It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
reply to post by poet1b
I'm glad I could make you laugh. I hope it helps calm your nerves somewhat.
The Arctic ice sheet is approximately 12 million km², which is a little larger than Europe, about the size of the United States and Mexico combined, and smaller than Russia - just to put things into perspective. That's about 2.5% of the planet's surface. I would call that localized.
I would love to see your proof that a concentration of 0.00018% methane and 0.04% carbon dioxide will cause a temperature rise sufficient to melt 12 million km² of ice. I mean, if we're going to start asking for numbers, shouldn't you make yours available as well?
(I actually made mine available some time back in another thread.)
If ice is melting, heat energy is being added. Heat energy also tends to thaw other frozen things besides ice.
Undersea, not underground... and it does so by adding heat to everything in contact with the emissions, which will dissipate outward via conduction and convection. Are you saying heat doesn't thaw frozen tundra?
Yes, I am quite familiar with hydrates. What do the hydrates have to do with the source of the heat? Are you saying the methane in the permafrost is melting itself?
TheRedneck
I'm glad I could make you laugh. I hope it helps calm your nerves somewhat.
Do you realize that the Arctic Ocean is a lot larger than that few square inches on a globe?
The Arctic ice sheet is approximately 12 million km², which is a little larger than Europe, about the size of the United States and Mexico combined, and smaller than Russia - just to put things into perspective. That's about 2.5% of the planet's surface. I would call that localized.
Where is your proof that an underwater volcano has ever created a temperature rise of the extent we are now seeing in the Arctic Ocean over an area the size of the Arctic Ocean?
I would love to see your proof that a concentration of 0.00018% methane and 0.04% carbon dioxide will cause a temperature rise sufficient to melt 12 million km² of ice. I mean, if we're going to start asking for numbers, shouldn't you make yours available as well?
(I actually made mine available some time back in another thread.)
How do explain frozen tundra in the Arctic on land defrosting and being exposed to the air for the first time in over a million years?
If ice is melting, heat energy is being added. Heat energy also tends to thaw other frozen things besides ice.
How does you underground volcano un-freeze land as well?
Undersea, not underground... and it does so by adding heat to everything in contact with the emissions, which will dissipate outward via conduction and convection. Are you saying heat doesn't thaw frozen tundra?
Do you understand the source of the Methane? Do you know what methane hydrates are?
Yes, I am quite familiar with hydrates. What do the hydrates have to do with the source of the heat? Are you saying the methane in the permafrost is melting itself?
TheRedneck
reply to post by eriktheawful
When a poster ridicules me, I ridicule back. I know how big the Arctic Ocean is, I have seen a globe, I understand that a 2d representation distorts size at the poles.
I don't need to prove the theory wrong, it is up to the poster to prove the theory.
I posted clear evidence as to why the volcano theory is wrong, and both of you ignore the clear facts I posted.
There are no examples of a volcano heating up an area as large as the Arctic Ocean. The Arctic Ocean might be small compared to the Pacific Ocean, but it is still a very extensive body of water. There are known underwater volcanoes, and they have not change the temperature of smaller bodies of water, like the Mediterranean Sea, the way the Arctic Ocean has heated up, which means the claim has no solid basis for consideration.
If TheRedneck wants an intelligent discussion, he shouldn't ignore all the evidence I have presented and insult my intelligence by claiming I don't know the difference between a 2d map and a globe.
When facts are ignored and insults are used, then the claim becomes a joke.
How do you explain Arctic Tundra melting? This must be some super volcano to change the temperature to the entire top of the planet, even on land.
While you are at telling people how to have an intelligent discussion, maybe you can address all the anti-GW poster who throw their cheap shots up on the thread.
When a poster ridicules me, I ridicule back. I know how big the Arctic Ocean is, I have seen a globe, I understand that a 2d representation distorts size at the poles.
I don't need to prove the theory wrong, it is up to the poster to prove the theory.
I posted clear evidence as to why the volcano theory is wrong, and both of you ignore the clear facts I posted.
There are no examples of a volcano heating up an area as large as the Arctic Ocean. The Arctic Ocean might be small compared to the Pacific Ocean, but it is still a very extensive body of water. There are known underwater volcanoes, and they have not change the temperature of smaller bodies of water, like the Mediterranean Sea, the way the Arctic Ocean has heated up, which means the claim has no solid basis for consideration.
If TheRedneck wants an intelligent discussion, he shouldn't ignore all the evidence I have presented and insult my intelligence by claiming I don't know the difference between a 2d map and a globe.
When facts are ignored and insults are used, then the claim becomes a joke.
How do you explain Arctic Tundra melting? This must be some super volcano to change the temperature to the entire top of the planet, even on land.
While you are at telling people how to have an intelligent discussion, maybe you can address all the anti-GW poster who throw their cheap shots up on the thread.
Anthropogenic CO2 increase[edit]
While CO2 absorption and release is always happening as a result of natural processes, the recent drastic rise in CO2 levels in the atmosphere is known to be mainly due to human activity.[23] Researchers know this both by calculating the amount released based on various national statistics, and by examining the ratio of various carbon isotopes in the atmosphere,[23] as the burning of long-buried fossil fuels releases CO2 containing carbon of different isotopic ratios to those of living plants, enabling them to distinguish between natural and human-caused contributions to CO2 concentration.
