It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
10
share:
Yesterday my wife was complaining about a bad reaction after drinking "red cap" milk, it's not the first time I must say. So I finally got tired of it
and decide to share my thoughts here and try to investigate it at the same time. Let's see what we got here.
Even if it's been discussed a few times, it doesn't seem to be clear and no further conclusion has been reached, maybe because there are too many factors to consider.
Before we go deeper, we must make understandable for members in other countries our milk classification (USA), since we are not sure if all other countries follow the same standards. Probably it's where all the polemic starts.
CLASSIFICATION AND GRADING OF MILK.
Everything starts here. Originally, milk pricing was defined by the volume. But raw milk is not uniform and the amount of butterfat and other elements varies constantly. For example, in summer cows produce less milk and also their diet is different than winter lowering the butterfat and protein in the milk.
www.ers.usda.gov...
In other words, establishing standards for milk permited to improve the commercial benefits of the milk. Extracting excess of butterfat in order to acomplish these standards, expanded the business with other products to offer, like heavy cream and skim milk. A gold mine was discovered.

www.ncbi.nlm.nih.gov...
Then we also had milk powder, an incredible way to storage milk for longer periods of time which allowed shipping to further territories.
www.wisebread.com...
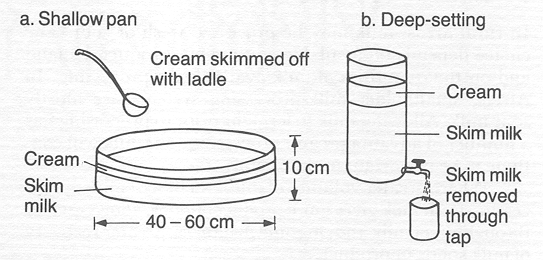
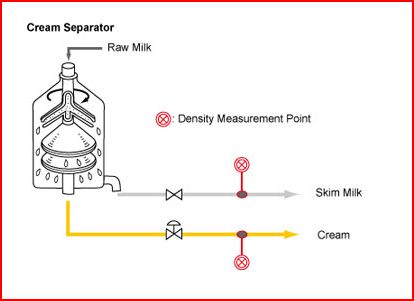
To impove efficience skiming milk, technology started to develope methods based in cetrifuge force, instead of let the milk separates by natural process.
www.ilri.org...
www.yokogawa.com...
At this point we can confirm without a doubt, the milk that people used to drink and used for other products like butter or cheese long time ago was richer in its elements, it wasn't altered as it is now. The milk on the bucked was the final product, as the milk we all got from mama when we were babies.
Now we have a cap color system that tells us what type of milk is supose to be inside the bottle.

www.wisebread.com...
Well, I think that covers well that point, now it's time to talk about hormones. I promise it will be controversial.
HORMONES AND MILK
I personally believe that there is nothing we need to add to milk, Mother Nature already did all and her designs are always perfect. Anyway It's also neccessary to consider other points of view. Like this one from Canada:
www.healthunit.org...
Once again, it's all about money and we were considered as collateral damage. Pay attention to the word "UNNATURALLY" here.
Also, if it's not good for the cows, it's not good for us neither right ?
www.godairyfree.org...
Remenber, it's not me saying all this :
Continue next post....
Even if it's been discussed a few times, it doesn't seem to be clear and no further conclusion has been reached, maybe because there are too many factors to consider.
Before we go deeper, we must make understandable for members in other countries our milk classification (USA), since we are not sure if all other countries follow the same standards. Probably it's where all the polemic starts.
CLASSIFICATION AND GRADING OF MILK.
Everything starts here. Originally, milk pricing was defined by the volume. But raw milk is not uniform and the amount of butterfat and other elements varies constantly. For example, in summer cows produce less milk and also their diet is different than winter lowering the butterfat and protein in the milk.
Milk was priced, like most liquids, on the basis of simple volume until the very late 19th century.
