It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
11
share:
No Rise of Atmospheric Carbon Dioxide Fraction in Past 160 Years, New Research Finds
www.sciencedaily.com
(visit the link for the full news article)
ScienceDaily (Dec. 31, 2009) — Most of the carbon dioxide emitted by human activity does not remain in the atmosphere, but is instead absorbed by the oceans and terrestrial ecosystems. In fact, only about 45 percent of emitted carbon dioxide stays in the atmosphere.
While I am sitting squarely on the fence on the AGW issue, it has become nearly impossible too put any faith in the science they have obviously
tinkered with in their climate models, statistics, etc that they feed to the public. Whatever their motives are, they have obviously been stacking the
numbers in favor of AGW.
www.sciencedaily.com
(visit the link for the full news article)
www.sciencedaily.com
(visit the link for the full news article)
You do know that the oceans are currently SATURATED with CO2 dont you? Due to this CO2 saturation in the oceans, it is turning more acidic which
hampers the devolopment of corals, shelled creatures such as crabs and the most important being plankton. IT also hurts the fish eggs with thier thin
membrane walls....
I remember when we called it "The Greenhouse Effect" then we called it "Global Warming" then we called it "Anthropogenic Global Warming" then we
called it "Climate Change".
This is all a lie to separate you from your earned wealth and your individual liberty. It's a ponzi scheme developed by the Rothschild family to collect more wealth and power. Now that the scheme is played out and everyone is feeling like they've been had, they trot out Al Gore as the fall guy.
But fear not, Al has been most handsomely rewarded for his loyalty.
This is all a lie to separate you from your earned wealth and your individual liberty. It's a ponzi scheme developed by the Rothschild family to collect more wealth and power. Now that the scheme is played out and everyone is feeling like they've been had, they trot out Al Gore as the fall guy.
But fear not, Al has been most handsomely rewarded for his loyalty.
Originally posted by DaRAGE
You do know that the oceans are currently SATURATED with CO2 dont you? Due to this CO2 saturation in the oceans, it is turning more acidic which hampers the devolopment of corals, shelled creatures such as crabs and the most important being plankton. IT also hurts the fish eggs with thier thin membrane walls....
Thats not really the point though is it ?
We have been told that rising CO2 in the atmosphere has caused global warming.
Now we find there has been no rise in CO2 in the atmosphere ... we have been lied to... simple as.
Please wake up.
"In fact, only about 45 percent of emitted carbon dioxide stays in the atmosphere."
I don't believe in the whole global warming thing, but even so 45% is still alot when put into perspective.
[edit on 31-12-2009 by keagan13]
I don't believe in the whole global warming thing, but even so 45% is still alot when put into perspective.
[edit on 31-12-2009 by keagan13]
speaking of, oceans,
Melting Glaciers Nourishing Oceans With Ancient
CarbonChristine
Dell'Amore
National Geographic News
December 30, 2009
news.nationalgeographic.com...
Melting Glaciers Nourishing Oceans With Ancient
CarbonChristine
Dell'Amore
National Geographic News
December 30, 2009
news.nationalgeographic.com...
The new study goes against a long-held belief that older carbon is less palatable to simple organisms, Hood added.
For instance, in most of the world's water bodies, the older the carbon, the less easily microbes can digest it.
"That's the stuff that's been worked over—it's no good," Hood said. "But in our case the older it was, the more the microbes wanted to eat it."
That's mainly because glacial carbon is made of dead microbes that have been essentially preserved in ice.
The dead microbes contain more easily digestible nitrogen and not much lignin, a plant compound that's tough for microbes to break down.
Overall, the contribution of glaciers to the productivity of rivers and oceans is "greatly underappreciated," the study authors write.
"It's good to understand the uniqueness of glacier ecosystems and the important role that they play as a source of water and nutrients," Hood added.
Originally posted by bigyin
Now we find there has been no rise in CO2 in the atmosphere ... we have been lied to... simple as.
Please wake up.
Re-read the article. No where does it say that there has been "rise in CO2 in the atmosphere".
What the article is saying is that the proportion of atmospheric CO2 has not increased compared to that absorbed by carbon sinks. Everyone agrees that atmospheric CO2 has increased.
The issue, as it's always been, is weather or not man's CO2 emissions are the cause of global warming.
reply to post by C.H.U.D.
The last paragraph states:
"In contradiction to some recent studies, he finds that the airborne fraction of carbon dioxide has not increased either during the past 150 years or during the most recent five decades."
