It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
originally posted by: Char-Lee
a reply to: METACOMET
Drinking blood would do nothing as it would be destroying in the digesting of it. If it had an effect then so should cow blood when people eat rare meat.
Prion proteins are not destroyed in digestion - but they are more stable than most ordinary proteins. Still, GDF11 might survive digestion, be absorbed into the lymphatic system and from there, get into the blood where it's needed.
...More research required.
HMMM. Seems likely GDF11 misfolds in response to environmental influences - the misfolded form spreads as we age, and by the time we hit 25-40, it's just a numbers game with the misfolded form winning out. ....A prion-related post-transcription epigenetic mechanism.
Bets anyone?
edit on 5/5/14 by soficrow because: add hmmm
a reply to: METACOMET
Its nothing new, in'fact was watching a piece on this some hours ago on the TV before I came online about how young blood will help revitalize and rejuvenate the body, stimulating everything from muscle to cognitive processes and memory, to even if not reverse aging at least keeping it at bay. Your tittle is a bit sensational as you cant drink blood for any of this to happen, but the truth is this is much worse in many ways.
Imagine some years from now that there will be a market on young blood transfusions to keep those with the funds going a few years longer. I mean a much bigger market then it is now. And what is worse some guy in a cape sucking on your neck at night, or a giant conglomeration and hidden operation whereby the sale of blood of the young and kids thousands and thousands of them becomes a very profitable business. Were there is a profit there is a way, never underestimate the depths of human depravity and greed. Vampire therapy indeed, fact is generally more stranger and much more scarier then fiction.
Its nothing new, in'fact was watching a piece on this some hours ago on the TV before I came online about how young blood will help revitalize and rejuvenate the body, stimulating everything from muscle to cognitive processes and memory, to even if not reverse aging at least keeping it at bay. Your tittle is a bit sensational as you cant drink blood for any of this to happen, but the truth is this is much worse in many ways.
Imagine some years from now that there will be a market on young blood transfusions to keep those with the funds going a few years longer. I mean a much bigger market then it is now. And what is worse some guy in a cape sucking on your neck at night, or a giant conglomeration and hidden operation whereby the sale of blood of the young and kids thousands and thousands of them becomes a very profitable business. Were there is a profit there is a way, never underestimate the depths of human depravity and greed. Vampire therapy indeed, fact is generally more stranger and much more scarier then fiction.
edit on 11pmMondaypm052014f1pmMon, 05 May 2014 23:25:44 -0500 by galadofwarthethird because: (no reason given)
lets not forget that some of those missing children could possibly be attributed to reptillians satisfying their appetites. I remember hearing that
blood itself is very beneficial to the body and that is one of the gov't "secrets" they keep from us. makes sense that the gov't wouldnt want ppl
to know this. One could imagine the amount of murders that would be linked to this.
The same thread on Medical Issues & Conspiracies forum :
www.abovetopsecret.com...
And the same kind of research last year :
www.dailymail.co.uk... -organ.html
And chinese ate babies and fetuses old news :
www.abovetopsecret.com...
And the same kind of research last year :
www.dailymail.co.uk... -organ.html
The vampire treatment that 'rejuvenates' ageing hearts: Dose of young blood can reverse life-threatening thickening of organ
And chinese ate babies and fetuses old news :
Injecting blood can also easily kill you, so dont anybody get any bright ideas. Not only do you run the risk of pathogens, but you run the risk of an
immediate hemolytic transfusion reaction. Futhermore, if you by chance live through that you can produce antibodies to the foreign blood dramatically
increasing your chance of having difficulty finding compatible blood should you actually need it. Now the possibilty of autologously extracting you
own blood when you are young and storing it for when you are older is intriguing. However, there is only one viable long term storage option for
whole blood that I am aware of, glycerolization. The storage of and deglycerization of said blood may prove moot due to destruction of protein of
interest. Probably crap science, at best, but interesting.
originally posted by: soficrow
originally posted by: Char-Lee
a reply to: METACOMET
Drinking blood would do nothing as it would be destroying in the digesting of it. If it had an effect then so should cow blood when people eat rare meat.