However, anyone making dairy products knew that milk was far from uniform. The composition of milk
varied by producer, individual cow, stage of lactation, season, and other less explainable factors. Even
today, some milk can easily contain twice as much fat as some other milk. The Babcock test, developed in
1890, was a simple, fast, and accurate way of measuring the milkfat content of milk. It fairly directly
established the amount of cream or butter that could be produced from milk and also provided a fairly
accurate means of predicting cheese yields. The Babcock test revolutionized milk pricing and was rapidly
adopted by plants buying milk.
As the commercial dairy industry developed, measurement of the quantity of milk shifted from a volume
basis to weight because accurate determination of weight was generally easier. The use of the Babcock
test and the change to weighing milk led to the milk pricing method that would dominate throughout
most of the 20th century: dollars per 100 pounds, adjusted for milkfat content with a butterfat differential.
Another pricing method, dollars per pound of milkfat has been used for cream and was sometimes
used for milk. It never was a satisfactory pricing method for many uses of milk and fell entirely out of
favor as skim solids became more valuable.
www.ers.usda.gov...
In other words, establishing standards for milk permited to improve the commercial benefits of the milk. Extracting excess of butterfat in order to acomplish these standards, expanded the business with other products to offer, like heavy cream and skim milk. A gold mine was discovered.

Articles from American Journal of Public Health (New York, N.Y. : 1912) are provided here courtesy of
www.ncbi.nlm.nih.gov...
Then we also had milk powder, an incredible way to storage milk for longer periods of time which allowed shipping to further territories.
www.wisebread.com...
To impove efficience skiming milk, technology started to develope methods based in cetrifuge force, instead of let the milk separates by natural process.

www.ilri.org...

www.yokogawa.com...
At this point we can confirm without a doubt, the milk that people used to drink and used for other products like butter or cheese long time ago was richer in its elements, it wasn't altered as it is now. The milk on the bucked was the final product, as the milk we all got from mama when we were babies.
Now we have a cap color system that tells us what type of milk is supose to be inside the bottle.

Red Cap: Whole Milk
Purple Cap: Reduced-Fat (2%) Milk
Green: Low-Fat (1%) Milk
Light Blue: Skim Milk
Brown Cap: Chocolate Milk
Pink Cap: Strawberry Milk
Yellow Cap: Buttermilk
Orange Cap: Half and Half
Magenta Cap: Whipping Cream
www.wisebread.com...
Well, I think that covers well that point, now it's time to talk about hormones. I promise it will be controversial.
HORMONES AND MILK
I personally believe that there is nothing we need to add to milk, Mother Nature already did all and her designs are always perfect. Anyway It's also neccessary to consider other points of view. Like this one from Canada:
Myth – Milk contains hormones that are harmful to humans
Fact – Canada does not allow the use of growth hormones to increase milk production in
cows. Therefore, no added growth hormones are found in any Canadian milk or milk
products. There are also no antibiotic residues in any Canadian milk or milk products.
Natural hormones from the cow do pass into the milk - however they have no effect on
humans when consumed and are digested like any other protein. Bovine Growth
Hormone (BGH), also known as recombinant Bovine Somatotropin (rBST), is currently
used in the United States to increase milk production in cows. rBST is not present in
milk that is produced in Canada.
www.healthunit.org...
Once again, it's all about money and we were considered as collateral damage. Pay attention to the word "UNNATURALLY" here.
The FDA approved the use of rBGH, a synthetic version of BGH, in 1993. The injection of rBGH into cows has become standard practice on many dairy farms, as it has the ability to unnaturally increase a cow’s output of milk by up to 20% (according to the rBGH manufacturer).
Also, if it's not good for the cows, it's not good for us neither right ?
Cows treated with rBGH were found to have a 25% increased risk of acquiring an udder infection (mastitis). Other major side effects (as noted by the manufacturer of rBGH) include infertility, lameness, cystic ovaries, uterine disorders, digestive disorders, lacerations, and calluses of the knee
www.godairyfree.org...