Burning fossil fuels such as coal and petroleum is the leading cause of increased anthropogenic CO2; deforestation is the second major cause. In 2010, 9.14 gigatonnes of carbon (33.5 gigatonnes of CO2) were released from fossil fuels and cement production worldwide, compared to 6.15 gigatonnes in 1990.[24] In addition, land use change contributed 0.87 gigatonnes in 2010, compared to 1.45 gigatonnes in 1990.[24] In 1997, human-caused Indonesian peat fires were estimated to have released between 13% and 40% of the average carbon emissions caused by the burning of fossil fuels around the world in a single year.[25][26][27] In the period 1751 to 1900 about 12 gigatonnes of carbon were released as carbon dioxide to the atmosphere from burning of fossil fuels, whereas from 1901 to 2008 the figure was about 334 gigatonnes.[28]
This addition, about 3% of annual natural emissions, as of 1997, is sufficient to exceed the balancing effect of sinks.[29] As a result, carbon dioxide has gradually accumulated in the atmosphere, and as of 2013, its concentration is almost 43% above pre-industrial levels.,[30][31] Various techniques have been proposed for removing excess carbon dioxide from the atmosphere in carbon dioxide sinks.
wiki
There is a way to tell how much Co2 is from us, and how much is from natural sources. Here's the real kicker in the whole ridiculous argument that shouldn't even exist... regardless of how much naturally released Co2 or methane is in the atmosphere, we are the scale tippers, our emissions became the dominant forcing of climate change because we are adding so much more to the natural... that is all Anthropogenic Global Warming has ever meant. Climate responds to it's most dominant forcing.
The natural carbon cycle emits about 750 gigatons of Co2 per year as of 2010 we emit about 29 gigatons (it's increased since then) of co2 a year. Between land and the ocean about 770 gigatons of Co2 is absorbed per year, so the planet does take back most of what we put up there, however there's a remaining balance of Co2 left to deal with and thus it remains in the atmosphere completely outside the normal carbon cycle. The ocean is better carbon sink than land so it does take some of that added Co2 and that's why the oceans are warming faster than the surface, that is exactly where we find our temperature discrepancies and why sea ice in the arctic is melting more from the bottom than the top.
Arctic land ice however is melting so rapidly because of soot (dirty ice), black deposits on the ice absorb more heat from the sun and create pocs all over the surface ice allowing heat to sink further and further down. Land only exposed for the first time in hundreds of thousands of years if not millions is also warming, the exposed permafrost is thawing, releasing more carbon dioxide, more methane etc...
It's also unlikely that any methane from deep ocean floors will make it to the surface, methane dissolves in salt water... most of the methane is coming from land, shallow sea waters and lakes. That doesn't rule out volcanoes but I think it's clearly less likely volcanic activity owns the bulk of responsibility for extra GHG's.
Hating Al Gore is no reason to discount a theory he didn't come up with. A theory that there happens to be scientific consensus on.
Gravity is also a theory there's scientific consensus on, disbelieving that however, won't help you levitate.
edit on 22-6-2013 by Kali74
because: Said something backwards
reply to post by Moshpet
The theory that Redneck proposed is not IF there are volcanoes under the Arctic Ocean, there ARE volcanoes under the Arctic Ocean:
Arctic Volcanoes Found At Unprecedented Depths - National Geographic
Fire Under Ice -- Science Daily
Volcanoes Erupting Under Arctic Ice -- NBC News
Volcanoes Erupt Beneath Arctic Ice - Live Science
And it's plural: Volcanoes. Not one massive one.
If you look at the articles (all dating back to 2008), you see that some of them say that they (scientist) do not "believe" that they could be causing Arctic ice melt. It's important to remember that word, "believe" as many a researcher has had their "belief" in something end up changing when further data was collected, or had their belief confirmed
The theory that Redneck proposed is not IF there are volcanoes under the Arctic Ocean, there ARE volcanoes under the Arctic Ocean:
Arctic Volcanoes Found At Unprecedented Depths - National Geographic
Fire Under Ice -- Science Daily
Volcanoes Erupting Under Arctic Ice -- NBC News
Volcanoes Erupt Beneath Arctic Ice - Live Science
And it's plural: Volcanoes. Not one massive one.
If you look at the articles (all dating back to 2008), you see that some of them say that they (scientist) do not "believe" that they could be causing Arctic ice melt. It's important to remember that word, "believe" as many a researcher has had their "belief" in something end up changing when further data was collected, or had their belief confirmed
reply to post by TheRedneck
Read the evidence I have already presented, it explains why the Arctic ocean, and the entire planet is heating up, and where the heat comes from.
Science predicted decades ago what we are currently seeing.
See my comments above on your volcano theory. If an underwater volcano could increase the temperature of such a large area as the Arctic circle, then underwater volcano's in other parts of the planet would increase temperatures in the same way to the same extent. That is how thermal convection works.
I don't think you understand how big of an area we are talking about, an area larger than the Arctic circle.
Read the evidence I have already presented, it explains why the Arctic ocean, and the entire planet is heating up, and where the heat comes from.
Science predicted decades ago what we are currently seeing.
See my comments above on your volcano theory. If an underwater volcano could increase the temperature of such a large area as the Arctic circle, then underwater volcano's in other parts of the planet would increase temperatures in the same way to the same extent. That is how thermal convection works.
I don't think you understand how big of an area we are talking about, an area larger than the Arctic circle.
reply to post by Moshpet
Very well-phrased questions! It will be my pleasure to try and answer them.
1a. Not all volcanic lava forms pumice. It may also form scoria, which is similar but denser than water. The high pressures underneath the ocean cause most oceanic lava to form scoria instead of pumice.