Prion proteins are not destroyed in digestion - but they are more stable than most ordinary proteins. Still, GDF11 might survive digestion, be absorbed into the lymphatic system and from there, get into the blood where it's needed.
...More research required.
HMMM. Seems likely GDF11 misfolds in response to environmental influences - the misfolded form spreads as we age, and by the time we hit 25-40, it's just a numbers game with the misfolded form winning out. ....A prion-related post-transcription epigenetic mechanism.
Bets anyone?
But it's not a prion.
well doesnt this Open Doors to alot of things
Vampire hmm
It makes you wonder
The Protein GDF11 aka Youth Protein that Dissipates when you Get older
when you look at the Bible/Torah of what Books, has Claimed
About Noah and his Descendants all living pass the 120 Marker that US Humans Have Now
When Some of the People in the Pre Flood ( Antediluvian Times ) that lived Close to A Thousand then After The Flood
the Age then diluted down from every Generation after. to the 120 year Mark DID this Protein last longer through Age ?
Then you have People Search for Fountain of Youth through out the Millennium ?
Blood Sacrifice and you have Holy Books telling YOU NOT to Drink Blood from Human and Animals
A Coincidence?
So I guess The Hebrew God Reduced this Protein to a Certain Limit. in Humans or even all life
So does this Make Some of those Legend and Holy Book to be Right ?
and what about The Pre Colombian Natives all Painted Up in Blue for Sacrifice Ritual For Draining Blood ?
and the King/Chief to Drink it ?
of what US Human can do to day is Reverse the Slowing Down this Protein Alter to Modify through BIO Engineering
and Mass Replicate it Self and to keep the Protein to last Longer
But this is just Whoa!!
Young Blood May Hold Key to Reversing Aging
www.nytimes.com...
Biomedicine – Part 10: Bioengineering the End to Aging
www.21stcentech.com...
Anti-Aging Research Advances Featured in QB3 Symposium
Chronic Diseases of Aging are Targeted in New Ways
www.ucsf.edu...
Vampire hmm
It makes you wonder
The Protein GDF11 aka Youth Protein that Dissipates when you Get older
when you look at the Bible/Torah of what Books, has Claimed
About Noah and his Descendants all living pass the 120 Marker that US Humans Have Now
When Some of the People in the Pre Flood ( Antediluvian Times ) that lived Close to A Thousand then After The Flood
the Age then diluted down from every Generation after. to the 120 year Mark DID this Protein last longer through Age ?
Then you have People Search for Fountain of Youth through out the Millennium ?
Blood Sacrifice and you have Holy Books telling YOU NOT to Drink Blood from Human and Animals
A Coincidence?
So I guess The Hebrew God Reduced this Protein to a Certain Limit. in Humans or even all life
So does this Make Some of those Legend and Holy Book to be Right ?
and what about The Pre Colombian Natives all Painted Up in Blue for Sacrifice Ritual For Draining Blood ?
and the King/Chief to Drink it ?
of what US Human can do to day is Reverse the Slowing Down this Protein Alter to Modify through BIO Engineering
and Mass Replicate it Self and to keep the Protein to last Longer
But this is just Whoa!!
“This should give us all hope for a healthier future,” said Prof Doug Melton, of Harvard's Department of Stem Cell and Regenerative Biology. “We all wonder why we were stronger and mentally more agile when young, and these two unusually exciting papers actually point to a possible answer. “There seems to be little question that, GDF11 has an amazing capacity to restore aging muscle and brain function.”
Last year the team discovered that the protein could repair damaged hearts. But the new study showed that that raising the levels of the GDF11 protein in older mice improved the function of every organ in the body. Harvard stem cell biologist Prof Lee Rubin added: “We do think that, at least in principal, there will be a way to reverse some of the decline of aging with a single protein.
"It isn't out of question that GDF11, or a drug developed from it, might be worthwhile in Alzheimer's Disease.”
It is likely that the protein is at least partly responsible for the parallel finding by Stanford University that young blood can reverse the signs of ageing. In the study, the blood of three-month-old mice was repeatedly injected into 18-month-old mice near the end of their natural life span. The "vampire therapy" improved the performance of the elderly mice in memory and learning tasks.