Remenber, it's not me saying all this :
Even more seriously, rBGH milk is contaminated with high levels of the natural Insulin-like Growth Factor-1 (IGF-1), which regulates cell growth, division and multiplication throughout life, particularly in infants and young children; Eli Lilly, in its application for registration of rBGH, admitted that IGF-1 blood levels of injected cows are increased up to ten-fold. IGF-1 is resistant to pasteurization and digestion, and is readily absorbed from the small intestine. Monsanto's own data revealed that feeding IGF-1 to adult rats for only two weeks significantly increased body and liver weights, and bone length.
More critically, increased IGF-1 blood levels have been incriminated as a major cause of cancer. IGF-1 induces uncontrolled growth of normal human breast cells in tissue culture, and has been incriminated in their transformation to cancer cells.
Continue next post....
edit on 24-3-2012 by Trueman because: (no reason given)
Who puts this crap in the milk ?
www.organicconsumers.org...
We cartainly have no way to personally confirm all this by a scientific method but somehow is hard to trust those who deny it.
www.washingtonpost.com...
If you don’t believe the scientists and activists, listen to the kid :
ANTIBIOTICS AND MILK
Now, this is serious. Everybody knows how dangerous is the excessive use of antibiotics Let’s analyze both sides. This gentleman explains in short terms how the usage of antibiotics in milk should be used, nothing wrong with that :
This farmer makes you feel safe and I wish all farmers do the same way. Unfortunatelly, even if the farmer follow all procedures, there is always something that can go wrong.
A real milk conspiracy, with all the evidence you want. Undebunkable.
www.nj.com...
It’s important to understand what the presence of antibiotic in milk really means. First of all, the term antibiotic itself in too generic. There are an specific class of them that we must pay attention to:
en.wikipedia.org...
Now we can have another conclusion here. Antibiotics in milk can be SERIOUSLY dangerous if not used in the right way and CONTROLLED properly. Otherwise, this long procedure wouldn’t have a reason to be :
www.in.gov...
That’s good for now folks.
Revealingly, Hudson uses the term rBST, recombinant Bovine Somatotropin, avoiding any reference to the word "Hormone" in Monsanto's original acronym rBGH.
www.organicconsumers.org...
We cartainly have no way to personally confirm all this by a scientific method but somehow is hard to trust those who deny it.
In 2009, President Obama appointed Michael Taylor as a senior adviser for the FDA. Consumer groups protested the appointment because Taylor had formerly served as a vice president for Monsanto, the controversial agricultural multinational at the forefront of genetically modified food
www.washingtonpost.com...
If you don’t believe the scientists and activists, listen to the kid :
ANTIBIOTICS AND MILK
Now, this is serious. Everybody knows how dangerous is the excessive use of antibiotics Let’s analyze both sides. This gentleman explains in short terms how the usage of antibiotics in milk should be used, nothing wrong with that :
This farmer makes you feel safe and I wish all farmers do the same way. Unfortunatelly, even if the farmer follow all procedures, there is always something that can go wrong.
N.J. company charged with using tainted milk in its ricotta cheese
The company paid cash for the discounted milk from D.A. Landis Trucking Inc. of Lancaster, although dairy farmers had pledged to dispose of it, prosecutors said. Lebanon sold the ricotta cheese to restaurants, delis, ravioli manufacturers and others.
A real milk conspiracy, with all the evidence you want. Undebunkable.
Landis Trucking and President Dean Landis have agreed to plead guilty Friday in Allentown to conspiracy to falsify driver log books.
www.nj.com...
It’s important to understand what the presence of antibiotic in milk really means. First of all, the term antibiotic itself in too generic. There are an specific class of them that we must pay attention to:
β-Lactam antibiotics (beta-lactam antibiotics) are a broad class of antibiotics, consisting of all antibiotic agents that contains a β-lactam nucleus in their molecular structures. This includes penicillin derivatives (penams), cephalosporins (cephems), monobactams, and carbapenems.[1] Most β-lactam antibiotics work by inhibiting cell wall biosynthesis in the bacterial organism and are the most widely used group of antibiotics. Up until 2003, when measured by sales, more than half of all commercially available antibiotics in use were β-lactam compounds.[2]
Adverse drug reactions
Common adverse drug reactions (ADRs) for the β-lactam antibiotics include diarrhea, nausea, rash, urticaria, superinfection (including candidiasis).[3]
Infrequent ADRs include fever, vomiting, erythema, dermatitis, angioedema, pseudomembranous colitis.[3]
Pain and inflammation at the injection site is also common for parenterally administered β-lactam antibiotics.
