It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
USA / Europe - Radiation and Jetstream FORECAST UPDATE - march 27, 2011
Received on the 29 of March 2011
Don't look good, if this is true, already exposed.
Received on the 29 of March 2011
Don't look good, if this is true, already exposed.
Originally posted by 1beerplease
any effect to the southern hemisphere?
Jeffrey Stehr, an atmospheric research scientist at the University of Maryland, said that while the radiation from Japan has been widely detected, it could take as much as a year to spread throughout the Northern Hemisphere. It could take another year before it is widespread in the Southern Hemisphere because of blocking at the equator caused by rising air currents where winds from north and south collide.
Source: Associated press
reply to post by elevenaugust
So the areas around the equators would be the best place in an extreme worst case?
Hmm.
So the areas around the equators would be the best place in an extreme worst case?
Hmm.
Updates
Observations and simulation Japan map of the I131 diffusion from March 29th 00h UTC to March 31st 00UTC avalaible here
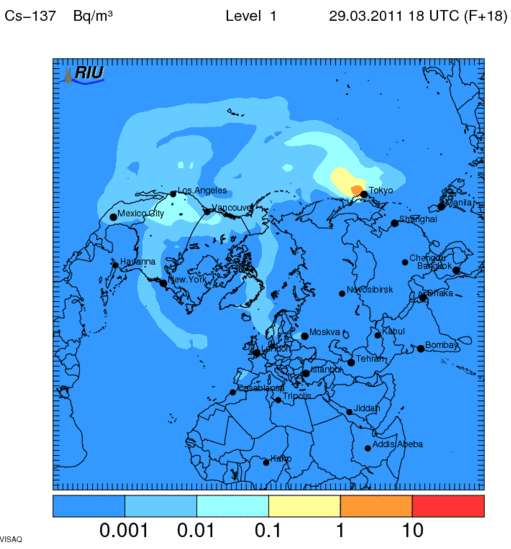
Observations and simulation worldwide of the Cs137 ground diffusion March 29th 18h UTC:
Composition of the estimated extend Cs137 cloud worldwide between March 30th and 31st:

I'll try to do an update everyday of this cloud extension
Observations and simulation Japan map of the I131 diffusion from March 29th 00h UTC to March 31st 00UTC avalaible here
Observations and simulation worldwide of the Cs137 ground diffusion March 29th 18h UTC:

Composition of the estimated extend Cs137 cloud worldwide between March 30th and 31st:

I'll try to do an update everyday of this cloud extension
edit on 29-3-2011 by elevenaugust because: (no reason given)
edit on 29-3-2011 by elevenaugust because: (no reason
given)
edit on 29-3-2011 by elevenaugust because: trying desesperatly to host a gif....
reply to post by markymint
HAHAHA!!! A proper ATS moment. I made that comment about it reaching Scotland and the next day the BBC report it in SCOTLAND and my home county!!! (Oxfordshire) ... bloody alienz and conspirasars and shtuff!
Anyhoo - keep up these images. They appear much more informative than just about anything in the media. I think my previous encounter proves these images have told me well in advance of any news report just where the radiation is. I urge others to keep an eye on this thread!
HAHAHA!!! A proper ATS moment. I made that comment about it reaching Scotland and the next day the BBC report it in SCOTLAND and my home county!!! (Oxfordshire) ... bloody alienz and conspirasars and shtuff!
Anyhoo - keep up these images. They appear much more informative than just about anything in the media. I think my previous encounter proves these images have told me well in advance of any news report just where the radiation is. I urge others to keep an eye on this thread!
edit on
29-3-2011 by markymint because: (no reason given)
Originally posted by eagledriver
It's quite simply that they do not want to start a panic. In the US, the slightiest amount of bad news mushrooms out of control, due to the Internet and mobile networks.
That's why everyone in the media, including Obama, downplays everything.edit on 28-3-2011 by eagledriver because: (no reason given)
Rule #1 Deny
Rule #2 Don't scare the sheeple
Rule #3 Deny some more.
Rule #4 Scare the sheeple just enough to keep them under control.