1b. If the vent were close enough to the surface, there would be noticeable vents for gases to escape. At deeper depths, however, most of these gasses are absorbed by the water and released slowly at the surface when the warmed water rises. Such a rise is not necessarily noticeable with the naked eye; it depends on how fast the water is rising. Also, the heat is dissipated by the surrounding water. Remember that water can absorb a lot of heat energy in a small temperature change. Any steam formed at the site of the vent will return to liquid form as soon as the heat dissipates, which can be well below the surface.
2. Noticeable tectonic movement is not a requirement for a volcano to erupt. Bear in mind that not al eruptions are catastrophic events; volcanoes can also erupt slowly, spewing lava and superheated gasses over a long period of time. Hydrothermal vents are one form of volcanic exhaust, although very slow, and are undetectable from the surface. Wiki article
3. There has been little actual examination of the sea floor under the Arctic ice; recall that the volcanic vents I referenced earlier were only discovered a few years ago. The Arctic ice sheet itself poses a problem with examination of the seafloor underneath it. There would be no altitude change in the ice itself, such as a ground bulge like that which can occur with land-based volcanoes, since the volcano is located on the sea floor and not on the ice sheet itself.
The volcanic activity is there; that much is verified scientific fact. The only question is whether there is sufficient activity to melt the ice. Considering the other possibility, a change of a mere degree or two of temperature in the air, it makes more sense to me that the heat is coming from the water than from the air, and observation seems to back this up since the water is showing greater temperature anomalies than the air.
I hope this answered your questions; feel free to ask for a clarification if not.
TheRedneck
Very well-phrased questions! It will be my pleasure to try and answer them.
1a. Not all volcanic lava forms pumice. It may also form scoria, which is similar but denser than water. The high pressures underneath the ocean cause most oceanic lava to form scoria instead of pumice.
1b. If the vent were close enough to the surface, there would be noticeable vents for gases to escape. At deeper depths, however, most of these gasses are absorbed by the water and released slowly at the surface when the warmed water rises. Such a rise is not necessarily noticeable with the naked eye; it depends on how fast the water is rising. Also, the heat is dissipated by the surrounding water. Remember that water can absorb a lot of heat energy in a small temperature change. Any steam formed at the site of the vent will return to liquid form as soon as the heat dissipates, which can be well below the surface.
2. Noticeable tectonic movement is not a requirement for a volcano to erupt. Bear in mind that not al eruptions are catastrophic events; volcanoes can also erupt slowly, spewing lava and superheated gasses over a long period of time. Hydrothermal vents are one form of volcanic exhaust, although very slow, and are undetectable from the surface. Wiki article
3. There has been little actual examination of the sea floor under the Arctic ice; recall that the volcanic vents I referenced earlier were only discovered a few years ago. The Arctic ice sheet itself poses a problem with examination of the seafloor underneath it. There would be no altitude change in the ice itself, such as a ground bulge like that which can occur with land-based volcanoes, since the volcano is located on the sea floor and not on the ice sheet itself.
The volcanic activity is there; that much is verified scientific fact. The only question is whether there is sufficient activity to melt the ice. Considering the other possibility, a change of a mere degree or two of temperature in the air, it makes more sense to me that the heat is coming from the water than from the air, and observation seems to back this up since the water is showing greater temperature anomalies than the air.
I hope this answered your questions; feel free to ask for a clarification if not.
TheRedneck
Originally posted by poet1b
reply to post by eriktheawful
While you are at telling people how to have an intelligent discussion, maybe you can address all the anti-GW poster who throw their cheap shots up on the thread.
I would if I could keep up with all of them....and that goes for the AGW side too. Plenty of examples of them taking cheap shots too.
I've yet to see any AGW thread here on ATS debated in a civil way. They may start out that way.....and then spiral down into name calling, insulting and as you put it, ridiculing threads that boil down to:
"Yes it is!"
"No it isn't!"
(repeat over and over).
However, it never hurts to ask. Maybe, just maybe some will think about it and respond that way.......
More than likely not however.
I'm neither for or against AGW. I've seen WAY too much political agenda, bad science, manipulated data, and declarations of "having it all figured out" from both sides.
Again, it's like seeing only a handful of stars in the sky and declaring that they know everything there is to know in the universe............
The only thing I know for sure from watching all this over the past 15 years is: as far as Climate Change goes.....we still have a lot to learn about Climate Change.
reply to post by poet1b
I have read your evidence. Impressive. But there is one question that I cannot get past, and that is the fact that the air cannot contain enough energy in the temperature ranges we have experienced to raise the melting point of the ice sheet above the freezing level.
Water can contain sufficient energy to do this, since the water has a higher specific heat capacity and shows greater temperature anomalies. Ergo, the heat is coming from the water.
Water can warm land as well as air. Heat conducts through any material, at a speed dictated by the temperature differential, the insulating value of the material, and the thickness of the material. Earth has similar properties to water itself, since it typically contains a substantial amount of water dissolved in the soil.
If you took insult at my posts, I would like to apologize. As for the map, when you expressed how large the Arctic ice sheet is, contrary to the actual numbers which I posted earlier, I simply thought you, like many others, had an inflated idea of how large the area was. My intention was to educate, not to ridicule. The single country of Russia is itself much larger than the Arctic ice sheet.
I still maintain that 2.5% of the planet is far from global, and therefore is a localized event.
TheRedneck
I have read your evidence. Impressive. But there is one question that I cannot get past, and that is the fact that the air cannot contain enough energy in the temperature ranges we have experienced to raise the melting point of the ice sheet above the freezing level.
Water can contain sufficient energy to do this, since the water has a higher specific heat capacity and shows greater temperature anomalies. Ergo, the heat is coming from the water.