Young Blood May Hold Key to Reversing Aging
www.nytimes.com...
Biomedicine – Part 10: Bioengineering the End to Aging
www.21stcentech.com...
Anti-Aging Research Advances Featured in QB3 Symposium
Chronic Diseases of Aging are Targeted in New Ways
www.ucsf.edu...
edit on 6-5-2014 by Wolfenz because: (no reason given)
a reply to: METACOMET
Very interesting article you found there.
But if you think that's weird... google this "Kevin Annett" guy lol...
Here's one of his youtube videos -
And of course the so-called Republic of Kanata
The entire world has gone insane.
I still always keep an open mind and try to look at things from all angles but at this point I've seen so much wild stuff who the hell knows anymore.
Very interesting article you found there.
But if you think that's weird... google this "Kevin Annett" guy lol...
Here's one of his youtube videos -
And of course the so-called Republic of Kanata
The entire world has gone insane.
I still always keep an open mind and try to look at things from all angles but at this point I've seen so much wild stuff who the hell knows anymore.
edit on 6-5-2014 by Time2Think because: fixed typos
Factor in the Reptilian Conspiracy or even the Elites in general, for their claimed link(and exposed) to youth. With that might be onto something
here. Wonder if this scientific find will spark whistle blowers in the know.
From what I recall, Reptilians, and possibly the Elites(link to kidnapping children from foster care) prefer(probably despite race but closely related to); brunette hair/ green or blue eyes are a favorite, redheads and blondes. Perhaps linked to blood types, or is the gene combos preferred?
originally posted by: Xcouncil=wisdom
Blood transfusions have been around for a long time now, I would think that if there was any real benefit to using the blood of the youth, its effects would have come up statistically already...would'nt it?
Some people thrive from their transfusions while others don't see any change...
I mean, I know about all the people that get Hepatitis and AIDS and all those other diseases getting tracked back to bad blood that they got transfused into them, so if the opposite happened and they saw super fast recovery, and anti aging then it would have also been tracked back to the blood they got of a young one.
But in a completely other line of thinking....is there a particular race who seem to have more of this protein in their young blood?
From what I recall, Reptilians, and possibly the Elites(link to kidnapping children from foster care) prefer(probably despite race but closely related to); brunette hair/ green or blue eyes are a favorite, redheads and blondes. Perhaps linked to blood types, or is the gene combos preferred?
originally posted by: dickrichards
Injecting blood can also easily kill you, so dont anybody get any bright ideas. Not only do you run the risk of pathogens, but you run the risk of an immediate hemolytic transfusion reaction. Futhermore, if you by chance live through that you can produce antibodies to the foreign blood dramatically increasing your chance of having difficulty finding compatible blood should you actually need it. Now the possibilty of autologously extracting you own blood when you are young and storing it for when you are older is intriguing. However, there is only one viable long term storage option for whole blood that I am aware of, glycerolization. The storage of and deglycerization of said blood may prove moot due to destruction of protein of interest. Probably crap science, at best, but interesting.
You think elites can't find the clear blood they need ?
originally posted by: NavyDoc
originally posted by: soficrow
originally posted by: Char-Lee
a reply to: METACOMET
Drinking blood would do nothing as it would be destroying in the digesting of it. If it had an effect then so should cow blood when people eat rare meat.
Prion proteins are not destroyed in digestion - but they are more stable than most ordinary proteins. Still, GDF11 might survive digestion, be absorbed into the lymphatic system and from there, get into the blood where it's needed.
...More research required.
HMMM. Seems likely GDF11 misfolds in response to environmental influences - the misfolded form spreads as we age, and by the time we hit 25-40, it's just a numbers game with the misfolded form winning out. ....A prion-related post-transcription epigenetic mechanism.
Bets anyone?
But it's not a prion.
I did not say GDF11 is a prion - I was musing about whether or not it might be stable enough to survive digestion if ingested, and then, about whether or not it misfolds as we age. I also said more research is required.
If you have information on research into GDF11's normal and misfolded states, please post the links.
