It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
reply to post by Moduli
Always a pleasure to see you too.
What? Who the heck is Nima??
Or maybe I didn't published it on a journal. Ever thought of that? How many ATSers, really, publish their posts in Nature?
Not bad. I like the name. So you do have imagination after all!
Many thanks. But actually you have to look for the page "platonic solids". Just so you know; you who knows all of mathematics.
Hm, my computer doesn't come with SuperSimulationProX300 version 11.5.8 slash summer update. I used what I had to make a visual draft. If you have a problem with Photoshop, I am sure Adobe will be most happy to hear about your complaints.
Of course not. You're a self-proclaimed "genius" who can't stop bragging about it. Big difference.
Not even close. It's:
-A plane,
-which is perpendicular to the momentum direction.
And you got lost at that?!
lol of course you have contacts... otherwise the Nobel committee wouldn't have noticed all the groundbreaking discoveries you've made.
Yes, we know you have a degree, you can't stop telling ATS about it.
Always a pleasure to see you too.
Plagiarizing the title of the media's reports on Nima's paper will not get you any credit with real scientists.
What? Who the heck is Nima??
Didn't fill out the right forms to get it published? I bet we can expect to see it any day now when these trivial little misunderstandings are cleared up.
Or maybe I didn't published it on a journal. Ever thought of that? How many ATSers, really, publish their posts in Nature?
I'm going to code name my criticism of you Mongoose B. In particular, the hierarchically recursive (reduced normal form) Mongoose B Criticism.
Not bad. I like the name. So you do have imagination after all!
A quick look at the wikipedia page for the number 6 is all that's needed to solve this conundrum! Masterful work.
Many thanks. But actually you have to look for the page "platonic solids". Just so you know; you who knows all of mathematics.
Which I have absolutely no doubt, that was created as the result of state-of-the-art particle physics simulation software available to actual researchers, and was not at all in any way produced in photoshop.
Hm, my computer doesn't come with SuperSimulationProX300 version 11.5.8 slash summer update. I used what I had to make a visual draft. If you have a problem with Photoshop, I am sure Adobe will be most happy to hear about your complaints.
Do you take me for some kind of crackpot who doesn't understand physics?
Of course not. You're a self-proclaimed "genius" who can't stop bragging about it. Big difference.
In the... am I getting it right... anti-time direction? I'll admit you've lost me here.
Not even close. It's:
-A plane,
-which is perpendicular to the momentum direction.
And you got lost at that?!
but some of my contacts on the Nobel committee
lol of course you have contacts... otherwise the Nobel committee wouldn't have noticed all the groundbreaking discoveries you've made.
mostly on account of my crippling student loan debt incurred by my actually degree in physics
Yes, we know you have a degree, you can't stop telling ATS about it.
edit on 24-11-2013 by swanne because: (no reason given)
reply to post by Thought Provoker
Interesting stuff, In water, when you take away energy it actually produces work. I had a thread about this here . Does your theory explain this?
Interesting stuff, In water, when you take away energy it actually produces work. I had a thread about this here . Does your theory explain this?
reply to post by jlafleur02
My theory? No; subatomic vibration has nothing directly to do with freezing water's expansion. I believe your answer involves density. The work being performed by the energy being removed is to force the H2O molecules into a more-rigid crystalline structure (ice), which is physically larger than the fluid, uncrystallized state. The energy being removed sort of "sucks" the molecules apart, per se. The volume expands with no change in mass as the brownian motion goes away, which lowers the density and exerts mechanical force in all directions that, if restrained, tries to overcome its container. Liquid water expands both when frozen or boiled, but far more so when boiled. Or plas- plas, ummm, plasmated? Plasmized? (There ought to be a word.) Water can only expand a small amount when frozen, but it is inexorable... like, ummm... it's not easy coming up with an analogy; it's quite a unique feature of water that it does this, not much else does. Water gets bigger when it freezes because of the shape of its molecules, and there is an upper limit to size, unlike when it gets hotter. No matter how much colder ice gets, it doesn't expand further; that's a one-off when crossing the freezing point, and it's due to the molecules pushing each other into a crystalline configuration and locking together like that, providing rigidity. Analogy, analogy, simile, metaphor, what, what...
