It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
0
share:
I was thinking about how everything seems to be drier than it used to be. We have not gotten a good amount of snow or rain in a long time.
Is our water vapor leaking through the hole in the ozone?
Is our water vapor leaking through the hole in the ozone?
edit on 14-2-2013 by MrBigDave because: my bad
reply to post by MrBigDave
the uk have seen the wettest summer in 100 years in 2012 and not much difference this year so far
the uk have seen the wettest summer in 100 years in 2012 and not much difference this year so far
reply to post by MrBigDave
Sorta sounds like a dumb question but I wonder... The earth's water cycle definitely has been disrupted - and while some places are experiencing the opposite of drought (heavy precipitation and flooding) I recall reading that our fresh water IS disappearing and not being "recycled."
Sorta sounds like a dumb question but I wonder... The earth's water cycle definitely has been disrupted - and while some places are experiencing the opposite of drought (heavy precipitation and flooding) I recall reading that our fresh water IS disappearing and not being "recycled."
some places are wetter this year some places are drier, I think the weather patterns have been disrupted somehow or maybe this is just a normal long
term weather cycle.
There seems to be plenty of water in the oceans, people keep claiming the ocean level is raising.
There seems to be plenty of water in the oceans, people keep claiming the ocean level is raising.
Originally posted by MrBigDave
I was thinking about how everything seems to be drier than it used to be. We have not gotten a good amount of snow or rain in a long time.
Is our water vapor leaking through the hole in the ozone?edit on 14-2-2013 by MrBigDave because: my bad
50% more snow this year compared to last year.
reply to post by MrBigDave
DUH.....
That's where the aliens come to our planet through.....That hole in the ozone I mean....
Everybody knows that......../sarcasm
DUH.....
That's where the aliens come to our planet through.....That hole in the ozone I mean....
Everybody knows that......../sarcasm
Originally posted by MrBigDave
I was thinking about how everything seems to be drier than it used to be. We have not gotten a good amount of snow or rain in a long time.
Is our water vapor leaking through the hole in the ozone?edit on 14-2-2013 by MrBigDave because: my bad
Brief answer: no.
Longer answer: 99% of our water vapor is in the lowest layer of the atmosphere, the troposphere. If any molecules (which are heavier than plain oxygen (02) because they have an attached hydrogen atom) get into the stratosphere, they usually get ionized.
So, water vapor is so heavy that 99% of it never makes it as far as the ozone layer.
reply to post by MrBigDave
More...
Our Planets Disappearing Drinking Water
World Water Day Pictures: Epic Disappearing Acts
More...
Our Planets Disappearing Drinking Water
World Water Day Pictures: Epic Disappearing Acts
Middle Eastern fresh water disappearing fast
The Middle East has lost fresh water reserves equivalent to the entire Dead Sea over the last ten years, data from NASA satellites shows.
Feedback Loops: The Basic Problem
Human consumption of water and interaction with the water cycle both contribute to water stress in a complex dynamic that has resulted in environmental damage and risks to human security.
[
Brief answer: no.
Longer answer: 99% of our water vapor is in the lowest layer of the atmosphere, the troposphere. If any molecules (which are heavier than plain oxygen (02) because they have an attached hydrogen atom) get into the stratosphere, they usually get ionized.
So, water vapor is so heavy that 99% of it never makes it as far as the ozone layer.
While your conclusion is correct, your chemistry is way off. Diatomic oxygen(O2), the natural state in which most oxygen exists has a molecular weight of 31.88 +/- 0.0008 grams/mol.. Water vapor (H2O) has a molecular weight of 15.83 g/mol. So, water vapor is about half the weight of diatomic oxygen. I'm guessing you got confused about the chemical formula for water and were thinking of HO2, which does exist in the atmosphere as a hydroperoxl radical, an important atmospheric cleansing agent.
The real reason that water vapor exists in the troposphere and not higher is that the adiabatic lapse rate results in saturation and precipitation as the air gets cooler. Each pound of dry air today in Miami (60% humidity) can carry 159 grams of water vapor. At 15,000 feet, it is down to about 14 grams water vapor per pound of air.At 39,000 over Miami today, the temperature is -65F. At that temperature the water vapor load carrying ability is almost 0. At an average, the ozone layer is at about 75,000 feet. So water vapor would condense or freeze and fall to earth long before it gets there.
In addition to all the informative replies posted here.
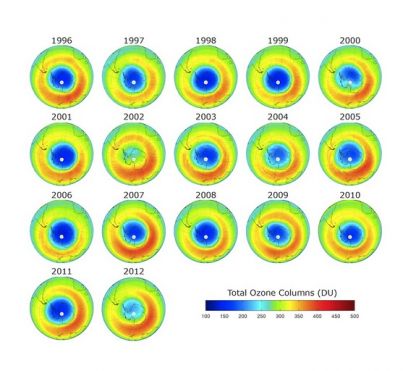
The hole is the ozone has gotten smaller lately.
Ozone Layer Is On The Road To Recovery
Is the hole in the ozone closing?
Ozone hole shrinks to record low
Smallest it's been in ten years.
The hole is the ozone has gotten smaller lately.
Ozone Layer Is On The Road To Recovery
Is the hole in the ozone closing?
Ozone hole shrinks to record low
Smallest it's been in ten years.

Perhaps there's an outside influence...
Remember in the miniseries "V" what the aliens were here for?
Remember in the miniseries "V" what the aliens were here for?