What does that mean ?
The last paragraph states:
"In contradiction to some recent studies, he finds that the airborne fraction of carbon dioxide has not increased either during the past 150 years or during the most recent five decades."
What does that mean ?
reply to post by DaRAGE
You are talking about carbonic acid (H2CO3) that naturally occurs when water comes into contact with carbon dioxide. It's a good thing it does, because that is how carbon dioxide gets into the water in the first place, so oceanic plant life can survive.
Carbonic acid under atmospheric pressure is not a strong acid. It is very weak compared to other acids like nitric acid (HNO3), sulfuric acid (H2SO4), or hydrochloric acid (HCl). Want a simple comparison?
Presently, the undeveloped and developing countries of the world are allowing massive amounts of sulfur impurity in their fuels. Ships have excessive amounts allowed from all countries. In the United States, diesel fuel allows only 15 ppm sulfur ad gasoline is limited to 30 ppm sulfur. impurities up to 5000 ppm are allowed elsewhere.
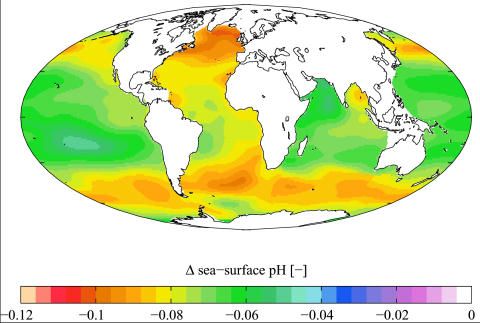
Take a look at these images. The first one is from Wikipedia and shows oceanic acidification levels geographically:
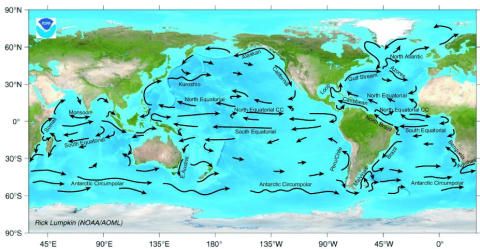
The second one shows oceanic currents, from NOAA:
Compare the two. Notice that the Atlantic seems to have more than its share of of acid especially south of Greenland. Now follow the currents from that area... down the coast of Newfoundland (medium traffic, 15 ppm sulfur), past the Eastern US seaboard (massive traffic, 15 ppm sulfur), past Brazil (high traffic, 500/2000 ppm sulfur), and on down past Argentina (medium traffic, 50/500 ppm sulfur). Those last two countries have some of the highest sulfur content in their fuel in the world! And of course, the traffic along our eastern seaboard is world-renowned.
Now look at the Antarctic circumpolar current... it aligns with an abnormally high acid concentration as well. Where does that circumpolar current originate from? Let's see... South Africa (unknown but assumed high sulfur content)... Argentina (50/500 ppm sulfur content)... Australia (High traffic, 10 ppm sulfur restrictions)... and some input from New Zealand (massive traffic and 10 ppm sulfur).
Check out the Pacific just off the coast of China... there is a nice little stretch of acidification right there in the middle of nowhere. It coincides with the Kuroshio current which runs right up the Chinese/Japanese shores. Need I say more? China is high traffic and 2000 ppm sulfur, while Japan is massive traffic and unknown sulfur.
So how about we stop trying to blame everything on carbon dioxide? There are other chemicals out there, you know.
TheRedneck
You are talking about carbonic acid (H2CO3) that naturally occurs when water comes into contact with carbon dioxide. It's a good thing it does, because that is how carbon dioxide gets into the water in the first place, so oceanic plant life can survive.
Carbonic acid under atmospheric pressure is not a strong acid. It is very weak compared to other acids like nitric acid (HNO3), sulfuric acid (H2SO4), or hydrochloric acid (HCl). Want a simple comparison?
- Sulfuric acid (dilute) is in your car battery.
- Carbonic acid (concentrated under high pressure) is in that can of soda you drank this morning.
Presently, the undeveloped and developing countries of the world are allowing massive amounts of sulfur impurity in their fuels. Ships have excessive amounts allowed from all countries. In the United States, diesel fuel allows only 15 ppm sulfur ad gasoline is limited to 30 ppm sulfur. impurities up to 5000 ppm are allowed elsewhere.