Allergy/hypersensitivity
Immunologically mediated adverse reactions to any β-lactam antibiotic may occur in up to 10% of patients receiving that agent (a small fraction of which are truly IgE-mediated allergic reactions, see amoxicillin rash). Anaphylaxis will occur in approximately 0.01% of patients.[3][4] There is perhaps a 5%-10% cross-sensitivity between penicillin-derivatives, cephalosporins, and carbapenems; but this figure has been challenged by various investigators.
Nevertheless, the risk of cross-reactivity is sufficient to warrant the contraindication of all β-lactam antibiotics in patients with a history of severe allergic reactions (urticaria, anaphylaxis, interstitial nephritis) to any β-lactam antibiotic.
A Jarisch-Herxheimer reaction may occur after initial treatment of a spirochetal infection such as syphilis with a β-lactam antibiotic.
en.wikipedia.org...
Now we can have another conclusion here. Antibiotics in milk can be SERIOUSLY dangerous if not used in the right way and CONTROLLED properly. Otherwise, this long procedure wouldn’t have a reason to be :
SCREENING TESTS NECESSARY TO IMPLEMENT THE PROVISIONS OF
APPENDIX N. FOR BULK MILK PICKUP TANKERS:
1. Performance Tests/Controls (+/-):
a. Each lot of kits purchased is tested by positive (+) and negative (-) controls.
b. Each screening facility runs a positive (+) and negative (-) control performance test each
testing day.
c. All NCIMS Approved Bulk Milk Pickup Tanker Screening Tests Include The Following
Format: All presumptive positive test results are to be repeated in duplicate as soon as
possible at the direction of the Regulatory Agency on the same sample with single positive
(+) and negative (-) controls by a certified analyst (Official State Laboratory, Officially
Designated Laboratory or Certified Industry Supervisor) using the same or equivalent test
(M-I-96-10, latest revision). If the duplicate tests, with appropriate control (+/-) results are
negative (-), the tanker is reported as negative. If one or both duplicate test(s) is positive (+),
the test result is reported to the Regulatory Agency of the State in which the testing was
conducted, as a screening positive.
d. All positive (+) controls used for drug residue testing kits are labeled to indicate a
specific drug and concentration level for that drug.
(1) For tests that only detect Penicillin, Ampicillin, Amoxicillin and Cephapirin, the
positive (+) control is Pen G @ 5 ± 0.5 ppb.
(2) For test kits validated for the detection of Cloxacillin, the positive (+) control may be
Cloxacillin @ 10 ± 1 ppb.
(3) For test kits validated for one (1) drug residue only, the positive (+) control is ± 10%
of the safe level/tolerance of the drug residue detected.
2. Work Area:
a. Temperature within specifications of the test kit manufacturer's labeling.
b. Adequate lighting for test kit procedure.
3. Test Kit Thermometers:
a. Thermometer traceable to a NIST Certified Thermometer.
b. Graduation interval not greater than 1°C.
c. Dial thermometers are not used to determine temperatures of samples, reagents,
refrigerators, or incubators in milk laboratories.
4. Refrigeration:
a. Test kit reagent storage temperature specified by manufacturer.
5. Balance (Electronic):
335
a. 0.01 g for preparation of positive (+) controls.
b. Balance with appropriate sensitivity for calibration of pipetting devices within a tolerance
of ± 5%. These devices may be calibrated at another location acceptable to the State LEO.
6. Screening Test Sampling Requirements:
a. Temperature of milk in the bulk milk pickup tanker determined and recorded.
b. Representative bulk milk pickup tanker sample for drug residue testing collected.
c. Samples tested within seventy-two (72) hours of collection.
