It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
scitechdaily.com...
The building blocks of life surrounding a young star !
How cool
Is the creation of life going to begin while Man watches?
The building blocks of life surrounding a young star !
How cool
Is the creation of life going to begin while Man watches?
Sugar Molecules Found in Gas Surrounding Sun-Like Star
August 29, 2012 by Staff
Using the Atacama Large Millimeter/submillimeter Array, a team of astronomers discovered molecules of a simple form of sugar in the gas surrounding a young binary star called IRAS 16293-2422, which is located roughly 400 light-years away.
“In the disc of gas and dust surrounding this newly formed star, we found glycolaldehyde, which is a simple form of sugar, not much different to the sugar we put in coffee,” explains Jes Jorgensen (Niels Bohr Institute, Denmark), the lead author of the paper. “This molecule is one of the ingredients in the formation of RNA, which — like DNA, to which it is related — is one of the building blocks of life.”
“What it is really exciting about our findings is that the ALMA observations reveal that the sugar molecules are falling in towards one of the stars of the system,” says team member Cécile Favre (Aarhus University, Denmark). “The sugar molecules are not only in the right place to find their way onto a planet, but they are also going in the right direction.”
scitechdaily.com...
This is a new star and has allready sending out waves of radition giving us the ability to to map it.
The glycolaldehyde Will hopfully have a safe progression to a star and bind with other complex particles, change to an intricate enough form, and merge into the beginings of a young planet.
Sweet! pun intended
Very exciting stuff happening most everyday now!
Found where the original info. was first placed on the net, entitled:
Sweet Result from ALMA
www.almaobservatory.org...
The research Paper:
www.almaobservatory.org...
A video on this can be found at:
vimeo.com...
Sweet Result from ALMA
www.almaobservatory.org...
The research Paper:
www.almaobservatory.org...
A video on this can be found at:
vimeo.com...
edit on 29-8-2012 by azureskys because: made bad choice corrected it
Heres a pic of the compound. red=Oxygen black=carbon white=hydrogen
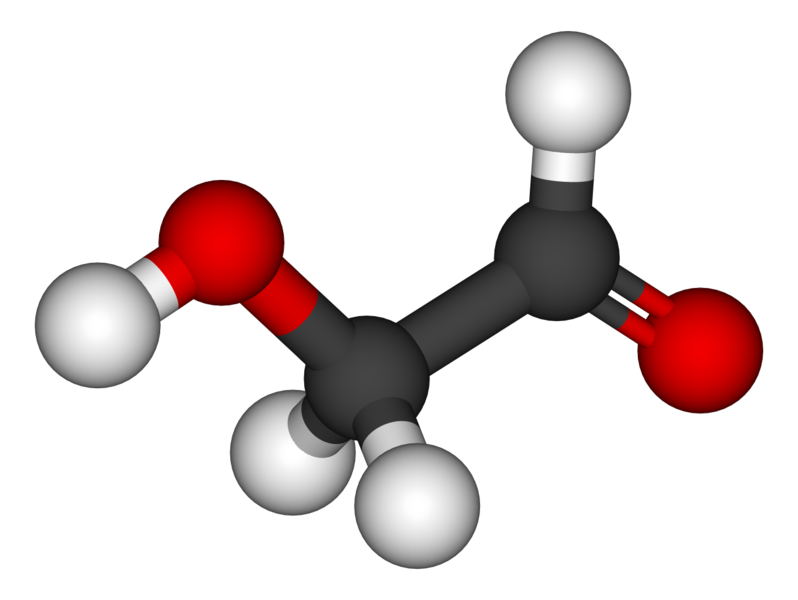
Fairly simple organic compound. I wounder what the synthesis was and what intermediates the star went through to get this aldehyde.

Fairly simple organic compound. I wounder what the synthesis was and what intermediates the star went through to get this aldehyde.
edit on
29-8-2012 by BriGuyTM90 because: (no reason given)
reply to post by BriGuyTM90
Oh yeah my biggest downfall in the consumption of edibles, carbohydrates!!