Jenga!! Yes, that's it. As you pull little blocks out of the bottom and stack them on top, the tower's mass stays the same but the density drops as it takes up more space. It's... sort of the same as water freezing, as close as I can think of right now. Nothing to do with particle vibrations, though.
Or, or, the energy which exerts the pressure is electromagnetic, the energy in each electron, their like charges trying to repel each other. Ultimately, all energy starts with vacuum energy, the energy that creates each particle and gives it mass, charge, and spin. Temperature doesn't affect those three fundamental properties. Perhaps your answer lies along those lines.
My theory? No; subatomic vibration has nothing directly to do with freezing water's expansion. I believe your answer involves density. The work being performed by the energy being removed is to force the H2O molecules into a more-rigid crystalline structure (ice), which is physically larger than the fluid, uncrystallized state. The energy being removed sort of "sucks" the molecules apart, per se. The volume expands with no change in mass as the brownian motion goes away, which lowers the density and exerts mechanical force in all directions that, if restrained, tries to overcome its container. Liquid water expands both when frozen or boiled, but far more so when boiled. Or plas- plas, ummm, plasmated? Plasmized? (There ought to be a word.) Water can only expand a small amount when frozen, but it is inexorable... like, ummm... it's not easy coming up with an analogy; it's quite a unique feature of water that it does this, not much else does. Water gets bigger when it freezes because of the shape of its molecules, and there is an upper limit to size, unlike when it gets hotter. No matter how much colder ice gets, it doesn't expand further; that's a one-off when crossing the freezing point, and it's due to the molecules pushing each other into a crystalline configuration and locking together like that, providing rigidity. Analogy, analogy, simile, metaphor, what, what...
Jenga!! Yes, that's it. As you pull little blocks out of the bottom and stack them on top, the tower's mass stays the same but the density drops as it takes up more space. It's... sort of the same as water freezing, as close as I can think of right now. Nothing to do with particle vibrations, though.
Or, or, the energy which exerts the pressure is electromagnetic, the energy in each electron, their like charges trying to repel each other. Ultimately, all energy starts with vacuum energy, the energy that creates each particle and gives it mass, charge, and spin. Temperature doesn't affect those three fundamental properties. Perhaps your answer lies along those lines.
edit on 11/24/2013 by Thought Provoker because: Or,
or...
reply to post by Moduli
Is it me, or you seem to be jealous of Swanne? Since the beginning of Swanne's Phoenix Theory, you have non-stop attacked him and mocking him instead of actually contributing in a meaningful way.
No, you seem to be very jealous; here you are, having a supposedly degree in physics, and yet, a simple civillian from Canada created a simple theory and graphics that finally solves the problem for preons, while you didn't contributed one bit to science's history. That's why you're spending so much time trying to discredit him. What's your next move, discredit Swanne so you can publish his theory in Nature, and get all the credit (and potentially a Nobel prize)?
Because frankly, you're not at all acting like a scientist. Scientist (at least, not the close-minded ones and stuck in a dogma) don't slash and mock people. Quantum Theory interpretations dictates alot more non-sense and stupid things than Swanne's Pheonix I/II theory, and yet, these Quantum interpretations are widely approved of, even if a third of it is non-sense.
But then, Quantum Theory came from someone with a PhD, not from a civillian. Would it have been from a civillian, scientists like yourselves would have slashed it to the ground.
People like you are why people like me and most other laymen can't understand science and physics. Because modern physics demands pages-long equations for just one aspect. Read a physics book to a layman, and it's all a big mumble-jumble.
But here's Swanne: he actually created a theory that is understandable. No page-long equations, no mumble-jumble. It's 6 preons, whose spins and flips creates charges and mass inside quarks and electrons. How complicated is that? No need for a PhD, which, in case you're not aware of, most of the population can't afford.