Water can warm land as well as air. Heat conducts through any material, at a speed dictated by the temperature differential, the insulating value of the material, and the thickness of the material. Earth has similar properties to water itself, since it typically contains a substantial amount of water dissolved in the soil.
If you took insult at my posts, I would like to apologize. As for the map, when you expressed how large the Arctic ice sheet is, contrary to the actual numbers which I posted earlier, I simply thought you, like many others, had an inflated idea of how large the area was. My intention was to educate, not to ridicule. The single country of Russia is itself much larger than the Arctic ice sheet.
I still maintain that 2.5% of the planet is far from global, and therefore is a localized event.
TheRedneck
reply to post by eriktheawful
That just needed to be posted again... I could not agree more!
(Oh, love the avater btw... big Molly Hatchet fan)
TheRedneck
The only thing I know for sure from watching all this over the past 15 years is: as far as Climate Change goes.....we still have a lot to learn about Climate Change.
That just needed to be posted again... I could not agree more!
(Oh, love the avater btw... big Molly Hatchet fan)
TheRedneck
reply to post by eriktheawful
Actually it is volcanic explosions, not plural volcanoes, from 1999, according to your own links.
How is it that a period of eruptions in 1999, over a 4 square mile area, is continuing to produce more heat to accelerate warming of the entire arctic region? You don't think that is much of a stretch?
Two of the four articles don't even mention global warming, the interest in in how the eruption is believed to have taken place. One article states,
One person states "we don't believe".
From the your 4th link.
Science does not back the volcano explanation for obvious reasons.
Actually it is volcanic explosions, not plural volcanoes, from 1999, according to your own links.
How is it that a period of eruptions in 1999, over a 4 square mile area, is continuing to produce more heat to accelerate warming of the entire arctic region? You don't think that is much of a stretch?
Two of the four articles don't even mention global warming, the interest in in how the eruption is believed to have taken place. One article states,
Scientists don't see any significant connection, however.
One person states "we don't believe".
From the your 4th link.
With news this week that polar ice is melting dramatically, underwater Arctic pyrotechnics might seem like a logical smoking gun. Scientists don't see any significant connection, however.
"We don't believe the volcanoes had much effect on the overlying ice," Reeves-Sohn told LiveScience, "but they seem to have had a major impact on the overlying water column."
Science does not back the volcano explanation for obvious reasons.
edit on 23-6-2013 by poet1b because: Changed from 1999 to in 1999
reply to post by TheRedneck
I agree, air alone does not explain this heating. From what I get from the science, there are several factors.
Warm ocean water from the Atlantic is probably the biggest contributor of heat. Warmer water from the rivers flowing into the Arctic is also a contributor. Then there is heat from the sun, trapped by CO2 and Methane. As the ice has receded year after year, the ocean has also absorbed more heat from the sun, because the dark waters absorb a great deal more heat than the white ice.
The bigger problem now is that the Methane holds much more heat, keeping heat from the sun trapped in the atmosphere, so the air above the Arctic, with higher levels of methane has a great deal more ability to warm and melt the ice. With Methane, the air has much more latent heat.
As the Arctic Ocean melts, it also absorbs less and less heat from the Atlantic, and the air. The system begins to feed on itself, just like lighting a fire.
I understand water heats land, and cools land, but that would have had to have been a massive volcanic eruption to heat 2.5% of the planet's surface area that much, and a body of water that large, thermally connected to the Atlantic and Pacific Ocean, and considerable land masses.
Apology accepted, and I apologize for taking a shot back. I always take these exchanges with a sense of humor. I get a lot of cheap shots from the anti-GW crowd, so I have to keep a sense of humor about it.
I agree, air alone does not explain this heating. From what I get from the science, there are several factors.
Warm ocean water from the Atlantic is probably the biggest contributor of heat. Warmer water from the rivers flowing into the Arctic is also a contributor. Then there is heat from the sun, trapped by CO2 and Methane. As the ice has receded year after year, the ocean has also absorbed more heat from the sun, because the dark waters absorb a great deal more heat than the white ice.
The bigger problem now is that the Methane holds much more heat, keeping heat from the sun trapped in the atmosphere, so the air above the Arctic, with higher levels of methane has a great deal more ability to warm and melt the ice. With Methane, the air has much more latent heat.
As the Arctic Ocean melts, it also absorbs less and less heat from the Atlantic, and the air. The system begins to feed on itself, just like lighting a fire.
I understand water heats land, and cools land, but that would have had to have been a massive volcanic eruption to heat 2.5% of the planet's surface area that much, and a body of water that large, thermally connected to the Atlantic and Pacific Ocean, and considerable land masses.
Apology accepted, and I apologize for taking a shot back. I always take these exchanges with a sense of humor. I get a lot of cheap shots from the anti-GW crowd, so I have to keep a sense of humor about it.
reply to post by poet1b
Warm water from the Atlantic also begs the question: where did the heat come from to warm it? The Atlantic influx is nothing new, so if it is delivering more heat, then the water coming in through it is warmer. We're back to warm water from an unknown source. That source cannot be CH4/CO2, because those gasses are in the atmosphere.
I do admit, openly and easily, that methane is a serious greenhouse gas. Unlike CO2, CH4 can easily absorb and re-emit heat along the entire spectrum emitted from the earth. The question, which I cannot answer, is whether even the 1800ppb level is enough to cause problems, and if so, how much of a problem? I do not think even the climate scientists know the answer to this, but I will agree it is troublesome enough to be monitored.
My reasoning tells me that if the CH4 in question is that contained in the permafrost, then it is not new to the atmosphere. Regardless of how long it may have been locked up in the frozen hydrates, it had to exist in the atmosphere at one time. If the CH4 is coming from other sources, that is not necessarily the case.
