, the average is about 2.5%...ranging from minor rash...to death....a person in my office was paralyzed from the wast down from a flu shot...
Find me a single story of....."Joe/Jane in the prime of their perfectly healthy 25 year old or so lives..were taken by COVID way to early.." certainly with "600k+" deaths the media should have at least one of these stories (verifiable) to advance their narrative....last time someone sent me such a story, the person in question did not actually exist...
The vaccine probably kills more young healthy people than the actual virus....remember all vaccines have side effects, the average is about 2.5%...ranging from minor rash...to death....a person in my office was paralyzed from the wast down from a flu shot...
They are lying, as usual nobody can trust this people, they are going to create another wave as the first two jabs are not working anymore,
then blame on the unvaxed again, the boosters will save the day, because the vaxed will get sick with covid.
I will seat and watch the pony show, I guess the WHO knew something but big pharma greed kept pushing for it.
What is happening in Israel from the boosters they already gave to their people, that is not been talk about.
Something is going on and is not pretty.
I will seat and watch the pony show, I guess the WHO knew something but big pharma greed kept pushing for it.
What is happening in Israel from the boosters they already gave to their people, that is not been talk about.
Something is going on and is not pretty.
The number of daily deaths in America attributed to Covid-19 keeps increasing: www.worldometers.info...
But the FDA rules Booster shots are not needed.
Is this an admission the Vaccines don't work?
But the FDA rules Booster shots are not needed.
Is this an admission the Vaccines don't work?
a reply to: carewemust
The latest Israel data point outs that the boosters will give an immune response for the elderly and health compromised, but the boost seems to fall short and last for a short period of time, probably a few months, until another booster is needed.
It seems that the body is no really working well with the technology been tested in the jabs.
the body is learning to by pass the boost or immunity after a while.
The latest Israel data point outs that the boosters will give an immune response for the elderly and health compromised, but the boost seems to fall short and last for a short period of time, probably a few months, until another booster is needed.
It seems that the body is no really working well with the technology been tested in the jabs.
the body is learning to by pass the boost or immunity after a while.
a reply to: Vasa Croe
Actually, they did.
For the original EUAs:
Pfizer
Moderna
And for a third dose for some.
3rd Dose
But, if you aren't vaccinated it's not really a concern of yours anyway. Is it?
Would be nice to have seen what their recommendation would have been on the vaccine originally, but the FDA did not ask them then.
Actually, they did.
For the original EUAs:
Pfizer
Moderna
And for a third dose for some.
3rd Dose
This authorization of a third dose for immunocompromised individuals is separate and distinct from the booster dose reviewed by VRBPAC today. The third dose for immunocompromised individuals is meant to address the fact that these individuals sometimes do not build enough protection after two doses of the vaccine. In contrast, the booster dose recommended today by VRBPAC for EUA refers to an additional dose of the vaccine that is given to those who have built enough protection after the primary two-dose vaccination series, but may have decreased protection over time due to waning of immunity.
But, if you aren't vaccinated it's not really a concern of yours anyway. Is it?
edit on 9/17/2021 by Phage because: (no reason given)
new topics
-
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections: 1 hours ago -
Ode to Artemis
General Chit Chat: 1 hours ago -
Ditching physical money
History: 5 hours ago -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 5 hours ago -
Don't take advantage of people just because it seems easy it will backfire
Rant: 5 hours ago -
VirginOfGrand says hello
Introductions: 6 hours ago -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 7 hours ago -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 9 hours ago -
Geddy Lee in Conversation with Alex Lifeson - My Effin’ Life
People: 10 hours ago -
God lived as a Devil Dog.
Short Stories: 11 hours ago
top topics
-
Hate makes for strange bedfellows
US Political Madness: 15 hours ago, 20 flags -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 9 hours ago, 12 flags -
Police clash with St George’s Day protesters at central London rally
Social Issues and Civil Unrest: 12 hours ago, 9 flags -
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections: 1 hours ago, 7 flags -
TLDR post about ATS and why I love it and hope we all stay together somewhere
General Chit Chat: 13 hours ago, 7 flags -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 7 hours ago, 6 flags -
Don't take advantage of people just because it seems easy it will backfire
Rant: 5 hours ago, 4 flags -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 5 hours ago, 4 flags -
God lived as a Devil Dog.
Short Stories: 11 hours ago, 3 flags -
Ditching physical money
History: 5 hours ago, 3 flags
active topics
-
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections • 2 • : ImagoDei -
Russia Ukraine Update Thread - part 3
World War Three • 5717 • : Arbitrageur -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections • 44 • : budzprime69 -
Hundreds of teenagers flood into downtown Chicago, smashing car windows
Other Current Events • 111 • : 777Vader -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 125 • : Annee -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media • 22 • : ImagoDei -
Who guards the guards
US Political Madness • 4 • : kwaka -
New whistleblower Jason Sands speaks on Twitter Spaces last night.
Aliens and UFOs • 44 • : pianopraze -
"We're All Hamas" Heard at Columbia University Protests
Social Issues and Civil Unrest • 263 • : YourFaceAgain -
Remember These Attacks When President Trump 2.0 Retribution-Justice Commences.
2024 Elections • 50 • : Asher47