The true purpose of the Visitors' arrival on Earth was to conquer and subdue the planet, steal all of the Earth's water, and harvest the human race as food, leaving only a few as slaves and cannon fodder for the Visitors' wars with other alien races.
reply to post by MrBigDave
This↓ may not actually be relevant for this specific topic. It is something that I had forgotten about, until now. I just think that
it's quite interesting.(even though it is difficult to believe)
[color=A6DBFF]The Earth is pretty much a "closed system," like a terrarium. That means that the Earth as a whole, neither gains nor loses much matter, including water.
....very little of Earth's substances escape into outer space. This is certainly true about water.
This means that the same water that existed on Earth millions of years ago is still here. Thanks to the water cycle, the same water is continually being recycled all around the globe.
ga.water.usgs.gov...
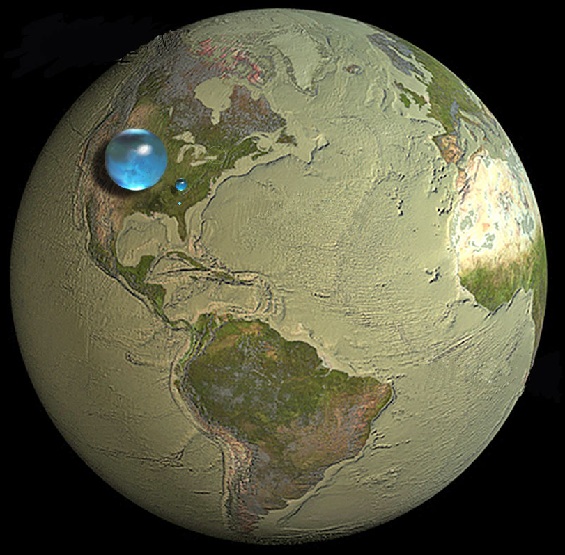
[color=87B1CF]The drawings below show various blue spheres representing relative amounts of Earth's water in comparison to the size of the Earth. Are you surprised that these water spheres look so small? They are only small in relation to the size of the Earth.
These images attempt to show three dimensions, so each sphere represents "volume." Overall, it shows that in comparison to the volume of the globe the amount of water on the planet is very small - and the oceans are only a "thin film" of water on the surface.
[color=A6DBFF]Large Sphere = ALL of Earth's water
Medium Sphere = Earth's liquid fresh water
Tiny Sphere= Water in lakes and rivers
ga.water.usgs.gov...
edit on 2/14/13 by BrokenCircles because: (no reason given)
I thought it was the aliens from V that were stealing our water! LoL
Good question Mr Big Dave, I am glad tha answer seems to be no. In Australia we had the wettest two years on record 2010/2011. The north east of the
country is still sodden and has just had huge floods again, but the south east has been in drought for the last 5 months. Where I am we have had only
1 mm (1/25 inch) of rain in the last 5 months!
edit on 15/2/13 by Cinrad because: (no reason given)
reply to post by BrokenCircles
That graphic of all the liquid water on earth has always puzzled me. It seems much too small, especially the fresh water and river water. It looks like if the bubbles broke and the water spread out it would fill a little bit of space in the United States and that's all. How can those bubbles represent all the fresh water in the world?
That graphic of all the liquid water on earth has always puzzled me. It seems much too small, especially the fresh water and river water. It looks like if the bubbles broke and the water spread out it would fill a little bit of space in the United States and that's all. How can those bubbles represent all the fresh water in the world?
new topics
-
This is our Story
General Entertainment: 30 minutes ago -
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections: 2 hours ago -
Ode to Artemis
General Chit Chat: 3 hours ago -
Ditching physical money
History: 6 hours ago -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 7 hours ago -
Don't take advantage of people just because it seems easy it will backfire
Rant: 7 hours ago -
VirginOfGrand says hello
Introductions: 8 hours ago -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 8 hours ago -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 11 hours ago
top topics
-
Hate makes for strange bedfellows
US Political Madness: 17 hours ago, 20 flags -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 11 hours ago, 12 flags -
Police clash with St George’s Day protesters at central London rally
Social Issues and Civil Unrest: 14 hours ago, 9 flags -
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections: 2 hours ago, 8 flags -
TLDR post about ATS and why I love it and hope we all stay together somewhere
General Chit Chat: 15 hours ago, 7 flags -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 8 hours ago, 6 flags -
Don't take advantage of people just because it seems easy it will backfire
Rant: 7 hours ago, 4 flags -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 7 hours ago, 4 flags -
God lived as a Devil Dog.
Short Stories: 12 hours ago, 3 flags -
Ditching physical money
History: 6 hours ago, 3 flags
active topics
-
New whistleblower Jason Sands speaks on Twitter Spaces last night.
Aliens and UFOs • 47 • : Ophiuchus1 -
Hate makes for strange bedfellows
US Political Madness • 39 • : 19Bones79 -
Lawsuit Seeks to ‘Ban the Jab’ in Florida
Diseases and Pandemics • 29 • : Cre8chaos79 -
TLDR post about ATS and why I love it and hope we all stay together somewhere
General Chit Chat • 8 • : Cre8chaos79 -
This is our Story
General Entertainment • 0 • : BrotherKinsMan -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 126 • : Asher47 -
Why to avoid TikTok
Education and Media • 17 • : mooncake -
The Superstition of Full Moons Filling Hospitals Turns Out To Be True!
Medical Issues & Conspiracies • 22 • : mooncake -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media • 23 • : BigDuckEnergy -
President BIDEN Vows to Make Americans Pay More Federal Taxes in 2025 - Political Suicide.
2024 Elections • 3 • : BingoMcGoof
0