There is one bit of good news concerning the amount of increased absorption due to the lack of ice... the area in question is at the polar region, where sunlight is spread thinly to start with. This will minimize the problem, but not eliminate it.
I believe you misunderstand my hypothesis on the volcanic "eruptions"... as I tried to explain in a previous post, an eruption is not necessarily a single catastrophic event. If it were, you are correct that the event would have had to be massive in order to melt all the ice that is missing. Instead, these eruptions are slow, steady events that have apparently been happening for quite some time. Instead of a sudden explosion of lava and gases, it is a slow leak of lava and gasses, but enough of these slow leaks over a wide area over a prolonged period of time would raise water temperatures pretty much worldwide (as we have seen), air temperatures worldwide to a lesser degree (as we have seen), and more anomalous readings in the area affected, the Arctic (as we have seen).
The discoveries are also of widespread venting, not single vents.
Apology accepted. I will admit to having a healthy dose of skepticism when it comes to reports of global warming, primarily because I once believed the theory. When the predictions turned out to be inaccurate and I began researching the reports on my own, I discovered flaw after flaw in the reasoning and a political money-grabbing plot behind them. That makes it hard for me to accept anything reported on this subject at face value now.
A good example, and if I may, a demonstration of why the little barbs are thrown: In your reply to erictheawful, you posted a snippet where scientists alleged that while the volcanic activity was affecting the waters above the vents, it was not affecting the ice above those waters. Now think a moment: when have you seen ice that would not melt when placed in warming waters but would melt when slightly heated from above? I have never heard of such a thing; every scientific principle I can think of screams that this is impossible.
Vet your sources, lest they make you a target for ridicule. I know I try to vet mine.
And I am off for the night.
TheRedneck
Warm water from the Atlantic also begs the question: where did the heat come from to warm it? The Atlantic influx is nothing new, so if it is delivering more heat, then the water coming in through it is warmer. We're back to warm water from an unknown source. That source cannot be CH4/CO2, because those gasses are in the atmosphere.
I do admit, openly and easily, that methane is a serious greenhouse gas. Unlike CO2, CH4 can easily absorb and re-emit heat along the entire spectrum emitted from the earth. The question, which I cannot answer, is whether even the 1800ppb level is enough to cause problems, and if so, how much of a problem? I do not think even the climate scientists know the answer to this, but I will agree it is troublesome enough to be monitored.
My reasoning tells me that if the CH4 in question is that contained in the permafrost, then it is not new to the atmosphere. Regardless of how long it may have been locked up in the frozen hydrates, it had to exist in the atmosphere at one time. If the CH4 is coming from other sources, that is not necessarily the case.
There is one bit of good news concerning the amount of increased absorption due to the lack of ice... the area in question is at the polar region, where sunlight is spread thinly to start with. This will minimize the problem, but not eliminate it.
I believe you misunderstand my hypothesis on the volcanic "eruptions"... as I tried to explain in a previous post, an eruption is not necessarily a single catastrophic event. If it were, you are correct that the event would have had to be massive in order to melt all the ice that is missing. Instead, these eruptions are slow, steady events that have apparently been happening for quite some time. Instead of a sudden explosion of lava and gases, it is a slow leak of lava and gasses, but enough of these slow leaks over a wide area over a prolonged period of time would raise water temperatures pretty much worldwide (as we have seen), air temperatures worldwide to a lesser degree (as we have seen), and more anomalous readings in the area affected, the Arctic (as we have seen).
The discoveries are also of widespread venting, not single vents.
Apology accepted. I will admit to having a healthy dose of skepticism when it comes to reports of global warming, primarily because I once believed the theory. When the predictions turned out to be inaccurate and I began researching the reports on my own, I discovered flaw after flaw in the reasoning and a political money-grabbing plot behind them. That makes it hard for me to accept anything reported on this subject at face value now.
A good example, and if I may, a demonstration of why the little barbs are thrown: In your reply to erictheawful, you posted a snippet where scientists alleged that while the volcanic activity was affecting the waters above the vents, it was not affecting the ice above those waters. Now think a moment: when have you seen ice that would not melt when placed in warming waters but would melt when slightly heated from above? I have never heard of such a thing; every scientific principle I can think of screams that this is impossible.
Vet your sources, lest they make you a target for ridicule. I know I try to vet mine.
And I am off for the night.
TheRedneck
reply to post by Kali74
Thanks for the contribution and the excellent explanation.
From what I have read, it was not expected that methane would boil to the surface, but then they found kilometer wide plumes of methane coming out of the Asian continental shelf, which is the largest continental shelf on the planet. I guess those would be the shallow seas, unfortunately, there is a great deal of shallow sea in the Arctic ocean.
In 2008 when Arctic ice experienced a huge loss, I started taking notice, but it wasn't until the methane plumes were discovered that I realized how serous, and how soon we may see TSHTF.
Thanks for the contribution and the excellent explanation.
From what I have read, it was not expected that methane would boil to the surface, but then they found kilometer wide plumes of methane coming out of the Asian continental shelf, which is the largest continental shelf on the planet. I guess those would be the shallow seas, unfortunately, there is a great deal of shallow sea in the Arctic ocean.
In 2008 when Arctic ice experienced a huge loss, I started taking notice, but it wasn't until the methane plumes were discovered that I realized how serous, and how soon we may see TSHTF.
reply to post by TheRedneck
Those gases are not only in the atmosphere. 30-40% end up in the ocean. That causes ocean acidification, which of course is happening and attributed to industry.