Is it me, or you seem to be jealous of Swanne? Since the beginning of Swanne's Phoenix Theory, you have non-stop attacked him and mocking him instead of actually contributing in a meaningful way.
No, you seem to be very jealous; here you are, having a supposedly degree in physics, and yet, a simple civillian from Canada created a simple theory and graphics that finally solves the problem for preons, while you didn't contributed one bit to science's history. That's why you're spending so much time trying to discredit him. What's your next move, discredit Swanne so you can publish his theory in Nature, and get all the credit (and potentially a Nobel prize)?
Because frankly, you're not at all acting like a scientist. Scientist (at least, not the close-minded ones and stuck in a dogma) don't slash and mock people. Quantum Theory interpretations dictates alot more non-sense and stupid things than Swanne's Pheonix I/II theory, and yet, these Quantum interpretations are widely approved of, even if a third of it is non-sense.
But then, Quantum Theory came from someone with a PhD, not from a civillian. Would it have been from a civillian, scientists like yourselves would have slashed it to the ground.
People like you are why people like me and most other laymen can't understand science and physics. Because modern physics demands pages-long equations for just one aspect. Read a physics book to a layman, and it's all a big mumble-jumble.
But here's Swanne: he actually created a theory that is understandable. No page-long equations, no mumble-jumble. It's 6 preons, whose spins and flips creates charges and mass inside quarks and electrons. How complicated is that? No need for a PhD, which, in case you're not aware of, most of the population can't afford.
reply to post by swanne
I don't understand a word you said. Nonetheless, I've marked the thread to come back to and educate myself about things I know almost nothing about. Thanks for your interest in the data and for sharing it with us.
I don't understand a word you said. Nonetheless, I've marked the thread to come back to and educate myself about things I know almost nothing about. Thanks for your interest in the data and for sharing it with us.
edit on 24-11-2013 by Aleister because: word (word)
Americanist
reply to post by swanne
To answer your question... Yes, because they're fundamental to nature.
Good point.
Now notice the placement of both flip and choke rings:
Ugh, I'm on dial-up. Videos take eternity to load, maybe a bit more. Would there be by any chance text or image about the phenomena described in the video?
Hi Swanne,
I'm very excited to see where your theory goes, S&F for this, keep up the outside the box thinking!
I'm very excited to see where your theory goes, S&F for this, keep up the outside the box thinking!
reply to post by parad0x122
reply to post by Aleister
Thanks!
But I fear the scientific community won't give my theory much thoughts. I don't really blame them... I know my theory doesn't have enough maths and quantization.
reply to post by Aleister
Thanks!
But I fear the scientific community won't give my theory much thoughts. I don't really blame them... I know my theory doesn't have enough maths and quantization.
reply to post by swanne
As you say, it's only a theory. Every theory begins with just an idea, now it's just a matter of combining brainpower with the right people who could make more technical headway in the direction you're trying to go with it. Best of luck :-)
As you say, it's only a theory. Every theory begins with just an idea, now it's just a matter of combining brainpower with the right people who could make more technical headway in the direction you're trying to go with it. Best of luck :-)
reply to post by swanne
For an amateur like myself would it be accurate to say, your theory (which is wonderfully illustrated) suggests the mechanism for the conversion of energy to matter (Higgs Boson/God particle) uses these primeons and logosons to bind/alter the spin rates phasing anti-matter into matter and vice versa?
I LOVE the name logoson, because it in effect could render true the verse "By faith, we know the Universe was framed (created/sustained) by the Logos of God." - HEB 11
I can actually see your theory marrying the following theories into one unified theory:
- String Theory (covered by your spin rate/frequency)
- Multiverse (since the understanding of the logoson would cover the need for the missing "matter")
- Higgs Boson Particle (God Particle)
- Relativity/Gravity
- and AMAZINGLY, the faith chapter of Hebrews from scripture!