Take a look at these images. The first one is from Wikipedia and shows oceanic acidification levels geographically:
The second one shows oceanic currents, from NOAA:
Compare the two. Notice that the Atlantic seems to have more than its share of of acid especially south of Greenland. Now follow the currents from that area... down the coast of Newfoundland (medium traffic, 15 ppm sulfur), past the Eastern US seaboard (massive traffic, 15 ppm sulfur), past Brazil (high traffic, 500/2000 ppm sulfur), and on down past Argentina (medium traffic, 50/500 ppm sulfur). Those last two countries have some of the highest sulfur content in their fuel in the world! And of course, the traffic along our eastern seaboard is world-renowned.
Now look at the Antarctic circumpolar current... it aligns with an abnormally high acid concentration as well. Where does that circumpolar current originate from? Let's see... South Africa (unknown but assumed high sulfur content)... Argentina (50/500 ppm sulfur content)... Australia (High traffic, 10 ppm sulfur restrictions)... and some input from New Zealand (massive traffic and 10 ppm sulfur).
Check out the Pacific just off the coast of China... there is a nice little stretch of acidification right there in the middle of nowhere. It coincides with the Kuroshio current which runs right up the Chinese/Japanese shores. Need I say more? China is high traffic and 2000 ppm sulfur, while Japan is massive traffic and unknown sulfur.
- Sulfur contents are as of latest regulations on diesel fuel and are taken from
en.wikipedia.org...
Some countries have been omitted for brevity.
So how about we stop trying to blame everything on carbon dioxide? There are other chemicals out there, you know.
TheRedneck
reply to post by bigyin
As I understand it, it's referring to the ratio of CO2 in the atmosphere as compared with the ratio in carbon sinks like the sea. It's saying the ratio has not changed, although the total amount in the system (ie atmosphere and sinks) has. So atmospheric CO2 can go up, but that ratio remains unaffected.
As I understand it, it's referring to the ratio of CO2 in the atmosphere as compared with the ratio in carbon sinks like the sea. It's saying the ratio has not changed, although the total amount in the system (ie atmosphere and sinks) has. So atmospheric CO2 can go up, but that ratio remains unaffected.
reply to post by C.H.U.D.
This is the way I read it.
The article is stating the the percentage (fraction) of airborne co2 has not increased in the last 150 years. They also state that most climate models assume that this fraction is increasing. So, this would further indicate that all those climate models that are assuming this fraction is rising are invalid.
This article does not draw any conclusions about the amount of co2 that has been absorbed into the ocean, but simply notes that most of the total amount of co2 released is absorbed and does not stay airborne.
This is the way I read it.
The article is stating the the percentage (fraction) of airborne co2 has not increased in the last 150 years. They also state that most climate models assume that this fraction is increasing. So, this would further indicate that all those climate models that are assuming this fraction is rising are invalid.
This article does not draw any conclusions about the amount of co2 that has been absorbed into the ocean, but simply notes that most of the total amount of co2 released is absorbed and does not stay airborne.
Geo-engineering is going to take a crazy toll on the worlds health, but it may take up to 20 years for results to kick in. This is the primary spray
they are using. This is going to increase ocean acidity too! Have a scan over what I dug up here if you like.
From Geo-engineering, Wikipedia
From the provincial government of Manitoba, outlining Sulphur Dioxide health risks:
www.gov.mb.ca...
But here's what the climate activists are saying ...
Can a Million Tons of Sulfur Dioxide Combat Climate Change?
www.wired.com...
Ontario's WSIB policy for workers affected by such illnesses ...
And welcome to the world of tomorrow ...
Images of Sulphur Pollution
www.google.ca... p&ct=title&resnum=4&ved=0CB0QsAQwAw
Aerosol formation
Primary aerosol formation, also known as homogeneous aerosol formation results when gaseous SO2 combines with water to form aqueous sulfuric acid (H2SO4). This acidic liquid solution is in the form of a vapor and condenses onto particles of solid matter, either meteoritic in origin or from dust carried from the surface to the stratosphere. Secondary or heterogeneous aerosol formation occurs when H2SO4 vapor condenses onto existing aerosol particles. Existing aerosol particles or droplets also run into each other, creating larger particles or droplets in a process known as coagulation. The larger the particles or droplets, the shorter their residence time in the stratosphere and the less effective they are at scattering visible sunlight.
From Geo-engineering, Wikipedia
From the provincial government of Manitoba, outlining Sulphur Dioxide health risks:
What are the effects of sulphur dioxide on people?
For longer exposures, sulphur dioxide levels above 0.15 ppm have been linked with increased hospital admissions for cardiac or respiratory illnesses. Long-term exposures to 0.027 to 0.031 ppm of sulphur dioxide with high levels of particulate matter in the air have been associated with an increase in respiratory illnesses in children.
www.gov.mb.ca...