The temperature rise in the oceans have everything to do with greenhouse gases and their effects globally. I'm not sure where you're not making the connection. Perhaps check the basic science regarding GHGs.
Those gases are not only in the atmosphere. 30-40% end up in the ocean. That causes ocean acidification, which of course is happening and attributed to industry.
The temperature rise in the oceans have everything to do with greenhouse gases and their effects globally. I'm not sure where you're not making the connection. Perhaps check the basic science regarding GHGs.
reply to post by poet1b
Thanks, I had a bit of insomnia last night and was kind of foggy and didn't explain some things quite right, I'll try to fix that here since it's too late to edit my post.
I said:
The ocean is a better carbon sink and takes some of the extra carbon dioxide out of the atmosphere however that causes ocean acidification. The warming is caused by the same process as that which is heating the surface, it just happens that their is more ocean on the surface of earth than land so of course it reabsorbs more of the energy trapped and reflected back down from the extra GHG's in the atmosphere.
You're right about the methane release being far more dangerous but I'm not sure it was never expected. I think it's been talked about for quite some time now. A quick google search tells me that the typical depth of ocean water over continental shelves is 200ft, which is indeed relatively shallow.
As far as heat from hydrothermal vents and magma at the arctic ocean floor... I don't know. The arctic sea floor is about 2 miles deep. Science tells us that the heat has to go somewhere it doesn't just disappear. What we don't seem to know is how much heat is coming up, how far up is it going?
We know that there are different layers of ocean water, different circulation rates... we know that some scientists think the heat is dissipated well before it could affect the ice, I just don't know how strong that consensus is... I'm curious to know that bit. I think it's reasonable that it could have some effect. Leading cause? Most definitely not. It certainly does not explain warming at the surface waters the world over nor to depths of 700m, we do have consensus on that cause.
Why anyone tries to deny that very simple science is flabbergasting.
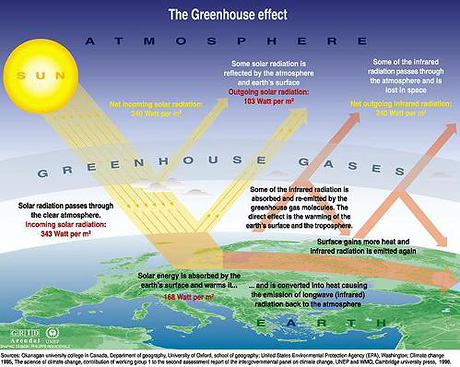
GHG's trap and release energy from the sun, GHG's in the stratosphere help keep the planet cool by trapping and releasing energy back into space, in the troposphere they trap and release that energy back toward the surface of the planet causing it to warm. The more GHG's in the troposphere, the more energy trapped and redirected back to earths surface. We are emitting upwards of 29 gigatons, of Co2 alone, per year... of which a portion of remains outside the carbon cycle and hangs around in the atmosphere.
Thanks, I had a bit of insomnia last night and was kind of foggy and didn't explain some things quite right, I'll try to fix that here since it's too late to edit my post.
I said:
The ocean is better carbon sink than land so it does take some of that added Co2 and that's why the oceans are warming faster than the surface, that is exactly where we find our temperature discrepancies
The ocean is a better carbon sink and takes some of the extra carbon dioxide out of the atmosphere however that causes ocean acidification. The warming is caused by the same process as that which is heating the surface, it just happens that their is more ocean on the surface of earth than land so of course it reabsorbs more of the energy trapped and reflected back down from the extra GHG's in the atmosphere.
You're right about the methane release being far more dangerous but I'm not sure it was never expected. I think it's been talked about for quite some time now. A quick google search tells me that the typical depth of ocean water over continental shelves is 200ft, which is indeed relatively shallow.
As far as heat from hydrothermal vents and magma at the arctic ocean floor... I don't know. The arctic sea floor is about 2 miles deep. Science tells us that the heat has to go somewhere it doesn't just disappear. What we don't seem to know is how much heat is coming up, how far up is it going?
We know that there are different layers of ocean water, different circulation rates... we know that some scientists think the heat is dissipated well before it could affect the ice, I just don't know how strong that consensus is... I'm curious to know that bit. I think it's reasonable that it could have some effect. Leading cause? Most definitely not. It certainly does not explain warming at the surface waters the world over nor to depths of 700m, we do have consensus on that cause.
Why anyone tries to deny that very simple science is flabbergasting.

GHG's trap and release energy from the sun, GHG's in the stratosphere help keep the planet cool by trapping and releasing energy back into space, in the troposphere they trap and release that energy back toward the surface of the planet causing it to warm. The more GHG's in the troposphere, the more energy trapped and redirected back to earths surface. We are emitting upwards of 29 gigatons, of Co2 alone, per year... of which a portion of remains outside the carbon cycle and hangs around in the atmosphere.
Good morning everyone.
reply to post by wehavenoclue
In order to act as a greenhouse gas, CH4/CO2 must be in a position to absorb and re-emit infrared radiation. If they cannot absorb and re-emit infrared radiation, they cannot act as a greenhouse gas. That is the very basis of the theory.
In the atmosphere, radiation from the earth can encounter a greenhouse gas molecule, be absorbed, then be re-emitted in a random direction, meaning a certain percentage will be be re-emitted toward the earth. In the ocean, there is no absorption and re-emission, because there is no radiation emitted through the water; if any is emitted, water itself will insulate so well as to make any dissolved gasses irrelevant. All oceanic heat emission is from the surface into the atmosphere.