In any event, I stand by my speculation that GDF11 might misfold as we age and thus, lose it's rejuvenating power - perhaps more likely, an epigenetic mechanism may "turn off" the GDF11 gene, and prevent its expression.
they get their elixir of youth, half of all placental blood of your newborn at the same time forbidding you your elixir of life fresh milk and whey
you buy dairy products it glue for the vessels and after the autopsy surgeon can not cut an artery with a sharp scalpel p.s. your wife will soon give
birth? and you want to harm the vampire backstage? poison all the placenta or give empty without blood
edit on 7/5/14 by mangust69 because:
p.s.
a reply to: candlestick
Wow this thread has gone to the bizarre. Read up on transfusion medicine and the human RBC antigen system then get back to me.
Wow this thread has gone to the bizarre. Read up on transfusion medicine and the human RBC antigen system then get back to me.
Public records of using blood for 'healing' date back to the 1600's. There is no doubt the big focus with big money was about 'reversing aging'
and no doubt much experimentation occurred without making it into the public record. Even Nazi experiments were big into anti-aging. We're just
seeing the tip of the tip of the iceberg here.
Matthew Rowlinson, “On the First Medical Blood Transfusion Between Human Subjects, 1818″
Abstract
The first experiments in blood transfusion took place in the seventeenth century, using blood drawn from animals. After the death of a French patient and the trial of his physician for manslaughter, transfusion was abandoned for a century and a half. When it resumed in the nineteenth century, the first trials used human blood. They were conducted by the obstetrician James Blundell, who developed transfusion to treat women suffering from hemorrhage after childbirth. During the course of the century, transfusion was applied as a remedy to different kinds of sicknesses and injuries, and performed at different times with various fluids. By the 1880s, British doctors had abandoned transfusion with blood in favor of infusion with saline solution; the practice was only again resumed after the recognition of human blood types in the twentieth century. Developments in the practice of transfusion and infusion during the century reflect shifting views of how gender and species identity are embodied, and of relations of affinity and immunity between bodies of different kinds. They also reflect a shift from a vitalist to a mechanist understanding of blood and its function.
Blood transfusion as a medical practice dates to the seventeenth century;[1]> the first experiments were performed a few decades after William Harvey published his demonstration that blood circulates in the body in De Motu Cordis (1628). Harvey’s discovery showed that blood circulated indefinitely, rather than being either consumed within the body or expelled from it. To cure pathologies of the blood was thus no longer exclusively a matter of stimulating or preventing its evacuation; now the goal was to affect blood’s circulation within the body and its interaction with other organs. The earliest blood transfusions involving human subjects were carried out almost simultaneously in France and England in 1667. Unlike modern transfusion, these first experiments did not treat subjects who had suffered losses of blood. Rather than deficiency in the quantity of the circulation, they hoped to cure defects in its quality, as expressed by the temperament of the subject. They were thus in every case transfusions between species and in no case aimed to treat hemorrhage or any other type of blood loss. In England on 23 November, a 22-year-old Bachelor of Arts was transfused with the blood of a sheep, in order to moderate what was considered the excessive heat of his brain. The recipient survived the operation, and Pepys records in his diary that after six days he gave a report to the Royal Society, speaking in Latin, in which he pronounced himself better. In the same year in France a series of similar experiments were made by Jean-Baptiste Denis, who was physician to Louis XIV. In the fourth and most celebrated of his cases, he on two occasions transfused calf’s blood into the veins of a certain Antoine Mauroy, in order to endow him with docility. Denis claimed temporary successes—Mauroy seems to have gone into shock—but was unable to continue the treatment with a third transfusion owing to the patient’s refusal. When Mauroy shortly after died, Denis was tried for manslaughter; though he was acquitted, transfusion was made illegal in France and abandoned throughout Europe for over a century (Maluf 64-67).