I must say Swanne, this is great work. Thanks,
God Bless,
For an amateur like myself would it be accurate to say, your theory (which is wonderfully illustrated) suggests the mechanism for the conversion of energy to matter (Higgs Boson/God particle) uses these primeons and logosons to bind/alter the spin rates phasing anti-matter into matter and vice versa?
I LOVE the name logoson, because it in effect could render true the verse "By faith, we know the Universe was framed (created/sustained) by the Logos of God." - HEB 11
I can actually see your theory marrying the following theories into one unified theory:
- String Theory (covered by your spin rate/frequency)
- Multiverse (since the understanding of the logoson would cover the need for the missing "matter")
- Higgs Boson Particle (God Particle)
- Relativity/Gravity
- and AMAZINGLY, the faith chapter of Hebrews from scripture!
I must say Swanne, this is great work. Thanks,
God Bless,
edit on 5-12-2013 by ElohimJD because: (no reason given)
reply to post by parad0x122
reply to post by ElohimJD
You guys are probably right. Thanks for the kind word.
Antimatter/matter are determined by the identity of the preons - 6xA for a positron, 6xB for an electron. As for the spin, it is created by the preon configuration inside the particle, which in turn determines the nature of the particle (matter or energy).
Indeed, many of these theories are in my mind. I am relieved you noticed! Thanks!
reply to post by ElohimJD
You guys are probably right. Thanks for the kind word.
ElohimJD
reply to post by swanne
For an amateur like myself would it be accurate to say, your theory (which is wonderfully illustrated) suggests the mechanism for the conversion of energy to matter (Higgs Boson/God particle) uses these primeons and logosons to bind/alter the spin rates phasing anti-matter into matter and vice versa?
Antimatter/matter are determined by the identity of the preons - 6xA for a positron, 6xB for an electron. As for the spin, it is created by the preon configuration inside the particle, which in turn determines the nature of the particle (matter or energy).
ElohimJDI can actually see your theory marrying the following theories into one unified theory:
- String Theory (covered by your spin rate/frequency)
- Multiverse (since the understanding of the logoson would cover the need for the missing "matter")
- Higgs Boson Particle (God Particle)
- Relativity/Gravity
- and AMAZINGLY, the faith chapter of Hebrews from scripture!
Indeed, many of these theories are in my mind. I am relieved you noticed! Thanks!
edit on 8-12-2013 by swanne because: (no reason given)
Hi swanne, I've got a few suggestions if you don't mind.
Remove any claims about your model, just makes you an easy target as you are missing any supporting calculations. All you are doing currently is simply playing with some numbers, nothing wrong with that.
You have recognized, that there are certain conserved properties. Why not work further on this to have something qualitative (sidestepping the math for now)?
Look at the quarks numbers here: hyperphysics.phy-astr.gsu.edu...
Leptons: hyperphysics.phy-astr.gsu.edu...
Can you fit this numbers into your model? What does it mean for your preons?
And the conservation laws: hyperphysics.phy-astr.gsu.edu...
Do they work with your model?
Remove any claims about your model, just makes you an easy target as you are missing any supporting calculations. All you are doing currently is simply playing with some numbers, nothing wrong with that.
You have recognized, that there are certain conserved properties. Why not work further on this to have something qualitative (sidestepping the math for now)?
Look at the quarks numbers here: hyperphysics.phy-astr.gsu.edu...
Leptons: hyperphysics.phy-astr.gsu.edu...
Can you fit this numbers into your model? What does it mean for your preons?
And the conservation laws: hyperphysics.phy-astr.gsu.edu...
Do they work with your model?
edit on 9-12-2013 by moebius because: fix typo
reply to post by moebius
Good suggestion - I'll take a look and come back to you shortly.