Water vapor and CH4 are excellent greenhouse gasses on the earth, because they can absorb the entire range of frequencies of infrared radiation emitted by the plant. CO2 is not as much of a greenhouse gas, because it has narrow absorption bands which do not perfectly coincide with the radiation frequencies emitted by the earth. CO2 tends to absorb only the lower frequencies. If the earth were to cool, CO2 absorption bands would be more aligned with the emitted frequencies and CO2 would be more effective as a greenhouse gas; if we were to warm, the alignment would be less exact and CO2 would be even less effective as a greenhouse gas. I have not mentioned this before because I wanted to focus on the Arctic situation instead of the basic science, but since I am having to explain it anyway...
Ocean acidification also aligns geographically with shipping lanes and oceanic currents around them. Considering that ships are allowed to burn fuel extremely high in sulfur content (5% in some cases, as opposed to 0.015% for diesel trucks in the US), and given that sulfuric acid is much more potent than carbonic acid, I tend to blame the sulfur before I do the CO2.
Please don't try and tell me how potent carbonic acid is... I'm drinking some right now: Mountain Dew. I have no intention of drinking battery acid (sulfuric acid).
CH4 is not an issue in water, as it tends to break down in contact with the hydroxyl ions in seawater into CO2 and H20 in the equation CH4 + 8OH- --> CO2 + 6H2O + 8e-. On further consideration, I would doubt that the elevated methane levels in the Arctic are directly associated with volcanic activity; they are probably a result of permafrost melting.
Finally, in order for heat energy to move into a substance, there must be a negative temperature differential between the substance being heated and the substance doing the heating. If the air is responsible for the heating as in greenhouse gas effects, it is simply not possible for the water underneath the air to increase in temperature to a level greater than the air. Conversely, if the water is doing the heating, the air cannot become warmer than the water. Check the anomaly levels around the Arctic and you will see that the water is experiencing greater temperature anomalies than the air.
TheRedneck
reply to post by wehavenoclue
In order to act as a greenhouse gas, CH4/CO2 must be in a position to absorb and re-emit infrared radiation. If they cannot absorb and re-emit infrared radiation, they cannot act as a greenhouse gas. That is the very basis of the theory.
In the atmosphere, radiation from the earth can encounter a greenhouse gas molecule, be absorbed, then be re-emitted in a random direction, meaning a certain percentage will be be re-emitted toward the earth. In the ocean, there is no absorption and re-emission, because there is no radiation emitted through the water; if any is emitted, water itself will insulate so well as to make any dissolved gasses irrelevant. All oceanic heat emission is from the surface into the atmosphere.
Water vapor and CH4 are excellent greenhouse gasses on the earth, because they can absorb the entire range of frequencies of infrared radiation emitted by the plant. CO2 is not as much of a greenhouse gas, because it has narrow absorption bands which do not perfectly coincide with the radiation frequencies emitted by the earth. CO2 tends to absorb only the lower frequencies. If the earth were to cool, CO2 absorption bands would be more aligned with the emitted frequencies and CO2 would be more effective as a greenhouse gas; if we were to warm, the alignment would be less exact and CO2 would be even less effective as a greenhouse gas. I have not mentioned this before because I wanted to focus on the Arctic situation instead of the basic science, but since I am having to explain it anyway...
Ocean acidification also aligns geographically with shipping lanes and oceanic currents around them. Considering that ships are allowed to burn fuel extremely high in sulfur content (5% in some cases, as opposed to 0.015% for diesel trucks in the US), and given that sulfuric acid is much more potent than carbonic acid, I tend to blame the sulfur before I do the CO2.
Please don't try and tell me how potent carbonic acid is... I'm drinking some right now: Mountain Dew. I have no intention of drinking battery acid (sulfuric acid).
CH4 is not an issue in water, as it tends to break down in contact with the hydroxyl ions in seawater into CO2 and H20 in the equation CH4 + 8OH- --> CO2 + 6H2O + 8e-. On further consideration, I would doubt that the elevated methane levels in the Arctic are directly associated with volcanic activity; they are probably a result of permafrost melting.
Finally, in order for heat energy to move into a substance, there must be a negative temperature differential between the substance being heated and the substance doing the heating. If the air is responsible for the heating as in greenhouse gas effects, it is simply not possible for the water underneath the air to increase in temperature to a level greater than the air. Conversely, if the water is doing the heating, the air cannot become warmer than the water. Check the anomaly levels around the Arctic and you will see that the water is experiencing greater temperature anomalies than the air.
TheRedneck
edit on 6/23/2013 by TheRedneck because: (no reason given)
According to a report in climate depot, the arctic is having its coldest start to the summer since records began, in 1958. Also another reports
states that Antarctic ice is up 10% from 'normal' .
Originally posted by TheRedneck
Good morning everyone.
reply to post by wehavenoclue
In order to act as a greenhouse gas, CH4/CO2 must be in a position to absorb and re-emit infrared radiation. If they cannot absorb and re-emit infrared radiation, they cannot act as a greenhouse gas. That is the very basis of the theory.
Agreed.
In the atmosphere, radiation from the earth can encounter a greenhouse gas molecule, be absorbed, then be re-emitted in a random direction, meaning a certain percentage will be be re-emitted toward the earth. In the ocean, there is no absorption and re-emission, because there is no radiation emitted through the water; if any is emitted, water itself will insulate so well as to make any dissolved gasses irrelevant. All oceanic heat emission is from the surface into the atmosphere.
You have some of this correct, and some of this incorrect.
The radiation starts from the sun, penetrates the atmosphere, bounces off the earth, and then may be reflected back to the earth in the atmosphere, in higher quantities the more greenhouse gases there are.