When transfusion experiments resumed in the nineteenth century it was on different principles. James Blundell’s original article on the subject appeared in 1818, shortly before his first attempt on a human subject. In it, he describes the death of a female patient from uterine hemorrhage, and reflects that she “might very probably have been saved by transfusion . . . by means of the syringe” (Blundell “Experiments” 56-57). Following upon these reflections, Blundell undertook a series of experiments with dogs, draining them of blood until the point of what he terms “apparent death” (Blundell “Experiments” 63, 64, 74), and then reviving them by transfusion. In these experiments, Blundell established that arterial and venous blood were equally effective in restoring life; that small quantities of air could be introduced into the circulation without risk; and that blood would not be rendered unfit by its passage through the syringe. He also came to the conclusion that the prevailing view regarding the interchangeability of blood between different animals was mistaken; experiments in which dogs were transfused with human blood and sheep’s blood, though they could produce an immediate improvement, usually led to death within a few days. As a result, Blundell throughout his work in transfusion advocated that “in . . . transfusion on the human body, the human blood alone should be employed” (Blundell Researches 92).
originally posted by: soficrow
originally posted by: NavyDoc
originally posted by: soficrow
originally posted by: Char-Lee
a reply to: METACOMET
Drinking blood would do nothing as it would be destroying in the digesting of it. If it had an effect then so should cow blood when people eat rare meat.
Prion proteins are not destroyed in digestion - but they are more stable than most ordinary proteins. Still, GDF11 might survive digestion, be absorbed into the lymphatic system and from there, get into the blood where it's needed.
...More research required.
HMMM. Seems likely GDF11 misfolds in response to environmental influences - the misfolded form spreads as we age, and by the time we hit 25-40, it's just a numbers game with the misfolded form winning out. ....A prion-related post-transcription epigenetic mechanism.
Bets anyone?
But it's not a prion.
I did not say GDF11 is a prion - I was musing about whether or not it might be stable enough to survive digestion if ingested, and then, about whether or not it misfolds as we age. I also said more research is required.
If you have information on research into GDF11's normal and misfolded states, please post the links.
In any event, I stand by my speculation that GDF11 might misfold as we age and thus, lose it's rejuvenating power - perhaps more likely, an epigenetic mechanism may "turn off" the GDF11 gene, and prevent its expression.
Part of aging, is that various proteins are made in less quantities across the board. Every cell makes proteins that perform various tasks. If this protein is connected to activation of other cells or to turn off the signals that make other cells stop functioning, it could improve life length or quality but it would only be part of a cascade of signals.
Would drinking blood get the benefit? I doubt it as digestion denatures proteins and getting this particular protein into cells so that it can do its job is a lot more complicated than that. Synthesize an injectable form like HGH? That is very possible.
originally posted by: NavyDoc
originally posted by: soficrow
originally posted by: NavyDoc
originally posted by: soficrow
originally posted by: Char-Lee
a reply to: METACOMET
Drinking blood would do nothing as it would be destroying in the digesting of it. If it had an effect then so should cow blood when people eat rare meat.
Prion proteins are not destroyed in digestion - but they are more stable than most ordinary proteins. Still, GDF11 might survive digestion, be absorbed into the lymphatic system and from there, get into the blood where it's needed.
...More research required.
HMMM. Seems likely GDF11 misfolds in response to environmental influences - the misfolded form spreads as we age, and by the time we hit 25-40, it's just a numbers game with the misfolded form winning out. ....A prion-related post-transcription epigenetic mechanism.
Bets anyone?
But it's not a prion.
I did not say GDF11 is a prion - I was musing about whether or not it might be stable enough to survive digestion if ingested, and then, about whether or not it misfolds as we age. I also said more research is required.
If you have information on research into GDF11's normal and misfolded states, please post the links.
In any event, I stand by my speculation that GDF11 might misfold as we age and thus, lose it's rejuvenating power - perhaps more likely, an epigenetic mechanism may "turn off" the GDF11 gene, and prevent its expression.
Part of aging, is that various proteins are made in less quantities across the board. Every cell makes proteins that perform various tasks. If this protein is connected to activation of other cells or to turn off the signals that make other cells stop functioning, it could improve life length or quality but it would only be part of a cascade of signals.
Great overview of general information, but I'm interested in specifics on GDF11. Also, while proteins are made in less quantity as we age, they also misfold - and often, the diminished production results from misfolded proteins that govern gene expression and turn off the gene in question.
Again, if you have information on research into GDF11's normal and misfolded states, please post the links.