7. Screening Test Volumetric Measuring Devices:
a. Single use devices provided by kit manufacturers are acceptable for Appendix N.
screening analysts.
b. NCIMS Certified Laboratories require calibrated pipetting/dispensing devices. These
devices may be calibrated at another location acceptable to the State LEO.
c. Measuring devices with tips bearing calibration lines provided by test kit manufacturers
are acceptable for Appendix N. screening.
IV. ESTABLISHED TOLERANCES AND/OR SAFE LEVELS OF DRUG RESIDUES
"Safe levels" are used by FDA as guides for prosecutorial discretion. They do not legalize
residues found in milk that are below the safe level. In short, FDA uses the "safe levels" as
prosecutional guidelines and in full consistency with CNI v. Young stating, in direct and
unequivocal language, that the "safe levels" are not binding. They do not dictate any result; they
do not limit the Agency's discretion in any way; and they do not protect milk producers, or milk
from court enforcement action.
"Safe levels" are not and cannot be transformed into tolerances that are established for animal
drugs under Section 512 (b) of the FFD&CA as amended . "Safe levels" do not:
1. Bind the courts, the public, including milk producers, or the Agency, including individual
FDA employees; and
2. Do not have the "force of law" of tolerances, or of binding rules.
Notification, changes or additions of "safe levels" will be transmitted via Memoranda of
Information (M-I's).
www.in.gov...
That’s good for now folks.
edit on 24-3-2012 by Trueman because: (no reason given)
While I have just read enough of this thread to get a summary (which is well put together, if I may say) I wanted to add something that bewildered
me.
I have a cat (approximately 9 months old) and while I know it's not good to give cats cow's milk per se I decided to give him a little saucer of it one night as a treat.
When I was a kid we did this pretty often as cats (any cat that my family had that is) always LOVED milk.
However, when I tried to give my cat milk (red cap, slightly warmed in the microwave) around 2 weeks ago, he smelled it and made a face likened to what he would have made had I gave him a lemon instead. I was perplexed and thought well maybe he just doesn't like milk, OR they're putting so much crap in it now that the cat is smart enough to steer clear.
Additionally, there is a wondering cat that comes around occasionally so just to settle my own conspiratorial notions I decided to try and give him/her a saucer of the same brand of milk, lo' and behold, it had the same reaction to the milk.
There in, I feel that posting this thread is spot on!
I have a cat (approximately 9 months old) and while I know it's not good to give cats cow's milk per se I decided to give him a little saucer of it one night as a treat.
When I was a kid we did this pretty often as cats (any cat that my family had that is) always LOVED milk.
However, when I tried to give my cat milk (red cap, slightly warmed in the microwave) around 2 weeks ago, he smelled it and made a face likened to what he would have made had I gave him a lemon instead. I was perplexed and thought well maybe he just doesn't like milk, OR they're putting so much crap in it now that the cat is smart enough to steer clear.
Additionally, there is a wondering cat that comes around occasionally so just to settle my own conspiratorial notions I decided to try and give him/her a saucer of the same brand of milk, lo' and behold, it had the same reaction to the milk.
There in, I feel that posting this thread is spot on!
edit on 3/24/2012 by UberL33t because: (no reason given)
reply to post by UberL33t
Thank you. I liked that story about your cat, I think he is wise enough to follow his instincts.
Thank you. I liked that story about your cat, I think he is wise enough to follow his instincts.
When I switched to Organic Milk and all Dairy Products years ago I stopped getting all stuffed up and not feeling up to par most of the time.
The Milk producers and FDA are trying to tell us that it isn't healthy to drink organic milk, makes you wonder why.. I know why, they don't want the competition and obviously don't want us healthy. There is a reason organic products are more expensive, it costs more to raise and produce them.
Opening poster switch to organic milk and see what happens with your wife.
The Milk producers and FDA are trying to tell us that it isn't healthy to drink organic milk, makes you wonder why.. I know why, they don't want the competition and obviously don't want us healthy. There is a reason organic products are more expensive, it costs more to raise and produce them.