Rule #5 Deny yet again.
reply to post by elevenaugust
how to read this? the blue lines and blobs are readings of higher concentrations of radioactive particles in the air?
or the ground? is this from satellite is this from topographical, on site analysis?
sorry im not sure how to read this graph.
how to read this? the blue lines and blobs are readings of higher concentrations of radioactive particles in the air?
or the ground? is this from satellite is this from topographical, on site analysis?
sorry im not sure how to read this graph.
The question I ask is how much effect did the 3 mile island have on Europe. I realize this is probably worse than the 3 mile island, but just
wondering.
Well, the radiation level just hit 105 cpm AT NIGHT in Denver, CO!
That ain't good...
www.abovetopsecret.com...
That ain't good...
www.abovetopsecret.com...
reply to post by YourPopRock
ok I do a lot of research online for my job so I got the home page of the website itself figured out..
www.zamg.ac.at...
that is a legit site they posted. it is in Vienna very interesting info...
thanks for sending that to us!
ok I do a lot of research online for my job so I got the home page of the website itself figured out..
www.zamg.ac.at...
that is a legit site they posted. it is in Vienna very interesting info...
thanks for sending that to us!
Originally posted by mutantgenius
Radiation network is showing high readings (above 130) over alabama rught now and since at least 830 am est (first time I checked today) Just thought everyone should know.
Here is the link
I'm showing NO (zero, none) high readings or even alerts on that map as of 3-29-11 @2126hrs (9:26pm)PST So, not sure if it was a glitch or what..
UPDATE: (from the site linked above)
Update: 3/29/11, 6:15 A.M. Radiation Monitoring by Government vs. Private Citizen
So what can we take away from yesterday's incident of the false alert? It seems to me that we must conclude that there are tradeoffs in the ways that we monitor radiation. Government operation of monitoring stations is under tight and direct control, but at the same time leaves open the question of transparency when it comes to the Government's understandable position of walking a fine line between keeping the public informed while averting panic.
Private citizen radiation monitoring networks like ours, on the other hand, are partly dependent on the integrity of its individual Monitoring Stations, but on the other hand, the reporting of radiation levels is quite transparent. So if we as citizens are to do our own monitoring of radiation, which is a very sensitive subject matter, and then report that data to the public over a network, there are certain guidelines that wisdom dictates we must follow:
~ There still must be some degree of central control. That includes being discriminating in issuing licenses to Monitoring Stations in the first place, to minimize the chance of admission to the network of those bent on mischief.
~ It then follows that the network must retain the ability to remove any Monitoring Stations that abuse their privilege.
~ The network should provide for multiple means of communication between client Monitoring Stations and the Network Server, along with the ability for real time discourse among Monitoring Stations through Chat and the like.
In summary, this is a fledgling Radiation Network, and we are in uncharted territory, so we must continue to learn from experience, and refine the network over time. Dealing with the false alert from yesterday was a small test for us. Generally, I think that we passed the test and took appropriate action to maintain the integrity of the network, while keeping the public informed of the facts.
The other positive take away from the incident is that it demonstrated our Radiation Alert system. Whether you were a participating Monitoring Station or a passive viewer of the National Radiation Map online, from hundreds or even thousands of miles away, you knew within just 1 minute of an elevated radiation condition, along with the location of the alert, and then the actual level of radiation being detected. Think about it - that's pretty amazing!!
edit on 29-3-2011 by Komodo because: (no reason given)
reply to post by elevenaugust
Thanks for the maps OP. star for you
For those who question the maps. They are from the CTBTO . The group was formed by a treaty between countries to monitor nuclear testing around the world. The CTBTO was reluctant to put out the maps and stating they needed a mandate about 10 days ago. It appears they found a way to get the data out to the public. Well looks like Germany is the only reporting country of the CTBTO treaty though. The station monitors are in the pacific and all around the world, these maps are the real deal people pay attention to them closely, because the data is from the CTBTO. ( they ,(CTBTO )have nothing to hide, except the countries that fail to report the data from them. I trust the data and maps way more than the EPA data.
Why in the world is The USA not reporting from the CTBTO? The USA is also a member of the treaty.
I wrote the CTBTO last week. All they said in a reply that what they needed was mandate and could not confirm any findings. Big thanks to Germany.