Would drinking blood get the benefit? I doubt it as digestion denatures proteins and getting this particular protein into cells so that it can do its job is a lot more complicated than that. Synthesize an injectable form like HGH? That is very possible.
I agree - and I'd also hate to see the general population get into the travesties the uber-wealthy have been conducting for the past several centuries. Best keep everyone ignorant, right? 'Cuz a little knowledge is dangerous, right? And mistaken assumptions made by a limited population of elite are preferable to mistakes made en masse by billions of plebs, right?
Got that everyone? Do NOT join a vampire cult! Hold out for the synthetic GDF11. And be prepared to pay through the nose for it.
A couple of experiments with GDF11 looked like this:
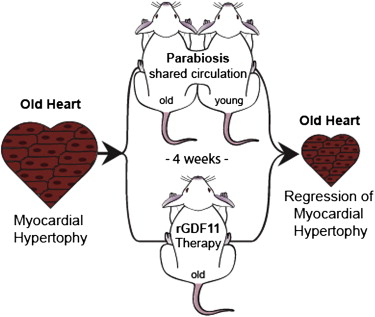
At the top, two mice were joined and had a shared circulatory system. One was old, the other young. The old mouse with a bad heart had it's heart improved. The old mouse generally got "younger". Interestingly, the young mouse got "older".
The bottom rGDF11 Therapy involved injecting an older mouse with GDF11 and it's heart also improved.
So I can see this a way to treat some types of heart failure and maybe Alzheimer's, simply by injecting GDF11.
Link
GDF11 stands for growth differentiation factor 11. It is in high concentrations when born and goes down as we age. This makes sense since we grow a lot after birth.
This is also scary. Can it cause cancer?
Link
Cancer cells produce more GDF11 than normal cells.
Ok, so we need to do more research to make sure we don't give ourselves cancer. But if you're about to die from heart failure, GDF11 treatment may be an option.
If we work out any problems, we could, eventually, turn on or increase the amount of GDF11 by placing the needed genetic material (DNA, RNA) in a virus and "infecting" the recipient.
Then we get into the moral ethics regarding immortality.

At the top, two mice were joined and had a shared circulatory system. One was old, the other young. The old mouse with a bad heart had it's heart improved. The old mouse generally got "younger". Interestingly, the young mouse got "older".
The bottom rGDF11 Therapy involved injecting an older mouse with GDF11 and it's heart also improved.
So I can see this a way to treat some types of heart failure and maybe Alzheimer's, simply by injecting GDF11.
Link
GDF11 stands for growth differentiation factor 11. It is in high concentrations when born and goes down as we age. This makes sense since we grow a lot after birth.
This is also scary. Can it cause cancer?
Quantitative real-time reverse transcription-PCR in colorectal cancer specimens obtained from 130 patients showed that GDF11 mRNA expression in cancer tissue was significantly higher than in normal tissue (p=0.001).
Link
Cancer cells produce more GDF11 than normal cells.
Ok, so we need to do more research to make sure we don't give ourselves cancer. But if you're about to die from heart failure, GDF11 treatment may be an option.
If we work out any problems, we could, eventually, turn on or increase the amount of GDF11 by placing the needed genetic material (DNA, RNA) in a virus and "infecting" the recipient.
Then we get into the moral ethics regarding immortality.
a reply to: ionwind
Here's more. What do you make of the contradictory results? [Aging effects on young mice v/s no aging effects on young mice]
Here's more. What do you make of the contradictory results? [Aging effects on young mice v/s no aging effects on young mice]
Parabiosis Points to GDF-11 as a Means to Reverse Age-Related Cardiac Hypertrophy
Parabiosis involves joining the circulatory systems of two animals. This is of interest for a number of studies in which old mice and young mice are linked together, known as heterochronic parabiosis. The young mice acquire a little of the metabolic, cellular, and gene expression changes characteristic of old mice, while in the the old mice some of these measures reverse towards more youthful levels. In stem cell activity in particular, the environment of signals present in the blood seems to dictate age-related decline as much as does any inherent damage to stem cells or their niches. This reinforces the view of stem cell aging as an evolved reaction to the cellular damage of aging that acts to extend life by reducing cancer risk, but at the cost of a slow decline into death due to ever more poorly maintained tissues and organs.