Yum
Oh yeah my biggest downfall in the consumption of edibles, carbohydrates!!
Yum
reply to post by azureskys
All I do is eat star dust. So we ve found Ethyl Alcohol and simple sugars around stars now we just need to find a star with menthol and we could make our selves the biggest Mojito ever made.
All I do is eat star dust. So we ve found Ethyl Alcohol and simple sugars around stars now we just need to find a star with menthol and we could make our selves the biggest Mojito ever made.
If its 400 light years away, wouldn't this already have potentially happened if it was going to?
How old is the light we are observing from this star now?
How old is the light we are observing from this star now?
reply to post by Signals
400 years ago, but if its happening around this star its more than likly happening around a bunch of stars threw out our universe. so the fact that that this happened 400 years ago in this star system doesn't make the finding any less meaningful
400 years ago, but if its happening around this star its more than likly happening around a bunch of stars threw out our universe. so the fact that that this happened 400 years ago in this star system doesn't make the finding any less meaningful
edit on 29-8-2012 by BriGuyTM90 because:
(no reason given)
reply to post by azureskys
how do they detect a chemical compound 400 light years away, that i cant detect 4 inches away?
how do they detect a chemical compound 400 light years away, that i cant detect 4 inches away?
edit on 29-8-2012 by ImaFungi because: (no
reason given)
reply to post by ImaFungi
Spectroscopy. Im not exactly sure which type they used to find out this specific compound. but probly some sort of diffraction spectroscopy.
Spectroscopy. Im not exactly sure which type they used to find out this specific compound. but probly some sort of diffraction spectroscopy.
reply to post by ImaFungi
Through the beauty of spectral analyses. When you split that light up into it's base components it leaves telltale signatures for the various elements within the star and it's surrounding proto-disk.
Through the beauty of spectral analyses. When you split that light up into it's base components it leaves telltale signatures for the various elements within the star and it's surrounding proto-disk.
Hopefully it's not too much sugar.
I'd hate for such a new young planet to live life with diabetes.
Tragic, really. Are we going to find something out there low-carb next?
I'd hate for such a new young planet to live life with diabetes.
Tragic, really. Are we going to find something out there low-carb next?
reply to post by LaughingatHumanity
ok thanks.,,.., when they split the light up,,,, how do they weed through all the other debris that light traveled past before being received on earth? how do they know they werent getting readings from the light passing by these compounds on earth right before it was received?
ok thanks.,,.., when they split the light up,,,, how do they weed through all the other debris that light traveled past before being received on earth? how do they know they werent getting readings from the light passing by these compounds on earth right before it was received?
reply to post by BriGuyTM90
Response to:
Well, we know formaldehyde molecules and heat were part of it some how and heat.
And just how long did it take to get to the point of forming the simplest if sugars? Amazing
Glycolaldehyde (HCOCH2OH)
Response to:
"Fairly simple organic compound. I wounder what the synthesis was and what intermediates the star went through to get this aldehyde."
Well, we know formaldehyde molecules and heat were part of it some how and heat.
And just how long did it take to get to the point of forming the simplest if sugars? Amazing
Glycolaldehyde (HCOCH2OH)
edit on 29-8-2012 by azureskys because: added more
Originally posted by ImaFungi
reply to post by LaughingatHumanity
ok thanks.,,.., when they split the light up,,,, how do they weed through all the other debris that light traveled past before being received on earth? how do they know they werent getting readings from the light passing by these compounds on earth right before it was received?
when an electron of a atom get excited to its next energy level it emits certain frequencies of light. that light is then detected on the earth and analyzed. Now there is oxygen and nitrogen in our atmosphere and those frequencies have to be removed by calibrating the instrument to ignore them or a trained person has to manually go threw the data and pull out the meaningful spectrum. Also positions of things in the night sky can be triangulated if they are relatively close to earth.
edit on 29-8-2012 by BriGuyTM90 because: (no reason given)
edit on 29-8-2012 by
BriGuyTM90 because: (no reason given)
reply to post by ImaFungi
A recognized radiation signature is what they picked up on to identify them.
www.sci-news.com...