Good suggestion - I'll take a look and come back to you shortly.
edit on 11-12-2013 by swanne because: (no reason given)
reply to post by moebius
All right. In the links you provided, these next parameters are those which checks OK as I compare them with the OP:
Spin number (quarks): accounted for
Charge number (quarks): accounted for
Generation (quarks): accounted for
Spin number (leptons): accounted for
Charge number (leptons): accounted for
Generation (leptons): accounted for
Spin number (bosons): accounted for
Charge number (bosons): accounted for
These are the numbers which are not covered by the OP:
Mass number (quarks): pattern's logic not found
Mass number (leptons): pattern's logic not found
Mass number (bosons): pattern's logic not found
Now, as for conservation laws: I've checked these and found that these laws really concerns baryonic and mesonic decay, while the OP is more about the inner composition of a given elementary particle. That's why I feel the Standard Model is something to keep in mind when it comes to such laws. It's a bit like... hm, a bit like quantum model versus Newtonian model: one is great at predicting atomic events while the other is great at explaining larger bodies.
That being said, my theory strongly supports the Standard Model's statement that "there is a separate lepton number for muons which must also be conserved". In my OP, it is shown that electrons, muons and tauons have different dimensional properties, properties which can hardly be modified by any known forces. Which strongly concurs with the SM's conclusion.
All right. In the links you provided, these next parameters are those which checks OK as I compare them with the OP:
Spin number (quarks): accounted for
Charge number (quarks): accounted for
Generation (quarks): accounted for
Spin number (leptons): accounted for
Charge number (leptons): accounted for
Generation (leptons): accounted for
Spin number (bosons): accounted for
Charge number (bosons): accounted for
These are the numbers which are not covered by the OP:
Mass number (quarks): pattern's logic not found
Mass number (leptons): pattern's logic not found
Mass number (bosons): pattern's logic not found
Now, as for conservation laws: I've checked these and found that these laws really concerns baryonic and mesonic decay, while the OP is more about the inner composition of a given elementary particle. That's why I feel the Standard Model is something to keep in mind when it comes to such laws. It's a bit like... hm, a bit like quantum model versus Newtonian model: one is great at predicting atomic events while the other is great at explaining larger bodies.
That being said, my theory strongly supports the Standard Model's statement that "there is a separate lepton number for muons which must also be conserved". In my OP, it is shown that electrons, muons and tauons have different dimensional properties, properties which can hardly be modified by any known forces. Which strongly concurs with the SM's conclusion.
This looks interesting and may be part of the way to get this elegant idea talking to the mathematics.
www.abovetopsecret.com...
www.wired.com...
arxiv.org...
Download the PDF of the last one and start by scanning through to see the figures. Then get excited and use Feynman's advice to his sister on approaching physics books: read attentively and keep going till you get completely lost. Then start at the beginning again, every time you get further
From looking through very quickly I got this sneaking suspicion that you could use their methods to model your idea succinctly.
www.abovetopsecret.com...
www.wired.com...
arxiv.org...
Download the PDF of the last one and start by scanning through to see the figures. Then get excited and use Feynman's advice to his sister on approaching physics books: read attentively and keep going till you get completely lost. Then start at the beginning again, every time you get further
From looking through very quickly I got this sneaking suspicion that you could use their methods to model your idea succinctly.
reply to post by asciikewl
Good advice. Hm, thanks for the links!
Although the "jewel" in some of those link refers not to a solid form but to a mathematical concept, one of your link rings a bell with its "Amplituhedron". I'll have to check this "Amplituhedron" further, but the name reminds me so much of the octahedron amplitudes which I describe in my OP:
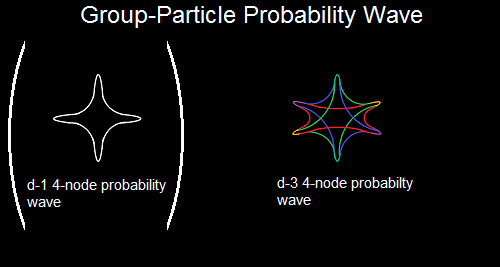
If only some physicist could tell me what an amplituhdron is exactly... it would save me alot of searching around... I'm still just a layman...
Good advice. Hm, thanks for the links!