But here's what the climate activists are saying ...
Can a Million Tons of Sulfur Dioxide Combat Climate Change?
A million tons of sulfur dioxide would be needed to begin the cooling process. Luckily SO2, a byproduct of coal-burning power plants, is a common industrial chemical.
www.wired.com...
Ontario's WSIB policy for workers affected by such illnesses ...
Policy
Chronic Obstructive Lung Disease (COLD) in smelter workers is accepted as an occupational disease under sections 1(1)(n) and 122 of the Workers' Compensation Act as peculiar to and characteristic of exposure to Sulphur Dioxide (SO2) and particulates.
Guidelines
Acceptance of a claim is dependent on objective medical evidence of COLD and impaired pulmonary function.
A diagnosis of primary bronchiectasis, chronic upper respiratory disease, or primary asthma without the presence of COLD is not considered an acceptable basis to claim under this policy.
Entitlement criteria
Based on medical studies, claims for COLD with impairment shall be favourably considered when all of the following circumstances apply.
Nature of exposure
A clear and adequate history of exposure to any smelting process involving the production of sulphur dioxide and particulates as a significant by-product of the process must be established. This includes roasting, smelting, converting of metallic ores, and the secondary production of sulphuric acid.
Intensity of exposure
Sulphur dioxide exposure is established when levels of more than 5 parts per million (5 p.p.m.) are recorded repeatedly over the exposure period.
Duration of exposure
A minimum of 20 years established exposure.
And welcome to the world of tomorrow ...
Images of Sulphur Pollution
www.google.ca... p&ct=title&resnum=4&ved=0CB0QsAQwAw
new topics
-
Weinstein's conviction overturned
Mainstream News: 53 minutes ago -
Supreme Court Oral Arguments 4.25.2024 - Are PRESIDENTS IMMUNE From Later Being Prosecuted.
Above Politics: 2 hours ago -
Krystalnacht on today's most elite Universities?
Social Issues and Civil Unrest: 2 hours ago -
Chris Christie Wishes Death Upon Trump and Ramaswamy
Politicians & People: 2 hours ago -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media: 5 hours ago -
Any one suspicious of fever promotions events, major investor Goldman Sachs card only.
The Gray Area: 7 hours ago -
God's Righteousness is Greater than Our Wrath
Religion, Faith, And Theology: 11 hours ago
top topics
-
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News: 16 hours ago, 11 flags -
Krystalnacht on today's most elite Universities?
Social Issues and Civil Unrest: 2 hours ago, 7 flags -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues: 17 hours ago, 6 flags -
Sunak spinning the sickness figures
Other Current Events: 16 hours ago, 5 flags -
Supreme Court Oral Arguments 4.25.2024 - Are PRESIDENTS IMMUNE From Later Being Prosecuted.
Above Politics: 2 hours ago, 5 flags -
Weinstein's conviction overturned
Mainstream News: 53 minutes ago, 4 flags -
Electrical tricks for saving money
Education and Media: 14 hours ago, 4 flags -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media: 5 hours ago, 2 flags -
Any one suspicious of fever promotions events, major investor Goldman Sachs card only.
The Gray Area: 7 hours ago, 2 flags -
Chris Christie Wishes Death Upon Trump and Ramaswamy
Politicians & People: 2 hours ago, 1 flags
active topics
-
HORRIBLE !! Russian Soldier Drinking Own Urine To Survive In Battle
World War Three • 41 • : CristianVictoria -
Chris Christie Wishes Death Upon Trump and Ramaswamy
Politicians & People • 9 • : Hecate666 -
Nearly 70% Of Americans Want Talks To End War In Ukraine
Political Issues • 80 • : FlyersFan -
Supreme Court Oral Arguments 4.25.2024 - Are PRESIDENTS IMMUNE From Later Being Prosecuted.
Above Politics • 29 • : xuenchen -
University of Texas Instantly Shuts Down Anti Israel Protests
Education and Media • 103 • : DBCowboy -
VP's Secret Service agent brawls with other agents at Andrews
Mainstream News • 45 • : network dude -
Weinstein's conviction overturned
Mainstream News • 10 • : xuenchen -
Remember These Attacks When President Trump 2.0 Retribution-Justice Commences.
2024 Elections • 57 • : TzarChasm -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 670 • : cherokeetroy -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 159 • : 5thHead
11