A recognized radiation signature is what they picked up on to identify them.
once a star has been formed in the middle of a rotating cloud of gas and dust, it heats the inner parts of the cloud to around room temperature, evaporating the chemically complex molecules, and forming gases that emit their characteristic radiation as radio waves that can be mapped using powerful radio telescopes such as ALMA.
www.sci-news.com...
reply to post by BriGuyTM90
Here's one synth that seems feasible in that environment CO(+)H+(+)CH2O= C2H4O2 using a cobalt catalyst. Or Carbon monoxide + hydrogen ion +Formaldehyde(methanal) using a cobalt catalyst will result in glycoladehyde. Now IDK if the cobalt catalyst is just used to reach activation energy level if so this is not needed because you have a star next to the reaction. But either way cobalt is not that rare of an element and I'm sure it is present in protoplanetary disk. Also this route has a low yield but this is happening on a stellar scale.
Here's one synth that seems feasible in that environment CO(+)H+(+)CH2O= C2H4O2 using a cobalt catalyst. Or Carbon monoxide + hydrogen ion +Formaldehyde(methanal) using a cobalt catalyst will result in glycoladehyde. Now IDK if the cobalt catalyst is just used to reach activation energy level if so this is not needed because you have a star next to the reaction. But either way cobalt is not that rare of an element and I'm sure it is present in protoplanetary disk. Also this route has a low yield but this is happening on a stellar scale.
edit on 29-8-2012 by
BriGuyTM90 because: (no reason given)
To answer your second question first:
The light we see today is 400 years old.
400 years is just a blink of an eye (even less than that) in the timescale of the universe. It is almost certain that the star has not changed much in the 400 years it took for that light to reach us. How we see the star (with 400 year-old information) is likely exactly the way the star really is right now.
Originally posted by Signals
How old is the light we are observing from this star now?
The light we see today is 400 years old.
Originally posted by Signals
If its 400 light years away, wouldn't this already have potentially happened if it was going to?
400 years is just a blink of an eye (even less than that) in the timescale of the universe. It is almost certain that the star has not changed much in the 400 years it took for that light to reach us. How we see the star (with 400 year-old information) is likely exactly the way the star really is right now.
edit on 8/29/2012 by Soylent Green Is People because: (no reason given)
I was under the impression that all it took was,
two formaldehyde molecules condensing to make glycolaldehyde
Am I wrong?
Found this to back me up:
www.ch.ic.ac.uk...
two formaldehyde molecules condensing to make glycolaldehyde
Am I wrong?
Found this to back me up:
Glycolaldehyde is thought to have been synthesised on the prebiotic Earth via the Formose reaction(4). This is where molecules of formaldehyde polymerise, or react together to form the larger molecule of Glycolaldehyde.
There is abundant gaseous formaldehyde in dense clouds and also some holding on tight to grains. This could also be the reaction forming Glycolaldehyde in space. The reaction would be catalysed or kick stated by UV radiation from stars.
www.ch.ic.ac.uk...
edit on 29-8-2012 by azureskys because: added more information
edit on 29-8-2012 by azureskys because: dito
reply to post by azureskys
Im not 100% but Im pretty sure you cant, Glycolaldehyde has Two carbons with two different functional groups. C(1) has Hydrogen and it has a double bonded Oxygen(Ketone)When a ketone is on the end of the molecule its called an Aldehyde. Now carbon(2) has a Hydoxyl group and two Hydrogen. It would be a reduction reaction not condensation that's just has to do with phase change. I don't think Formaldehyde will reduce with its self and break the double bound of the ketone. Also if you do happen to get them to reduce I don't think the product would be Glycoladehyde. Here's a pic I made with labels to help you see what I'm talking about the second pic is the molecule I think would come out of the reduction of Formaldehyde with its self and its systematic name I cant find anything on the web about it so its probably not stable.