Although the "jewel" in some of those link refers not to a solid form but to a mathematical concept, one of your link rings a bell with its "Amplituhedron". I'll have to check this "Amplituhedron" further, but the name reminds me so much of the octahedron amplitudes which I describe in my OP:

If only some physicist could tell me what an amplituhdron is exactly... it would save me alot of searching around... I'm still just a layman...
edit on 18-12-2013 by swanne because: (no reason given)
Ha!
For those who think that science is "settled" against the existence of preons:
Source: List of unsolved problems in physics
My answer: Yes they are (composite particles)!
For those who think that science is "settled" against the existence of preons:
Are any of the particles in the standard model of particle physics actually composite particles too tightly bound to observe as such at current experimental energies?
Source: List of unsolved problems in physics
My answer: Yes they are (composite particles)!
Oh my word! I just discovered that my preon model gives an explanation for the presence of colour charges... And why only quarks have 3 colour (and 3
anti-colour) charges! My model also explains why particles such as electron & positron annihilate into photons and NOT neutrinos.
This is becoming quite interesting.
This is becoming quite interesting.
edit on 30-4-2015 by swanne because: (no reason given)
originally posted by: swanne
particles such as electron & positron annihilate into photons and NOT neutrinos.
How can electron and positron collide and them as the particles they were completely disappear, and all that then exists is photons? Would this not mean that electrons and positrons, are absolutely nothing but photons?
new topics
-
Electrical tricks for saving money
Education and Media: 20 minutes ago -
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News: 1 hours ago -
Sunak spinning the sickness figures
Other Current Events: 2 hours ago -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues: 2 hours ago -
Late Night with the Devil - a really good unusual modern horror film.
Movies: 4 hours ago -
Cats Used as Live Bait to Train Ferocious Pitbulls in Illegal NYC Dogfighting
Social Issues and Civil Unrest: 5 hours ago -
The Good News According to Jesus - Episode 1
Religion, Faith, And Theology: 7 hours ago -
HORRIBLE !! Russian Soldier Drinking Own Urine To Survive In Battle
World War Three: 9 hours ago
top topics
-
SETI chief says US has no evidence for alien technology. 'And we never have'
Aliens and UFOs: 16 hours ago, 8 flags -
Cats Used as Live Bait to Train Ferocious Pitbulls in Illegal NYC Dogfighting
Social Issues and Civil Unrest: 5 hours ago, 8 flags -
Florida man's trip overseas ends in shock over $143,000 T-Mobile phone bill
Social Issues and Civil Unrest: 12 hours ago, 8 flags -
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News: 1 hours ago, 6 flags -
Former Labour minister Frank Field dies aged 81
People: 14 hours ago, 4 flags -
Bobiverse
Fantasy & Science Fiction: 12 hours ago, 3 flags -
HORRIBLE !! Russian Soldier Drinking Own Urine To Survive In Battle
World War Three: 9 hours ago, 3 flags -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues: 2 hours ago, 3 flags -
Sunak spinning the sickness figures
Other Current Events: 2 hours ago, 3 flags -
Late Night with the Devil - a really good unusual modern horror film.
Movies: 4 hours ago, 2 flags
active topics
-
Is there a hole at the North Pole?
ATS Skunk Works • 38 • : Therealbeverage -
Cats Used as Live Bait to Train Ferocious Pitbulls in Illegal NYC Dogfighting
Social Issues and Civil Unrest • 11 • : charlyv -
SETI chief says US has no evidence for alien technology. 'And we never have'
Aliens and UFOs • 41 • : TheMisguidedAngel -
Naked Eye Supernova Erupting in the T Coronae Borealis
Space Exploration • 13 • : Therealbeverage -
The Real Reason Behind the Sean Combes Exposure
Political Conspiracies • 45 • : ThatSmellsStrange -
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News • 14 • : lilzazz -
Electrical tricks for saving money
Education and Media • 0 • : annonentity -
Late Night with the Devil - a really good unusual modern horror film.
Movies • 4 • : DBCowboy -
Hate makes for strange bedfellows
US Political Madness • 41 • : Solvedit -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues • 11 • : DBCowboy