Opening poster switch to organic milk and see what happens with your wife.
Good information on the dangers of milk and what is used in it.
The video with the 11 year old kid was good too... very simply explained.
There is so much poison in our foods... I wonder who owns the farms that the elites get their food from. The ones where celebrities pay nutritionists and personal chefs to get their food from. Would be interesting to find out.
~Namaste
ETA: I switched to organic milk a while ago... tastes much better and I don't ever get stomach aches after drinking it or eating things made with it.
The video with the 11 year old kid was good too... very simply explained.
There is so much poison in our foods... I wonder who owns the farms that the elites get their food from. The ones where celebrities pay nutritionists and personal chefs to get their food from. Would be interesting to find out.
~Namaste
ETA: I switched to organic milk a while ago... tastes much better and I don't ever get stomach aches after drinking it or eating things made with it.
edit on 24-3-2012 by SonOfTheLawOfOne because: (no reason given)
Originally posted by Aquarius1
.. I know why, they don't want the competition and obviously don't want us healthy.
That phrase resumes my 2 pages thread initial post.
She has been using Lactaid (blue), but that day we only had the regular red cap in the fridge. Now she knows.
reply to post by SonOfTheLawOfOne
Thanks. I use red cap milk in my early morning coffee, makes me go bathroom when I feel little constipated. Makes me run
Thanks. I use red cap milk in my early morning coffee, makes me go bathroom when I feel little constipated. Makes me run
I own a dairy and am proud to have an organic operation.
I have to undergo 2 inspections each hear - they search my records, take samples of the soil, hay and feed I use. It’s a rather invasive process to be honest.
I have never given a cow a hormone and don't intend to. I have used antibiotics but if I do I am required to take the cow off the line during and for six weeks after the incident. I do everything I can to not have to run an antibiotic because I lose a lot of production from it. I have to keep meticulous records of all vet bills, visits and such.
I also have a heard of dairy goats - the industry standards for that are a little different. I would classify them as not as stringent.
I do not pasteurize milk here at the farm - I sell in bulk to a tanker who picks it up three times a week. However, I know it goes to "organic" diary processors because they pay the higher price.
I personally have consumed raw milk and raw milk products my whole life - I have never gotten sick from drinking raw unpasteurized milk from either a goat or a cow.
I probably drink upwards of 32 oz of whole raw milk a day - bovine and caprine.
My dairy cow herd averages between 7-10 percent milk fat in the raw form while the goats average 30-50% depending.
If you want good milk find a dairy operation in your area and get it fresh from the source and unpasteurized.
I sell to people all the time but I have to put "for animal use only" on the containers to protect myself from the law. It's dumb I have to do so but it's not a huge issue I just print off labels each week for them.
I don't care what they do with it when they get it home .
Great thread and good accurate research on the industry. Factory farming is the devil IMO the animals are not treated right and the product is #!
I have to undergo 2 inspections each hear - they search my records, take samples of the soil, hay and feed I use. It’s a rather invasive process to be honest.
I have never given a cow a hormone and don't intend to. I have used antibiotics but if I do I am required to take the cow off the line during and for six weeks after the incident. I do everything I can to not have to run an antibiotic because I lose a lot of production from it. I have to keep meticulous records of all vet bills, visits and such.
I also have a heard of dairy goats - the industry standards for that are a little different. I would classify them as not as stringent.
I do not pasteurize milk here at the farm - I sell in bulk to a tanker who picks it up three times a week. However, I know it goes to "organic" diary processors because they pay the higher price.
I personally have consumed raw milk and raw milk products my whole life - I have never gotten sick from drinking raw unpasteurized milk from either a goat or a cow.
I probably drink upwards of 32 oz of whole raw milk a day - bovine and caprine.
My dairy cow herd averages between 7-10 percent milk fat in the raw form while the goats average 30-50% depending.
If you want good milk find a dairy operation in your area and get it fresh from the source and unpasteurized.
I sell to people all the time but I have to put "for animal use only" on the containers to protect myself from the law. It's dumb I have to do so but it's not a huge issue I just print off labels each week for them.