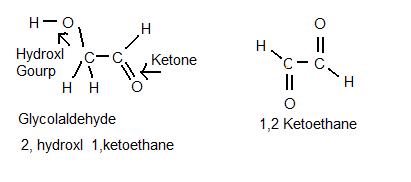
oh well nvm I was wrong guess you can
Im not 100% but Im pretty sure you cant, Glycolaldehyde has Two carbons with two different functional groups. C(1) has Hydrogen and it has a double bonded Oxygen(Ketone)When a ketone is on the end of the molecule its called an Aldehyde. Now carbon(2) has a Hydoxyl group and two Hydrogen. It would be a reduction reaction not condensation that's just has to do with phase change. I don't think Formaldehyde will reduce with its self and break the double bound of the ketone. Also if you do happen to get them to reduce I don't think the product would be Glycoladehyde. Here's a pic I made with labels to help you see what I'm talking about the second pic is the molecule I think would come out of the reduction of Formaldehyde with its self and its systematic name I cant find anything on the web about it so its probably not stable.

edit on 29-8-2012 by BriGuyTM90 because: (no reason given)
edit on 29-8-2012 by BriGuyTM90 because:
(no reason given)
edit on 30-8-2012 by BriGuyTM90 because: (no reason given)
edit on 30-8-2012 by BriGuyTM90 because:
(no reason given)
oh well nvm I was wrong guess you can
edit on 30-8-2012 by BriGuyTM90 because: (no reason given)
new topics
-
That which the "news" never talks about; Truth about election fraud
Mainstream News: 29 minutes ago -
Biden doesnt want the votes of "Death to America" chanters
US Political Madness: 36 minutes ago -
Horizon Post office scandal
Regional Politics: 1 hours ago -
Joe Biden and Donald Trump are both traitors
US Political Madness: 3 hours ago -
I'm new here. Avid conspiracy fan.
Introductions: 3 hours ago -
Denmark's Notre-Dame moment - 17th Century Borsen goes up in Flames
Mainstream News: 4 hours ago
top topics
-
Suspected Iranian agent working for Pentagon while U.S. coordinated defense of Israel
US Political Madness: 12 hours ago, 16 flags -
The Baloney aka BS Detection Kit
Social Issues and Civil Unrest: 12 hours ago, 7 flags -
That which the "news" never talks about; Truth about election fraud
Mainstream News: 29 minutes ago, 5 flags -
Denmark's Notre-Dame moment - 17th Century Borsen goes up in Flames
Mainstream News: 4 hours ago, 4 flags -
How does my computer know
Education and Media: 15 hours ago, 3 flags -
I'm new here. Avid conspiracy fan.
Introductions: 3 hours ago, 3 flags -
Joe Biden and Donald Trump are both traitors
US Political Madness: 3 hours ago, 2 flags -
Horizon Post office scandal
Regional Politics: 1 hours ago, 1 flags -
Biden doesnt want the votes of "Death to America" chanters
US Political Madness: 36 minutes ago, 0 flags
active topics
-
It has begun... Iran begins attack on Israel, launches tons of drones towards the country
World War Three • 748 • : Oldcarpy2 -
SHORT STORY WRITERS CONTEST -- April 2024 -- TIME -- TIME2024
Short Stories • 18 • : argentus -
Candidate TRUMP Now Has Crazy Judge JUAN MERCHAN After Him - The Stormy Daniels Hush-Money Case.
Political Conspiracies • 240 • : Threadbarer -
Abortions in first 12 weeks should be legalised in Germany, commission says
Medical Issues & Conspiracies • 18 • : Consvoli -
That which the "news" never talks about; Truth about election fraud
Mainstream News • 1 • : watchitburn -
Joe Biden and Donald Trump are both traitors
US Political Madness • 34 • : andy06shake -
Biden doesnt want the votes of "Death to America" chanters
US Political Madness • 1 • : Disgusted123 -
How does my computer know
Education and Media • 18 • : MrGashler -
Truth Social goes public, be careful not to lose your money
Mainstream News • 114 • : matafuchs -
NYAG Letitia James Prepares to Begin Seizing TRUMP Assets Even Though There Was No Crime.
Above Politics • 285 • : matafuchs