Now Call your Representatives in the USA and give them hell.
Thanks for the maps OP. star for you
For those who question the maps. They are from the CTBTO . The group was formed by a treaty between countries to monitor nuclear testing around the world. The CTBTO was reluctant to put out the maps and stating they needed a mandate about 10 days ago. It appears they found a way to get the data out to the public. Well looks like Germany is the only reporting country of the CTBTO treaty though. The station monitors are in the pacific and all around the world, these maps are the real deal people pay attention to them closely, because the data is from the CTBTO. ( they ,(CTBTO )have nothing to hide, except the countries that fail to report the data from them. I trust the data and maps way more than the EPA data.
Why in the world is The USA not reporting from the CTBTO? The USA is also a member of the treaty.
I wrote the CTBTO last week. All they said in a reply that what they needed was mandate and could not confirm any findings. Big thanks to Germany.
Now Call your Representatives in the USA and give them hell.
edit on 30-3-2011 by SJE98 because: (no reason given)
OMG this is getting worser and worser. I even saw a bit of that cloud forming over The Netherlands (where i come from). Lets all hope for the best, if
possible.
reply to post by elevenaugust
I hope this is OK I post this here. It is in reference to the radiation in the water. Masaru Emoto whose intuitive logic gives him an idemic Einsteinian philosophy is perhaps the worlds greatest researcher of morphogenetic fields effecting water. If you are unfamiliar with his work please Google him and see some images of experimental results of thoughts effect on water fast frozen into crystals with good and bad intent/feeling/language. If it is a fact that we are so intimately connected in time that our thoughts effect everything, lets be sure and send our regards to the wind and the airs that surround us in time. And of course the water.
Masaru Emoto resides in Japan, was in the quake and has a request of the worlds people.
His request is pasted below. This is for the water, and I suspect that because of the nature of water and crystallization it sets off a mass effect through the ocean, scencient awareness . Now in his words...
"Citizens of Earth: March 31st, 12 NOON (your timezone)". "The Water of Fukushima Nuclear Plant, we are sorry to make you suffer. Please forgive us. We thank you, and we love you." This is all it takes. Please say it aloud or in your mind 3 times as you put your hands together in a prayer position. Please offer your sincere prayer." ---- Dr. Masaru Emoto
emotopeaceproject.blogspot.com...
I hope this is OK I post this here. It is in reference to the radiation in the water. Masaru Emoto whose intuitive logic gives him an idemic Einsteinian philosophy is perhaps the worlds greatest researcher of morphogenetic fields effecting water. If you are unfamiliar with his work please Google him and see some images of experimental results of thoughts effect on water fast frozen into crystals with good and bad intent/feeling/language. If it is a fact that we are so intimately connected in time that our thoughts effect everything, lets be sure and send our regards to the wind and the airs that surround us in time. And of course the water.
Masaru Emoto resides in Japan, was in the quake and has a request of the worlds people.
His request is pasted below. This is for the water, and I suspect that because of the nature of water and crystallization it sets off a mass effect through the ocean, scencient awareness . Now in his words...
"Citizens of Earth: March 31st, 12 NOON (your timezone)". "The Water of Fukushima Nuclear Plant, we are sorry to make you suffer. Please forgive us. We thank you, and we love you." This is all it takes. Please say it aloud or in your mind 3 times as you put your hands together in a prayer position. Please offer your sincere prayer." ---- Dr. Masaru Emoto
emotopeaceproject.blogspot.com...
Originally posted by Ashyr
reply to post by elevenaugust
how to read this? the blue lines and blobs are readings of higher concentrations of radioactive particles in the air?
or the ground? is this from satellite is this from topographical, on site analysis?
sorry im not sure how to read this graph.
Well, firstly, one needs to know the natural proportion of Ce137 radioactivity in the atmosphere, which is around 0.001 to 0.01Bq/m2.
The medium blueish area have an average Cs137 radiation dose between 0.01 and 0.1Bq/m2
The light-blue area have an average Cs137 radiation dose between 0.1 and 1Bq/m2
The yellowish area have an average Cs137 radiation dose between 1 and 10Bq/m2
The orange area have an average Cs137 radiation dose between 10 and 100Bq/m2
The red area have an average Cs137 radiation dose more than 100Bq/m2
Deposition: The dry (and wet) deposition display the total accumulated deposition along the path of the plume, to the ground then.