I don't care what they do with it when they get it home .
Great thread and good accurate research on the industry. Factory farming is the devil IMO the animals are not treated right and the product is #!
reply to post by Golf66
Excellent ! The point of view of an organic milk farmer, what an honor for me, thank you. I also work with milk but unfortunatelly not organic.
I remember years ago, the first time I've heard the term "Organic", I had no clue what that means.
Later I found out "Organic" means the natural "Regular" way it used to be.
"Regular" was "Organic.
Excellent ! The point of view of an organic milk farmer, what an honor for me, thank you. I also work with milk but unfortunatelly not organic.
I remember years ago, the first time I've heard the term "Organic", I had no clue what that means.
Later I found out "Organic" means the natural "Regular" way it used to be.
"Regular" was "Organic.
Originally posted by Golf66
I personally have consumed raw milk and raw milk products my whole life - I have never gotten sick from drinking raw unpasteurized milk from either a goat or a cow.
Glad to hear you raise organic cows and produce organic milk, it all I use, happen to buy Horizon..
I have also drank raw milk from time to time but no longer live near a place where it's sold.. Sadly I have read that in some states the sale of raw milk is banned.
I have a question, why is that Organic Milk has a much longer shelf life then the other stuff?
OMG, this thread is so informative. I am deathly allergic to penicillin and have avoided milk in large quantities for years due to it giving me
"hives" same as the penicillin did when I was young.
You seriously hit the nail on the head with this thread. I'm not allergic to milk as I thought, it's the crap in it causing the reactions.
A_L
You seriously hit the nail on the head with this thread. I'm not allergic to milk as I thought, it's the crap in it causing the reactions.
A_L
Originally posted by Aquarius1
I have also drank raw milk from time to time but no longer live near a place where it's sold.. Sadly I have read that in some states the sale of raw milk is banned.
I have a question, why is that Organic Milk has a much longer shelf life then the other stuff?
In MO you can't sell raw milk to people for human consumption - that's why I have to put a label in the milk that I know the people I sell it to will ignore. It’s a stupid game we all play... I can sell it for "animal consumption only". As long as the buyer and I don't talk about it we are all hunky dory.
Organic and raw are two different things - raw milk is not pasteurized. However, organic milk often has to travel farther and longer to market than regular milk and therefore it is usually - ultra pasteurized.
It’s simply heated to a higher temperature say upward of 200°F instead of the typical 161° which destroys more of the organisms responsible for spoiling milk. With those bugs knocked out, organic milk lasts 25 to 40 days longer than the ordinary stuff.
The raw milk I use for my family never lasts long we seem to drink a lot. I love to have an 8oz glass of warm milk with a large dollop or spoonful of honey and a dash of cinnamon before bed. Nice treat.
Originally posted by another_lurker
OMG, this thread is so informative. I am deathly allergic to penicillin and have avoided milk in large quantities for years due to it giving me "hives" same as the penicillin did when I was young.
You seriously hit the nail on the head with this thread. I'm not allergic to milk as I thought, it's the crap in it causing the reactions.
A_L
I think you should define what kind of milk is the right one for you. Remember allergic reactions to milk are related to casein, which is the protein in the milk. This is different from lactose intolerance.
Anyway, in your case, the antibiotics seem to be the problem since you mention your allergy to penicillin, But I'm not an expert and I can be wrong.
Definitelly, the best option is always organic milk. If you still have bad reactions, then lactose should be the problem. Again, I can be wrong.
Consult your doctor before take a decision, ALWAYS.
en.wikipedia.org...
www.mayoclinic.com...=causes
edit on 24-3-2012 by Trueman because: (no reason given)
Here is something to add to this thread. Milk fat may be good for you. Especially milk that is not fed lots of hormones and is fed grains high in
pesticides and herbicides. www.medicalnewstoday.com...
originally posted by: rickymouse
Here is something to add to this thread. Milk fat may be good for you. Especially milk that is not fed lots of hormones and is fed grains high in pesticides and herbicides. www.medicalnewstoday.com...