The ocean most certainly absorbs and remits radiation. I'm not sure why you think it doesn't. Even if this wasn't the case, all it would take is radiation bouncing off the land, and being reflected at an angle in the atmosphere to land it in the ocean for it to be absorbed.
A pool is heated by the sun in the summer, and chills in the winter. Why would you think the ocean doesn't do the same
reply to post by wehavenoclue
The ocean certainly absorbs and re-emits radiation at its surface, not in the deep ocean depths. Heat transfer through the water is accomplished by conduction and convection, not by radiation. Any radiation emitted by water molecules in the ocean depths is immediately absorbed by neighboring molecules... this immediate absorption is considered conduction rather than radiation because there is no time between emission and absorption for the properties of radiation to become apparent; for all practical purposes, it acts no differently than a purely kinetic energy exchange. Thus, no amount of greenhouse gas dissolved in sea water makes a single iota of difference in heat transfer characteristics.
All this is starting to sound like an argument over semantics. My point is that greenhouse gas theory is only applicable in the atmosphere. Can you agree with that statement?
TheRedneck
The ocean certainly absorbs and re-emits radiation at its surface, not in the deep ocean depths. Heat transfer through the water is accomplished by conduction and convection, not by radiation. Any radiation emitted by water molecules in the ocean depths is immediately absorbed by neighboring molecules... this immediate absorption is considered conduction rather than radiation because there is no time between emission and absorption for the properties of radiation to become apparent; for all practical purposes, it acts no differently than a purely kinetic energy exchange. Thus, no amount of greenhouse gas dissolved in sea water makes a single iota of difference in heat transfer characteristics.
All this is starting to sound like an argument over semantics. My point is that greenhouse gas theory is only applicable in the atmosphere. Can you agree with that statement?
TheRedneck
reply to post by TheRedneck
I see the disconnect, and yes it was mostly a matter of semantics.
I don't agree that the effects of greenhouse gases will only effect the surface. I understand that it won't be directly from the radiation, but tend to think that over time the accumulation of GHG's will cause a rise in temperature at greater depths in the oceans. Let me see if I can find some science to back up this assertion.
I see the disconnect, and yes it was mostly a matter of semantics.
I don't agree that the effects of greenhouse gases will only effect the surface. I understand that it won't be directly from the radiation, but tend to think that over time the accumulation of GHG's will cause a rise in temperature at greater depths in the oceans. Let me see if I can find some science to back up this assertion.
edit on 23-6-2013 by wehavenoclue because: (no reason given)
new topics
-
Thousands Of Young Ukrainian Men Trying To Flee The Country To Avoid Conscription And The War
Other Current Events: 8 minutes ago -
12 jurors selected in Trump criminal trial
US Political Madness: 2 hours ago -
Iran launches Retalliation Strike 4.18.24
World War Three: 3 hours ago -
Israeli Missile Strikes in Iran, Explosions in Syria + Iraq
World War Three: 3 hours ago -
George Knapp AMA on DI
Area 51 and other Facilities: 9 hours ago -
Not Aliens but a Nazi Occult Inspired and then Science Rendered Design.
Aliens and UFOs: 9 hours ago -
Louisiana Lawmakers Seek to Limit Public Access to Government Records
Political Issues: 11 hours ago
top topics
-
BREAKING: O’Keefe Media Uncovers who is really running the White House
US Political Madness: 16 hours ago, 25 flags -
George Knapp AMA on DI
Area 51 and other Facilities: 9 hours ago, 24 flags -
Biden--My Uncle Was Eaten By Cannibals
US Political Madness: 17 hours ago, 18 flags -
Israeli Missile Strikes in Iran, Explosions in Syria + Iraq
World War Three: 3 hours ago, 12 flags -
"We're All Hamas" Heard at Columbia University Protests
Social Issues and Civil Unrest: 17 hours ago, 7 flags -
Louisiana Lawmakers Seek to Limit Public Access to Government Records
Political Issues: 11 hours ago, 7 flags -
Not Aliens but a Nazi Occult Inspired and then Science Rendered Design.
Aliens and UFOs: 9 hours ago, 5 flags -
So I saw about 30 UFOs in formation last night.
Aliens and UFOs: 14 hours ago, 5 flags -
Iran launches Retalliation Strike 4.18.24
World War Three: 3 hours ago, 5 flags -
Do we live in a simulation similar to The Matrix 1999?
ATS Skunk Works: 15 hours ago, 4 flags
active topics
-
Elites disapearing
Political Conspiracies • 30 • : SchrodingersRat -
Not Aliens but a Nazi Occult Inspired and then Science Rendered Design.
Aliens and UFOs • 10 • : OmegaLogos -
12 jurors selected in Trump criminal trial
US Political Madness • 21 • : VictorVonDoom -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 64 • : Degradation33 -
Iran launches Retalliation Strike 4.18.24
World War Three • 14 • : Cloudbuster1 -
Thousands Of Young Ukrainian Men Trying To Flee The Country To Avoid Conscription And The War
Other Current Events • 0 • : Consvoli -
African "Newcomers" Tell NYC They Don't Like the Free Food or Shelter They've Been Given
Social Issues and Civil Unrest • 17 • : SchrodingersRat -
Canadian Forces bow out and loose interest in UFO’s
Aliens and UFOs • 20 • : Ophiuchus1 -
Israeli Missile Strikes in Iran, Explosions in Syria + Iraq
World War Three • 50 • : WeMustCare -
Max Loughan Boy Genius Or Something More?
Science & Technology • 69 • : SchrodingersRat