These maps are created by weather National services, and they used radionucleide ground sensor all around the world as well as weather balloons and rain water falls collectors.
----------------------------------------------------------
Updates
In addition to the GIF animation of the worlwide Cs137 isotope dry deposition accumulation, I'll now add the everyday update of [www.nuc.berkeley.edu...]UC Berkeley Nuclear Engineering Air Monitoring Station[/url], from the Department of the Nuclear Engeneering, University of California, Berkeley.
Here are the preliminary statements:
The UC Berkeley Department of Nuclear Engineering is currently performing measurements to detect a potential increase in radiation here in Berkeley that could be associated with the release of radioactive materials in Japan. We perform this measurement by sampling air flowing through a particle filter mounted at the top of Etcheverry Hall. We first calibrated the monitor to account for normal background radiation levels.
After a period of 8-12 hours of particle collection, we take the filter down to our counting station in our laboratory and, using highly sensitive detection instruments, can determine the concentration of target radioactive elements within the air sample collected by counting gamma-ray photons emitted. This is to determine if any radiation signatures are present above normal background radiation levels.
The detection instruments we are using are not only able to measure the amount of radiation in the sample, but also its energy. More specifically, we measure gamma rays and their energies in our detector, which provides a unique fingerprint of a specific radioisotope.
By measuring the energy of the gamma rays with high precision, we can not only determine the amount of radiation due to a specific radioisotope, but we can distinguish it from the natural background radiation we measure as well.
For example, we can measure gamma rays from isotopes of Cesium (Cs) such as Cs-134 or Cs-137, Iodine (e.g. I-131), or Tellurium (e.g. Te-132), which we would expect to see from the release due to the fission products in Japan. It is important to realize that all because we can detect radiation does not mean it is harmful. Our instruments are so sensitive that we can measure radiation far below the levels of the natural background radiation we live in day to day.
For example, even if we are able to observe a tens of counts per hour of I-131 or Cs-137, we have to compare that to a natural background radiation level of > 300000 counts per hour when expressed in these terms. Therefore, even if we were able to observe these small amounts of radiation, it will only lead to a very small increase in the radiation we are exposed to due to the natural radiation and will have no measurable health effect.
We express the biological effect of radiation in terms of doses and dose rates expressed for example in terms of millirem (mrem) or milli-Sievert (mSv) per hour, day, or year. One millisievert is equivalent to 100 millirems. The average dose of a person living in the U.S. is about 620 mrem (6.2 mSv) per year. This can be broken down into about 300 mrem per year due to natural background, such as radon and ultraviolet rays from the sun, and 320 mrem per year due to artificial exposures, such as X-rays or Computed Tomography (CT) scans. One Chest-CT scan produces about a 1,000 mrem dose of radiation exposure, or about three times the level due to just the natural exposure. The radiation levels we expect to measure due to the events in Japan will be 1,000 times smaller than the natural levels of background radiation.

The maximum measured dose rate at the Fukushima plant has been reported at 11.4 mSv/hr which puts the dose in one hour to be near a full-body CT scan and one-fifth the annual US dose limit for US radiation workers. No health effects have been proven below the 50mSv per year level.
-------------------------------------------
3/29 (11:35am): Our air results and rain water results have both been updated. The isotope amounts in both have leveled out, which means that we might not be observing a downward trend at this point. We are continuing these tests so that we can observe the eventual expected decline in activities.
3/28 (2:24pm): Latest Air and Water Results data/spectra is now posted. We note decreased levels from previous peaks. This could be due to a number of reasons to include the lack of rain in the past 48 hours to an actual lower amount of particles in the air. Note, this is not a trend unless we see a sustained reduction. We are heading into a period of high pressure in the Bay Area and the jet stream will shift away from our area and this may cause even lower readings. We continue to test run-off creek water, tap water, and milk.