Heterochronic parabiosis studies in mice have been taking place for some years now, and researchers are beginning to link differences in gene expression and protein levels in old tissues versus young tissues to specific age-related conditions. The next logical step is to see if age-related dysfunction can be reversed by changing these protein levels in old animals:
Young blood reverses heart decline in old mice
Quoteumping young blood around old bodies - at least in mice - can reverse cardiac hypertrophy - the thickening and swelling of the heart muscle that comes with age and is a major cause of heart failure. After just four weeks, the older mouse's heart had reverted to almost the same size as that of its younger counterpart. The hearts of the young mice were unaffected, even though they were pumping some blood from the older mice.
After ruling out the effect of reduced blood pressure on the older mice, the team identified a potential candidate: a protein called GDF11, which was present in much higher quantities in the blood of the young mice. To test the effect of GDF11, the researchers gave old mice with cardiac hypertrophy daily injections of it for 30 days. At the end of the treatment, their hearts were significantly smaller than those in a second group of mice of the same age and with the same condition, but that had been injected with saline.
I recall reading about parabiosis (surgical union to allow sharing of the blood circulation) in classical novels - and concluded it was a fairly
common experimental procedure for the wealthy elites, using street urchins and slave children. Apparently, when it worked it was miraculous; when it
failed, the patients and donors died. This circumstance motivated a lot of research that culminated in the discovery of 'blood types.'
new topics
-
Ode to Artemis
General Chit Chat: 47 minutes ago -
Ditching physical money
History: 4 hours ago -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 4 hours ago -
Don't take advantage of people just because it seems easy it will backfire
Rant: 4 hours ago -
VirginOfGrand says hello
Introductions: 5 hours ago -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 6 hours ago -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 8 hours ago -
Geddy Lee in Conversation with Alex Lifeson - My Effin’ Life
People: 9 hours ago -
God lived as a Devil Dog.
Short Stories: 9 hours ago -
Police clash with St George’s Day protesters at central London rally
Social Issues and Civil Unrest: 11 hours ago
top topics
-
Hate makes for strange bedfellows
US Political Madness: 14 hours ago, 20 flags -
Who guards the guards
US Political Madness: 17 hours ago, 13 flags -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media: 8 hours ago, 12 flags -
Police clash with St George’s Day protesters at central London rally
Social Issues and Civil Unrest: 11 hours ago, 9 flags -
TLDR post about ATS and why I love it and hope we all stay together somewhere
General Chit Chat: 12 hours ago, 7 flags -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections: 6 hours ago, 5 flags -
One Flame Throwing Robot Dog for Christmas Please!
Weaponry: 4 hours ago, 4 flags -
Don't take advantage of people just because it seems easy it will backfire
Rant: 4 hours ago, 4 flags -
God lived as a Devil Dog.
Short Stories: 9 hours ago, 3 flags -
Ditching physical money
History: 4 hours ago, 3 flags
active topics
-
Hundreds of teenagers flood into downtown Chicago, smashing car windows
Other Current Events • 110 • : 777Vader -
Should Biden Replace Harris With AOC On the 2024 Democrat Ticket?
2024 Elections • 43 • : YourFaceAgain -
British TV Presenter Refuses To Use Guest's Preferred Pronouns
Education and Media • 125 • : Annee -
University student disciplined after saying veganism is wrong and gender fluidity is stupid
Education and Media • 22 • : ImagoDei -
Who guards the guards
US Political Madness • 4 • : kwaka -
New whistleblower Jason Sands speaks on Twitter Spaces last night.
Aliens and UFOs • 44 • : pianopraze -
"We're All Hamas" Heard at Columbia University Protests
Social Issues and Civil Unrest • 263 • : YourFaceAgain -
Remember These Attacks When President Trump 2.0 Retribution-Justice Commences.
2024 Elections • 50 • : Asher47 -
Ode to Artemis
General Chit Chat • 0 • : BrotherKinsMan -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 635 • : cherokeetroy