When they do the process of creating the other types of milk by removing the milkfats, they are taking out most of what makes milk good and simply leaving in most of the sugars.
If you are going to drink milk, then you should go with the fat content.
I wonder how we can get so wrapped up in adding hormones to our feed unnaturally and yet be so certain it's a good thing to pump our kids full of unnatural hormones in the name of treating them.
originally posted by: ketsuko
originally posted by: rickymouse
Here is something to add to this thread. Milk fat may be good for you. Especially milk that is not fed lots of hormones and is fed grains high in pesticides and herbicides. www.medicalnewstoday.com...
When they do the process of creating the other types of milk by removing the milkfats, they are taking out most of what makes milk good and simply leaving in most of the sugars.
If you are going to drink milk, then you should go with the fat content.
I wonder how we can get so wrapped up in adding hormones to our feed unnaturally and yet be so certain it's a good thing to pump our kids full of unnatural hormones in the name of treating them.
Saying fat from milk is bad for you is Fad nutrition. Fad Nutrition is based on taking one property of food being good or bad and running with it. People try to make themselves prestigious with ill gotten or misinterpreted evidence. Others just parrot the fake information. The news agencies are not the only group spurning crap in this world.
I hardly ever get anything other than whole milk, I convinced the wife years ago to stop getting two percent because it does not taste right. Skim milk and one percent were used to feed pigs years back, I pittied the pigs for having to drink that stuff. We have been using real butter for over twenty five years, it tastes so much better. There was only a window of time of about ten years where I actually ate margarine, and that was Fletchmans which seemed much better than the others, but was way more expensive than other Oleos.
edit on 17-7-2018 by rickymouse because:
(no reason given)
new topics
-
Any one suspicious of fever promotions events, major investor Goldman Sachs card only.
The Gray Area: 1 hours ago -
God's Righteousness is Greater than Our Wrath
Religion, Faith, And Theology: 6 hours ago -
Electrical tricks for saving money
Education and Media: 9 hours ago -
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News: 10 hours ago -
Sunak spinning the sickness figures
Other Current Events: 11 hours ago -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues: 11 hours ago
top topics
-
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News: 10 hours ago, 9 flags -
Cats Used as Live Bait to Train Ferocious Pitbulls in Illegal NYC Dogfighting
Social Issues and Civil Unrest: 14 hours ago, 8 flags -
Electrical tricks for saving money
Education and Media: 9 hours ago, 4 flags -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues: 11 hours ago, 3 flags -
Sunak spinning the sickness figures
Other Current Events: 11 hours ago, 3 flags -
Late Night with the Devil - a really good unusual modern horror film.
Movies: 13 hours ago, 2 flags -
Any one suspicious of fever promotions events, major investor Goldman Sachs card only.
The Gray Area: 1 hours ago, 2 flags -
The Good News According to Jesus - Episode 1
Religion, Faith, And Theology: 16 hours ago, 1 flags -
God's Righteousness is Greater than Our Wrath
Religion, Faith, And Theology: 6 hours ago, 0 flags
active topics
-
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues • 21 • : FlyersFan -
God's Righteousness is Greater than Our Wrath
Religion, Faith, And Theology • 13 • : andy06shake -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 662 • : F2d5thCavv2 -
SETI chief says US has no evidence for alien technology. 'And we never have'
Aliens and UFOs • 61 • : andy06shake -
The Reality of the Laser
Military Projects • 47 • : F2d5thCavv2 -
Definitive 9.11 Pentagon EVIDENCE.
9/11 Conspiracies • 423 • : Lazy88 -
Election Year 2024 - Interesting Election-Related Tidbits as They Happen.
2024 Elections • 73 • : Threadbarer -
Sunak spinning the sickness figures
Other Current Events • 8 • : McUrnsalso -
HORRIBLE !! Russian Soldier Drinking Own Urine To Survive In Battle
World War Three • 36 • : TheMisguidedAngel -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 146 • : Consvoli
10