3/27 (2:00pm): Strawberry Creek run off results posted. We do observe all signatures in the run off creek water, but the dilution is from ~2% for I-131 to 15% for Cs137. However, Cs137 and Te132 are just below minimum detectability for our system and the real dilution is most likely closer to 2-5%. Reservoir and tap water sampling begins next week. These activities are factors of 10 to 50 below rain water results.
----------------------------------------------

The dry deposition displaying the total accumulated deposition along the path of the plume
edit on 30-3-2011 by elevenaugust because: (no reason given)
Well this might explain why we're making 60 Geiger counter housings at work this week!!?
Wow...good job i'v got Potassium iodide & respirators
Wow...good job i'v got Potassium iodide & respirators
reply to post by elevenaugust
maybe the power company is waiting for BP to make a merger offer... 19 days and counting
maybe the power company is waiting for BP to make a merger offer... 19 days and counting
new topics
-
I hate dreaming
Rant: 27 minutes ago -
Is the origin for the Eye of Horus the pineal gland?
Philosophy and Metaphysics: 2 hours ago -
Man sets himself on fire outside Donald Trump trial
Mainstream News: 2 hours ago -
Biden says little kids flip him the bird all the time.
2024 Elections: 2 hours ago -
The Democrats Take Control the House - Look what happened while you were sleeping
US Political Madness: 3 hours ago -
Sheetz facing racial discrimination lawsuit for considering criminal history in hiring
Social Issues and Civil Unrest: 3 hours ago -
In an Historic First, In N Out Burger Permanently Closes a Location
Mainstream News: 5 hours ago -
MH370 Again....
Disaster Conspiracies: 5 hours ago -
Are you ready for the return of Jesus Christ? Have you been cleansed by His blood?
Religion, Faith, And Theology: 7 hours ago -
Chronological time line of open source information
History: 9 hours ago
top topics
-
In an Historic First, In N Out Burger Permanently Closes a Location
Mainstream News: 5 hours ago, 14 flags -
The Democrats Take Control the House - Look what happened while you were sleeping
US Political Madness: 3 hours ago, 10 flags -
Thousands Of Young Ukrainian Men Trying To Flee The Country To Avoid Conscription And The War
Other Current Events: 15 hours ago, 8 flags -
A man of the people
Medical Issues & Conspiracies: 10 hours ago, 8 flags -
Man sets himself on fire outside Donald Trump trial
Mainstream News: 2 hours ago, 7 flags -
Biden says little kids flip him the bird all the time.
2024 Elections: 2 hours ago, 6 flags -
4 plans of US elites to defeat Russia
New World Order: 12 hours ago, 4 flags -
Is the origin for the Eye of Horus the pineal gland?
Philosophy and Metaphysics: 2 hours ago, 4 flags -
Are you ready for the return of Jesus Christ? Have you been cleansed by His blood?
Religion, Faith, And Theology: 7 hours ago, 3 flags -
Sheetz facing racial discrimination lawsuit for considering criminal history in hiring
Social Issues and Civil Unrest: 3 hours ago, 3 flags
active topics
-
Man sets himself on fire outside Donald Trump trial
Mainstream News • 23 • : FlyersFan -
The Democrats Take Control the House - Look what happened while you were sleeping
US Political Madness • 22 • : xuenchen -
12 jurors selected in Trump criminal trial
US Political Madness • 90 • : DontTreadOnMe -
I hate dreaming
Rant • 2 • : FlyersFan -
America's Infant Mortality Rate Increases for the First Time in 20 Years
Medical Issues & Conspiracies • 22 • : BasicResearchMethods -
Post A Funny (T&C Friendly) Pic Part IV: The LOL awakens!
General Chit Chat • 7126 • : imitator -
Biden says little kids flip him the bird all the time.
2024 Elections • 10 • : imitator -
Thousands Of Young Ukrainian Men Trying To Flee The Country To Avoid Conscription And The War
Other Current Events • 28 • : Xtrozero -
Is the origin for the Eye of Horus the pineal gland?
Philosophy and Metaphysics • 4 • : JoelSnape -
Fossils in Greece Suggest Human Ancestors Evolved in Europe, Not Africa
Origins and Creationism • 72 • : Xtrozero
